��Ŀ����
11��Ϊ�˽��軷���Ѻ�����ᣬ���ܼ��Ž���Ϊ�ҹ�����Ŀǰ�������ص㣬��˽�Լ��Դ�뽨������Դ�ǰ��ڵ�ǰ��һ�����⣮�����һ����ij��ѧѧϰ�о���С��������·���������Ϊ���������ǣ�������| A�� | �о���ú�������¼�������߲��������㹤ҵ�����Ŀ��ٷ�չ | |
| B�� | �Ӵ�̫���ܡ�ˮ�ܡ����ܡ������ܵ���Դ���ȣ����ٻ�ʯȼ�ϵ�ʹ�� | |
| C�� | �����ƹ����Ҵ�Ϊȼ�ϵ�����������ƹ�綯���� | |
| D�� | �����������У�Ŭ��ʵ����Դ��ѭ������ |
���� ���������Ϣ��֪�����軷���Ѻ�����ᣬӦ���Ͻ��ܼ��ŵĻ�ѧ��������ɫ��ѧ�ĺ�����
��� �⣺A��ú��ʯ��ʹ��Խ�࣬�Ի�����ȾԽ���������⣬��Aѡ��
B��̫���ܡ�ˮ�ܡ����ܵ��������Դ���ܼ���ʹ�û�ʯȼ�ϣ�������Ⱦ����ŷţ��������⣬��B��ѡ��
C�������ƹ����Ҵ�Ϊȼ�ϵ�����������ƹ�綯���������Լ��ٻ�ʯȼ�ϵ�ȼ�գ�������Ⱦ����ŷţ��������⣬��C��ѡ��
D����Դ��ѭ�����ã���ѭ��Լ��Դ�������˷ѵ�������Ͻ��ܽ��ĺͱ�������Ŀ�ģ���D��ѡ��
��ѡ��A��
���� ���⿼���˻�����Ⱦ��������Ⱦ�Ĵ�������Ŀ�ѶȲ�����ʱӦ���ս��ܽ��ĺͱ���������˼�룬ע������֪ʶ�Ļ��ۣ�

��ϰ��ϵ�д�
�����Ŀ
1�� �绯ѧ���������������ڼ�����NH3�ĺ������乤��ԭ����ͼ��ʾ��NH3������Ϊ���������ʣ�����˵��������ǣ�������
�绯ѧ���������������ڼ�����NH3�ĺ������乤��ԭ����ͼ��ʾ��NH3������Ϊ���������ʣ�����˵��������ǣ�������
 �绯ѧ���������������ڼ�����NH3�ĺ������乤��ԭ����ͼ��ʾ��NH3������Ϊ���������ʣ�����˵��������ǣ�������
�绯ѧ���������������ڼ�����NH3�ĺ������乤��ԭ����ͼ��ʾ��NH3������Ϊ���������ʣ�����˵��������ǣ�������| A�� | ��Һ��K+��缫b�ƶ� | |
| B�� | ��Ӧ���ĵ�NH3��O2�����ʵ���֮��Ϊ4��5 | |
| C�� | �����ڵ缫a�Ϸ���������Ӧ | |
| D�� | �����ĵ缫��ӦʽΪ��O2+4e-+2H20=4OH- |
2�����������У�����������ʵ��ǣ�������
| A�� | CH3CH2OH | B�� | NaNO3 | C�� | NH3•H2O | D�� | H2SO4 |
19��W��X��Y��Z��Ϊ������Ԫ�أ�W������������������������֮��Ϊ7��17��X��Wͬ���壻Y��ԭ��������W��X��ԭ������֮�͵�һ�룻��ZԪ�ص�������ɫ��ӦΪ��ɫ�������ж���ȷ���ǣ�������
| A�� | �����ԣ�Y��Z | B�� | ��һ�����ܣ�X��W | ||
| C�� | ���ӵĻ�ԭ�ԣ�X��W | D�� | ԭ�Ӽ����Ӱ뾶��Z��Y��X |
6����50mL0.1moL/LNa2CO3��Һ�е������ӵ����ʵ���Ũ����ͬ����Һ�ǣ�������
| A�� | 50mL0.2moL/L��NaCl��Һ | B�� | 100mL0.1moL/L��NaCl��Һ | ||
| C�� | 25mL0.2moL/L��Na2SO4��Һ | D�� | 10mL0.5moL/L��Na2CO3��Һ |
16���Ʊ���������茶����ʵ���У������й�˵�����������ǣ�������
| A�� | ���ձ������ȵı���̼������Һϴ����м�������������벢ϴ����м | |
| B�� | ���裬С����Ȼ����Һ����������������Ũ������Һ������־�ĤΪֹ | |
| C�� | ���˺��ñ�ˮϴ�Ӿ�����渽�ŵ����� | |
| D�� | �ᾧǰ����ҺŨ�Ƚ�С�����ڵõ��ϴ��������������茶��� |
20������˵������ȷ���ǣ�������
| A�� | �����ʵ���Ũ�ȵģ�NH4��2CO3��Һ�ͣ�NH4��2SO4��Һ��c��NH4+����ͬ | |
| B�� | FeCl3��Һ���������CaCO3������Ӧ | |
| C�� | һ�������£���Ӧ2Mg��s��+CO2��g���TC��s��+2MgO��s�������Է��ԣ����H��0 | |
| D�� | ��0.005mol Na2O2����ˮ�õ�1L��Һ������Һ�ڳ����µ�pH=12 |
16������ʵ������������ȷ���ǣ�������
| A�� | Ϊ�ⶨ������ˮ��pHֵ���ò�����պȡ����Һ����pH��ֽ�ϣ����ɫ�����ռ��� | |
| B�� | ������ʵ��ʱ�������Һ�����ǰ�������Ǽӷ�ʯ��Ӧ����ֹͣ���ȣ���ȴ�ӷ�ʯ | |
| C�� | ��δ֪Һ�еμ�BaCl2��Һ���ְ�ɫ��������ϡ���ᣬ�������ܽ⣬˵����δ֪Һ�д���SO32-��SO42- | |
| D�� | �ᴿ������������ص��Ȼ��ƣ������ڽϸߵ��¶����Ƴ�Ũ��Һ����ȴ�ᾧ�����ˡ�����ķ��� |
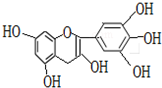 �����ҹ��Ĵ�ͳ��Ʒ����Ҷ�к��еIJ��ӿ������ʳƷ���Ӽ��ж������к��ĺϳɿ������������ڶ���ʳƷ���ʵȣ���ͼ1��ʾ�Dz����к�����ߵ�һ�ֶ�����A�Ľṹ��ʽ���������ֶ�����A���й�������ȷ���ǣ�������
�����ҹ��Ĵ�ͳ��Ʒ����Ҷ�к��еIJ��ӿ������ʳƷ���Ӽ��ж������к��ĺϳɿ������������ڶ���ʳƷ���ʵȣ���ͼ1��ʾ�Dz����к�����ߵ�һ�ֶ�����A�Ľṹ��ʽ���������ֶ�����A���й�������ȷ���ǣ�������