��Ŀ����
8����֪ij����ѩ��������Ҫ�ɷֵĻ�ѧʽΪXY2��X��Y��Ϊ���ڱ�ǰ20��Ԫ�أ�XԪ�ص�ԭ�Ӻ����20�ֲ�ͬ�˶�״̬�ĵ��ӣ�YԪ�ص�ԭ�Ӻ����5�ֲ�ͬ�ܼ��ĵ��ӣ���XY2�������Ӻ������ӵĵ��Ӳ�ṹ��ͬ����1������ѩ���Ļ�ѧʽ��CaCl2������ʽΪ��

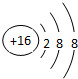
��2��Ԫ��D��Eԭ�ӵ�����������������Ӳ�����2����D��Y���ڣ���D�����ӽṹʾ��ͼ��
 ��D��E���γ�һ�ַǼ��Է��ӣ��÷��ӵĽṹʽΪS=C=S��E�ĵ����У����������Ե���ʯī����д�������ƣ���
��D��E���γ�һ�ַǼ��Է��ӣ��÷��ӵĽṹʽΪS=C=S��E�ĵ����У����������Ե���ʯī����д�������ƣ�����3��Ԫ��W��D��ͬһ���ڣ��䵥����ԭ�Ӿ��壻Ԫ��Z����̬���ʷ���Z2����3�����ۼ���W��Z���γ�һ���������ǽ������ϣ��仯ѧʽ������Si3N4��
��4��Ԫ��R��Yͬ���壬���⻯�������ڿ�ʴ������R2��NaOH��Һ��Ӧ�IJ���֮һ��OR2���÷�Ӧ�����ӷ���ʽΪ2F2+2OH-=2F-+OF2+H2O��
���� ��1��XԪ�ص�ԭ�Ӻ����20�ֲ�ͬ�˶�״̬�ĵ��ӣ���XΪCa���ɻ�ѧXY2��֪Y�Ļ��ϼ�Ϊ-1�ۣ�YԪ�ص�ԭ�Ӻ����5�ֲ�ͬ�ܼ��ĵ��ӣ���XY2�������Ӻ������ӵĵ��Ӳ�ṹ��ͬ�����Ӻ����������Ϊ18����Yԭ�Ӻ��������Ϊ17����YΪCl��
��2��Ԫ��D��Eԭ�ӵ�����������������Ӳ�����2����D����Ԫ�����ڣ���DΪ��Ԫ�أ�E��2�����Ӳ㣬����������Ϊ4����EΪ̼Ԫ�أ�
��3��Ԫ��W��D����ͬ���ڣ������ڵ������ڣ��䵥���ǰ뵼����ϣ���WΪSi��Ԫ��Z�ĵ��ʷ���Z2����3�����ۼ�����ZΪNԪ�أ�
��4��Ԫ��R��Y���ȣ�ͬ���壬���⻯�������ڿ�ʴ��������RΪCl��F2��NaOH��Һ��Ӧ�IJ���֮һ��OF2��������NaF��
��� �⣺��1��XԪ�ص�ԭ�Ӻ����20�ֲ�ͬ�˶�״̬�ĵ��ӣ���XΪCa���ɻ�ѧXY2��֪Y�Ļ��ϼ�Ϊ-1�ۣ�YԪ�ص�ԭ�Ӻ����5�ֲ�ͬ�ܼ��ĵ��ӣ���XY2�������Ӻ������ӵĵ��Ӳ�ṹ��ͬ�����Ӻ����������Ϊ18����Yԭ�Ӻ��������Ϊ17����YΪCl�������ѩ��ΪCaCl2������ʽΪ�� ��
��
�ʴ�Ϊ��CaCl2�� ��
��
��2��Ԫ��D��Eԭ�ӵ�����������������Ӳ�����2����D����Ԫ�����ڣ���DΪ��Ԫ�أ������ӽṹʾ��ͼΪ�� ��E��2�����Ӳ㣬����������Ϊ4����EΪ̼Ԫ�أ�D��E���γ�һ�ַǼ��Է��ӣ��÷��ӵĽṹʽΪS=C=S��E�ĵ����У����������Ե���ʯī��
��E��2�����Ӳ㣬����������Ϊ4����EΪ̼Ԫ�أ�D��E���γ�һ�ַǼ��Է��ӣ��÷��ӵĽṹʽΪS=C=S��E�ĵ����У����������Ե���ʯī��
�ʴ�Ϊ�� ��S=C=S��ʯī��
��S=C=S��ʯī��
��3��Ԫ��W��D����ͬ���ڣ������ڵ������ڣ��䵥���ǰ뵼����ϣ���WΪSi��Ԫ��Z�ĵ��ʷ���Z2����3�����ۼ�����ZΪNԪ�أ�Si��N�γ�һ���������ǽ�������ΪSi3N4��
�ʴ�Ϊ��Si3N4��
��4��Ԫ��R��Y���ȣ�ͬ���壬���⻯�������ڿ�ʴ��������RΪCl��F2��NaOH��Һ��Ӧ�IJ���֮һ��OF2��������NaF����Ӧ���ӷ���ʽΪ��2F2+2OH-=2F-+OF2+H2O��
�ʴ�Ϊ��2F2+2OH-=2F-+OF2+H2O��
���� ���⿼��ṹ����λ�ù�ϵ�����û�ѧ������ʽṹ�����ʵȣ��������ջ���֪ʶ��ע��OF2����Ԫ�ر��������ϼۣ����ڷ���������������

| ������ | IA | IIA | IIIA | IVA | VA | VIA | VIIA |
| 2 | a | c | e | g | |||
| 3 | b | d | f | h |
| A�� | a���ʵ��۵㡢�е��b���ʸ� | |
| B�� | ����Ԫ���У�bԪ�ص��ʵĻ�ѧ��������� | |
| C�� | ����Ԫ�صĵ����У������������ֹ��� | |
| D�� | c��d��f��h������������Ӧ��ˮ���������������ǿ |
��Al ��Al2O3 ��Al��OH��3 ��AlCl3��
| A�� | �٣��ڣ��ۣ��� | B�� | �ܣ��٣��ڣ��� | C�� | �ڣ���=�ۣ��� | D�� | �٣���=�ۣ��� |
| A�� | CH3CH2Cl | B�� | CH2Cl-CH2Cl | C�� | CH3-CH2OH | D�� | CH3 Cl |