��Ŀ����
7�����и���������ָ����Һ���ܴ���������� ��?������ɫ��Һ�У�K+��Cu2+��Na+��MnO4-��SO42-
��ʹpH=11����Һ�У�CO32-��Na+��AlO2-��NO3-
�ۼ���Al�ܷų�H2����Һ�У�Cl-��HCO3-��NO3-��NH4+
������ˮ�������c��OH-��=10-13mol•L-1����Һ�У�Na+��Ba2+��Cl-��I-
����ʹpH��ֽ��Ϊ��ɫ����Һ��Na+��C1-��S2-��SO32-
��������Һ�У�Fe2+��Al3+��NO3-��I-��Cl-��
| A�� | �٢ڢ� | B�� | �٢ۢ� | C�� | �ڢܢ� | D�� | �٢ڢ� |
���� ����ɫ��Һ�в�����ɫ���������ӡ�����������ӣ�
��pH=11����Һ�ʼ��ԣ�
�ۼ���Al�ܷų�H2����ҺΪǿ����Һ���ǿ����������Һ��
����ˮ�������c��OH-��=10-13mol•L-1����Һ�����Ի���ԣ�
����ʹpH��ֽ��Ϊ��ɫ����ҺΪ������Һ����������֮�䲻��Ӧ�����������������ӷ�Ӧ��
��������Һ�в�����������ܹ����������ӣ�
��� �⣺��Cu2+��MnO4-Ϊ��ɫ���ӣ�����Һ�в��ܴ������棬�ʢٴ���
��pH=11����Һ�ʼ��ԣ�CO32-��Na+��AlO2-��NO3-֮�䲻��Ӧ�����������������ӷ�Ӧ�������ܴ������棬�ʢ���ȷ��
�ۼ���Al�ܷų�H2����ҺΪǿ����Һ���ǿ����������Һ�����Ի���������¶����ܴ�������HCO3-�����������²��ܴ�������NH4+���ʢ۴���
����ˮ�������c��OH-��=10-13mol•L-1����Һ��ǿ���Ի�ǿ���ԣ�Na+��Ba2+��Cl-��I- ֮�䲻��Ӧ�Һ������ӻ����������Ӷ�����Ӧ�������ܴ������棬�ʢ���ȷ��
����ʹpH��ֽ��Ϊ��ɫ����Һ�д��ڴ������������ӣ�Na+��C1-��S2-��SO32- ֮�䲻��Ӧ�����������������ӷ�Ӧ������Һ���ܹ��������棬�ʢ���ȷ��
��������Һ�У�Fe2+��I-��NO3-����������ԭ��Ӧ�����ܴ������棬�ʢ���
��ѡC��
���� ���⿼�����ӹ��棬Ϊ��Ƶ���㣬��Ŀ�Ѷ��еȣ����ؿ��鸴�ֽⷴӦ��������ԭ��Ӧ����ȷ�������ʼ����ӹ��������ǽⱾ��ؼ���ע�������������ھ�����������ѧ�������Ӧ��������

 �������¿��ÿ�ʱ��ҵϵ�д�
�������¿��ÿ�ʱ��ҵϵ�д� Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�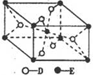 ��֪A��B��C��D��E����Ԫ�ص�ԭ���������ε�����A��B��C��Dλ�ڶ����ڣ�A��ԭ�Ӱ뾶��С��Ԫ�أ�B�Ļ�̬ԭ���е���ռ������������ͬ��ԭ�ӹ������ÿ�ֹ���еĵ���������ͬ��Dԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������3����E�С����������֮�ƣ�E4+���ԭ�ӵĺ�������Ų���ͬ��A��B��D����Ԫ����ɵ�һ�ֻ�����M����װ�����г����е�һ���к����壬A��B����Ԫ����ɵ�ԭ�Ӹ���֮��Ϊ1��1�Ļ�����N�dz������л��ܼ���
��֪A��B��C��D��E����Ԫ�ص�ԭ���������ε�����A��B��C��Dλ�ڶ����ڣ�A��ԭ�Ӱ뾶��С��Ԫ�أ�B�Ļ�̬ԭ���е���ռ������������ͬ��ԭ�ӹ������ÿ�ֹ���еĵ���������ͬ��Dԭ�ӵĺ���ɶԵ�������δ�ɶԵ�������3����E�С����������֮�ƣ�E4+���ԭ�ӵĺ�������Ų���ͬ��A��B��D����Ԫ����ɵ�һ�ֻ�����M����װ�����г����е�һ���к����壬A��B����Ԫ����ɵ�ԭ�Ӹ���֮��Ϊ1��1�Ļ�����N�dz������л��ܼ�����ش��������⣨����ʱ��A��B��C��D��E������Ӧ��Ԫ�ط��ű�ʾ����
��1��A2D2���ӵĵ���ʽΪ
 ��E�Ļ�̬ԭ�ӵ���Χ�����Ų�ʽΪ3d24s2��
��E�Ļ�̬ԭ�ӵ���Χ�����Ų�ʽΪ3d24s2����2��B��C��D����Ԫ�صĵ�һ��������С�����˳��ΪC��O��N��
��3������B��C������������ˮ�����Ũ��Һ�ȷ�Ӧ���仯ѧ����ʽΪC+4HNO3��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CO2��+4NO2��+2H2O
��4������������ȷ����ad������ţ���
a�� M�Ǽ��Է��ӣ�N�ǷǼ��Է��� b�� M��BD2�����е�����ԭ�Ӿ�����sp2�ӻ�
c�� N�����к���6���Ҽ���1���м� d�� BD2������۵㡢�е㶼�ȶ������辧��ĵ�
��5����֪����E��һ��������Q���侧���ṹ��ͼ��ʾ
��Q��s��+2Cl2��g���TECl4��l��+D2��g����H=+140kJ/mol
��2B��s��+D2��g���T2BD��g����H=-221kJ/mol
д������Q�ͽ�̿��������Ӧ����Һ̬ECI4��BD������Ȼ�ѧ����ʽ��TiO2��s��+2Cl2��g��+2C��s��=TiCl4��l��+2CO��g����H=-81kJ/mol
��6����0.5L���ܱ������У�һ������C2��A2�������»�ѧ��Ӧ��C2��g��+3A2��g���T2CA3��g����H��0���仯ѧƽ�ⳣ��K���¶�t�Ĺ�ϵ���±�����ش��������⣮
| t/�� | 200 | 300 | 400 |
| K | K1 | K2 | 0.5 |
����400��ʱ�������CA3��C2��A2�����ʵ����ֱ�Ϊ3mol��1mol��2molʱ����÷�Ӧ�� V��C2��������C2���棨��д����������=����������
| A�� | ��40g NaOH�ܽ���1Lˮ�� | |
| B�� | ��25g ��������ˮ���100mL CuSO4��Һ | |
| C�� | ��0.1L 10 mol•L-1Ũ�������0.9Lˮ�� | |
| D�� | ��22.4L HCl��������ˮ���1L��Һ |
| A�� | FeSO4��Fe��NO3��3�� Fe2��SO4��3��HNO3��N2O��H2O | |
| B�� | HNO3��FeSO4�� Fe2��SO4��3��Fe��NO3��3��N2O��H2O | |
| C�� | HNO3��Fe2��SO4��3�� FeSO4��Fe��NO3��3��N2O��H2O | |
| D�� | Fe2��SO4��3��N2O�� FeSO4��HNO3��Fe��NO3��3��H2O |
| A�� | ������ˮ | B�� | ��ҵ�̵� | C�� | ��ˮ��þ | D�� | ��س�� |
�ٵ�һ�ݼӹ���������������Һ����ȣ��ռ���0.02mol�����壬����������ͬʱ�õ���Һ�ף�
������Һ����ͨ�����Ķ�����̼���壬���ɰ�ɫ���������������ˣ�ϴ�����պõ�1.02g���壮
�۵ڶ��ݼ��������Ȼ�����Һ�����ɰ�ɫ������������������ϴ�ӣ�����õ�11.65g���壮
�ݴˣ���ͬѧ�õ��Ľ�����ȷ���ǣ�������
| A�� | ʵ����в���������Ϊ���������ɵ�ԭ��Һ��c��NH4+��=0.2 mol•L-1 | |
| B�� | ʵ����еİ�ɫ������һ����BaSO4����BaCO3 | |
| C�� | ԭ��Һ��һ����K+����c��K+��=0.4 mol•L-1 | |
| D�� | ��Ҫ�ж�ԭ��Һ���Ƿ���Cl-�������������ʵ����֤ |
| A�� | $\frac{3}{��m-n��}$mL | B�� | $\frac{��m-n��}{3}$ mL | C�� | $\frac{��4m-1��}{13}$ mL | D�� | $\frac{��m-n��}{3}$3 mL |
 ij����С������ͼװ�ý���ʵ�飬�Իش��������⣺
ij����С������ͼװ�ý���ʵ�飬�Իش��������⣺