��Ŀ����
��1��BCl3��NCl3����ԭ�ӵ��ӻ���ʽ�ֱ�Ϊ �� ����һ�����ܽ���B��N֮��ĵڶ�����Ԫ���� �֣�
��2����BCl3��XYmͨ��Bԭ����Xԭ�Ӽ����λ������γ����������������ṩ�¶Ե��ӵ�ԭ���� ��
��3��BF3��һ����ˮ�γɣ�H2O��2?BF3����Q��Q��һ�������¿�ת��ΪR��
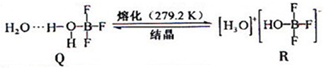
�پ���Q�и�����������������漰 ������ţ���
a�����Ӽ� b�����ۼ� c����λ�� d�����e�����»���
��R�������ӵĿռ乹��Ϊ �������ӵ�����ԭ�ӹ������ �ӻ���
��2����BCl3��XYmͨ��Bԭ����Xԭ�Ӽ����λ������γ����������������ṩ�¶Ե��ӵ�ԭ����
��3��BF3��һ����ˮ�γɣ�H2O��2?BF3����Q��Q��һ�������¿�ת��ΪR��

�پ���Q�и�����������������漰
a�����Ӽ� b�����ۼ� c����λ�� d�����e�����»���
��R�������ӵĿռ乹��Ϊ
���㣺ԭ�ӹ���ӻ���ʽ���ӻ������ж�,�жϼ��ӻ����ӵĹ���,�����ijɼ����
ר�⣺��ѧ���뾧��ṹ
��������1�����ݼ۲���ӶԻ�������ȷ��ԭ���ӻ���ʽ��ͬһ����Ԫ���У�Ԫ�ص�һ����������ԭ����������������������ƣ�����IIA�塢��VA��Ԫ�ص�һ�����ܴ�������Ԫ�أ�
��2�����йµ��ӶԺͺ��пչ����ԭ��֮�����γ���λ����Bԭ���к��пչ����
��3���ٷǽ���Ԫ��ԭ��֮�����γɹ��ۼ������пչ����ԭ�Ӻͺ��йµ��ӶԵ�ԭ��֮�����γ���λ��������֮����ڷ��»�����ˮ�����е�Oԭ�Ӻ����������е�Hԭ�����γ������
�ڸ��ݼ۲���ӶԻ�������ȷ��H3O+�ռ乹�͡�������������ԭ���ӻ���ʽ��
��2�����йµ��ӶԺͺ��пչ����ԭ��֮�����γ���λ����Bԭ���к��пչ����
��3���ٷǽ���Ԫ��ԭ��֮�����γɹ��ۼ������пչ����ԭ�Ӻͺ��йµ��ӶԵ�ԭ��֮�����γ���λ��������֮����ڷ��»�����ˮ�����е�Oԭ�Ӻ����������е�Hԭ�����γ������
�ڸ��ݼ۲���ӶԻ�������ȷ��H3O+�ռ乹�͡�������������ԭ���ӻ���ʽ��
���
�⣺��1��BCl3������Bԭ�Ӽ۲���ӶԸ���=3+
����3-3��1��=3�Ҳ����µ��Ӷԣ�����Bԭ�Ӳ���sp2�ӻ���NCl3������ԭ�Ӽ۲���ӶԸ���3+
����5-3��1��=4�Һ���һ���µ��Ӷԣ�����Nԭ�ӵ��ӻ���ʽΪsp3��
ͬһ����Ԫ���У�Ԫ�ص�һ����������ԭ����������������������ƣ�����IIA�塢��VA��Ԫ�ص�һ�����ܴ�������Ԫ�أ����Ե�һ�����ܽ���B��N֮��ĵڶ�����Ԫ����Be��C��O����Ԫ�أ�
�ʴ�Ϊ��sp2��sp3��3��
��2�����йµ��ӶԺͺ��пչ����ԭ��֮�����γ���λ����Bԭ���к��пչ��������Xԭ�����ṩ�µ��Ӷԣ��ʴ�Ϊ��X��
��3���ٷǽ���Ԫ��ԭ��֮�����γɹ��ۼ���Bԭ�Ӻ��пչ����Oԭ�Ӻ��йµ��Ӷԣ�����Bԭ�Ӻ�Oԭ��֮�������λ��������֮����ڷ��»�����ˮ�����е�Oԭ�Ӻ����������е�Hԭ�����γ���������Բ��漰�������Ӽ�����ѡa��
��H3O+��Oԭ�Ӽ۲���ӶԸ���=3+
����6-1-3��1��=4�Һ���һ���µ��Ӷԣ�����Ϊ�����νṹ��������������ԭ��Bԭ�Ӻ���4���Ҽ��Ҳ����µ��Ӷԣ�����Bԭ�Ӳ���sp3�ӻ���ʽ��
�ʴ�Ϊ�������Σ�sp3��
| 1 |
| 2 |
| 1 |
| 2 |
ͬһ����Ԫ���У�Ԫ�ص�һ����������ԭ����������������������ƣ�����IIA�塢��VA��Ԫ�ص�һ�����ܴ�������Ԫ�أ����Ե�һ�����ܽ���B��N֮��ĵڶ�����Ԫ����Be��C��O����Ԫ�أ�
�ʴ�Ϊ��sp2��sp3��3��
��2�����йµ��ӶԺͺ��пչ����ԭ��֮�����γ���λ����Bԭ���к��пչ��������Xԭ�����ṩ�µ��Ӷԣ��ʴ�Ϊ��X��
��3���ٷǽ���Ԫ��ԭ��֮�����γɹ��ۼ���Bԭ�Ӻ��пչ����Oԭ�Ӻ��йµ��Ӷԣ�����Bԭ�Ӻ�Oԭ��֮�������λ��������֮����ڷ��»�����ˮ�����е�Oԭ�Ӻ����������е�Hԭ�����γ���������Բ��漰�������Ӽ�����ѡa��
��H3O+��Oԭ�Ӽ۲���ӶԸ���=3+
| 1 |
| 2 |
�ʴ�Ϊ�������Σ�sp3��
���������⿼�������ӿռ乹�͵��жϡ�ԭ���ӻ���ʽ���жϡ���һ�����ܡ���ѧ����֪ʶ�㣬��Щ֪ʶ�㶼�Ǹ߿���Ƶ�㣬���ݼ۲���ӶԻ������ۡ�Ԫ�������ɵ�֪ʶ����������ɣ�ע���һ�����ܵĹ��ɼ��쳣����Ϊ�״��㣮

��ϰ��ϵ�д�
 ״Ԫ��ȫ��ͻ�Ƶ�����ϵ�д�
״Ԫ��ȫ��ͻ�Ƶ�����ϵ�д�
�����Ŀ
���������У���ȷ���ǣ�������
| A��H2SO4��Ħ��������98g |
| B����98gH2SO4�ܽ���500mLˮ�У�������Һ����������ʵ���Ũ��Ϊ2mol/L |
| C����������O2��O3����������ԭ������ͬ |
| D����״���£�1mol�κ����������Ϊ22.4L |
�ڴ������µ������£�C3H8�ܹ��������ѻ�����Ӧ����ʽ���£�C3H8��C2H4+CH4�������±��������ݣ�����˵����ȷ���ǣ�������
| ���ۼ����� | C-C | C=C | C-H |
| ����kJ/mol | 332 | 611 | 414 |
| A��C3H8��C2H4��CH4��Ϊͬϵ�� |
| B��C-C��C=C������ |
| C����1 mol C3H8��ȫ��Ӧ���ܹ�����53 kJ������ |
| D���������Ը�����س�ȥ��������е�C2H4���õ�������CH4 |
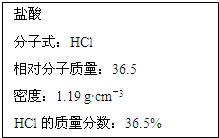 ��ͼΪʵ����ijŨ�����Լ�ƿ��ǩ�ϵ��й����ݣ��Ը��ݱ�ǩ�ϵ��й����ݻش��������⣺
��ͼΪʵ����ijŨ�����Լ�ƿ��ǩ�ϵ��й����ݣ��Ը��ݱ�ǩ�ϵ��й����ݻش��������⣺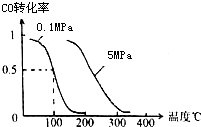 ��1����һ�������£��ݻ�Ϊ 100L�ܱ������з�����Ӧ��
��1����һ�������£��ݻ�Ϊ 100L�ܱ������з�����Ӧ��
