��Ŀ����
14�����û�ѧ��Ӧԭ���о�±��Ԫ�ص��й����ʾ�����Ҫ���壮��1��Cl2�ڹ�ҵ�����ż���㷺����;����ҵ��ͨ���ȼҵ����Cl2���䷴Ӧ�����ӷ���ʽΪ2Cl-+2H2O$\frac{\underline{\;���\;}}{\;}$2OH-+Cl2��+H2���������£��ö��Ե缫���100mL����ʳ��ˮ��������������״����11.2mL���壬������Һ����ı仯ʱ��������Һ��pH=12��
��2�����ú��⻯�صĵ�ҩƬ�ɿ��˷��䣮����ҩƬ��ȥ����Ƥ����������Թ����Ƶ���ɫˮ��Һ�������뼸�ε�����Һ��Ȼ�����Թ�����μ�����ˮ���۲쵽��Һ����ɫ��Ϊ��ɫ���������μ���ˮ����ɫ��Һ����Ϊ��ɫ����ɲ���ƽ��Һ����ɫ��Ϊ��ɫ�Ļ�ѧ����ʽ�������ѧ��������1��������д����5Cl2+I2+6H2O=2HIO3+10HCl��
���� ��1����ҵ�õ�ⱥ��ʳ��ˮ�ķ����Ʊ�������������������״����11.2mL���壬������0.0005mol���������ݷ���ʽ��������NaOH�����ʵ���������������Һ��pH��
��2�������μ���ˮ����ɫ��Һ����Ϊ��ɫ����������������ǿ�����ԣ�����ⷢ��������ԭ��Ӧ��������͵��ᣮ
��� �⣺��1����ҵ�õ�ⱥ��ʳ��ˮ�ķ����Ʊ���������Ӧ�ķ���ʽΪ2Cl-+2H2O$\frac{\underline{\;���\;}}{\;}$ 2OH-+Cl2��+H2����������������״����11.2mL���壬������0.0005mol������������NaOH0.001mol��c��OH-��=0.001mol��0.1L=0.01mol/L��c��H+��=10-12mol/L����pH=12��
�ʴ�Ϊ��2Cl-+2H2O$\frac{\underline{\;���\;}}{\;}$ 2OH-+Cl2��+H2����12��
��4�������μ���ˮ����ɫ��Һ����Ϊ��ɫ����������������ǿ�����ԣ�����ⷢ��������ԭ��Ӧ��������͵��ᣬIԪ�صĻ��ϼ���0����Ϊ+5�ۣ�ClԪ�صĻ��ϼ���0����Ϊ-1�ۣ���ԭ�ӡ������غ��֪����ʽΪ5Cl2+I2+6H2O�T2HIO3+10HCl��
�ʴ�Ϊ��5Cl2��I2��6H2O��2��10HCl��
���� ���⿼�����������ʣ�Ϊ��Ƶ���㣬�������ʵ����ʡ������ķ�Ӧ������Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬ע���⼰������ԭ��Ӧ��Ӧ�ã���Ŀ�ѶȲ���

 һ����ʦ�����Ծ�ϵ�д�
һ����ʦ�����Ծ�ϵ�д�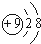 ��NH4+ ��O2-���������������ͬ���ǣ�������
��NH4+ ��O2-���������������ͬ���ǣ�������| A�� | �ڢۢ� | B�� | �٢ڢۢ� | C�� | �٢ڢ� | D�� | �٢ۢ� |
| A�� | �٢ڢۢ� | B�� | �٢ۢ� | C�� | �ۢ� | D�� | �٢ڢ� |
| A�� | Na��Mg��Al�ĵ�һ������������ | B�� | ���ȶ��ԣ�HF��H2O��NH3 | ||
| C�� | S2-��Cl-��K+�İ뾶������ | D�� | O��F��Ne�ĵ縺�������� |