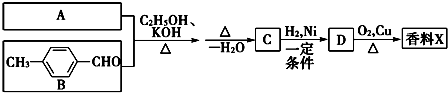
��Ŀ����
ij��ѧ�о���ѧϰС�����һ�����ԭ�ϣ������ֳ����Ķ����ڽ���������ɵĺϽ��ĩ������̽����ʵ���¼���£�
��֪����Mg2+�ļ��鷽����ȡ2�δ���Һ����2��2mol?L-1��NaOH��Һ���ټ���1��þ�Լ�����Ⱦ�ϣ���������ż�������ӣ��������ֳ���������þ�Լ�����Ⱦ�ϳ�����ɫ����ʾ����Һ�к���Mg2+��
��Ag+��Cu2+��Fe3+��NH4+�����Mg2+�����
��Mg��OH��2�������Ȼ��Ũ��Һ�����������IJ��ܼ��������Ȼ�泥�
�ܺܶ�Ͻ��ǽ����������ȷ���Ļ�ѧ��ɣ�
�ش��������⣺
��1���Լ�XΪ ���ѧʽ����
��2�������ԭ������ �� ���ֽ�����ɵĺϽ�
��3��ʵ��ʱ����ҺB�м�����������������Һ������У���е�Ŀ���� ��
��4��ȡ17.7g�Ͻ��������ϡ���������19.04L����״���£����壬�úϽ���һ�ֽ���������仯ѧʽΪ ��

��֪����Mg2+�ļ��鷽����ȡ2�δ���Һ����2��2mol?L-1��NaOH��Һ���ټ���1��þ�Լ�����Ⱦ�ϣ���������ż�������ӣ��������ֳ���������þ�Լ�����Ⱦ�ϳ�����ɫ����ʾ����Һ�к���Mg2+��
��Ag+��Cu2+��Fe3+��NH4+�����Mg2+�����
��Mg��OH��2�������Ȼ��Ũ��Һ�����������IJ��ܼ��������Ȼ�泥�
�ܺܶ�Ͻ��ǽ����������ȷ���Ļ�ѧ��ɣ�
�ش��������⣺
��1���Լ�XΪ
��2�������ԭ������
��3��ʵ��ʱ����ҺB�м�����������������Һ������У���е�Ŀ����
��4��ȡ17.7g�Ͻ��������ϡ���������19.04L����״���£����壬�úϽ���һ�ֽ���������仯ѧʽΪ

���㣺̽�����ʵ���ɻ�������ʵĺ���
ר�⣺
��������Ͻ������ᷴӦ����ҺA�н��백ˮ�����ɰ�ɫ������������ҺB������������Һ��к����þ�Լ�����Ⱦ�Ϻ���������ɫ������֪���Ͻ���һ������Mg����ɫ�����м�����������X��Һ��������þ�ܽ⣬�����ɵij����ܹ��ܽ����������ƺ����ᣬ˵���ó���Ϊ����������ԭ�Ͻ���һ������Al�������Ϣ��֪Xһ��ΪNH4Cl��
��1����Ϸ����ж�X�����Ƽ���ѧʽ��
��2�����ݷ����жϸúϽ����ɣ�
��3���������Ϣ��֪Mg��OH��2������Ũ���Ȼ����Һ��Ӧ�ų�NH4+�ĸ��ţ�
��4�����þ���������ʵ�����Ȼ���������������������������ʽ���㣬Ȼ�������úϽ�Ļ�ѧʽ��
��1����Ϸ����ж�X�����Ƽ���ѧʽ��
��2�����ݷ����жϸúϽ����ɣ�
��3���������Ϣ��֪Mg��OH��2������Ũ���Ȼ����Һ��Ӧ�ų�NH4+�ĸ��ţ�
��4�����þ���������ʵ�����Ȼ���������������������������ʽ���㣬Ȼ�������úϽ�Ļ�ѧʽ��
���
�⣺��Ͻ������ᷴӦ����ҺA�н��백ˮ�����ɰ�ɫ������������ҺB������������Һ��к����þ�Լ�����Ⱦ�Ϻ���������ɫ������֪���Ͻ���һ������Mg����ɫ�����м�����������X��Һ��������þ�ܽ⣬�����ɵij����ܹ��ܽ����������ƺ����ᣬ˵���ó���Ϊ����������ԭ�Ͻ���һ������Al�������Ϣ��֪Xһ��ΪNH4Cl��
��1�����ݷ�����֪��XΪNH4Cl��
�ʴ�Ϊ��NH4Cl��
��2���������Ϸ�����֪���úϽ�ΪMg��Al�Ͻ�
�ʴ�Ϊ��������Al����þ����Mg����˳��ɻ�������
��3������Mg��OH��2������Ũ���Ȼ����Һ�������þ�Լ�����Ⱦ�ϼ���Mg2+ʱҪ�ų�NH4+�ĸ��ţ�������Һ������������������Һ��NH4+ת��ΪNH3����н������ϳ���
�ʴ�Ϊ���ٽ�NH3�ݳ�����ֹNH4+����Mg2+�ļ��飻
��4��17.7g�Ͻ��������ϡ���������19.04L����״���£����壬��Ͻ��к���xmolMg��ymolAl�����ݺϽ�������ɵã�24x+27y=17.7�������������������ʵ�����֪��x+1.5y=
=0.85��������ã�x=0.4��y=0.3���Ͻ���Mg��Al�����ʵ���֮��Ϊ��0.4mol��0.3mol=4��3�����ԸúϽ�Ļ�ѧʽΪ��Mg4Al3��
�ʴ�Ϊ��Mg4Al3��
��1�����ݷ�����֪��XΪNH4Cl��
�ʴ�Ϊ��NH4Cl��
��2���������Ϸ�����֪���úϽ�ΪMg��Al�Ͻ�
�ʴ�Ϊ��������Al����þ����Mg����˳��ɻ�������
��3������Mg��OH��2������Ũ���Ȼ����Һ�������þ�Լ�����Ⱦ�ϼ���Mg2+ʱҪ�ų�NH4+�ĸ��ţ�������Һ������������������Һ��NH4+ת��ΪNH3����н������ϳ���
�ʴ�Ϊ���ٽ�NH3�ݳ�����ֹNH4+����Mg2+�ļ��飻
��4��17.7g�Ͻ��������ϡ���������19.04L����״���£����壬��Ͻ��к���xmolMg��ymolAl�����ݺϽ�������ɵã�24x+27y=17.7�������������������ʵ�����֪��x+1.5y=
| 19.04L |
| 22.4L/mol |
�ʴ�Ϊ��Mg4Al3��
���������⿼�����ʺ����IJⶨ����Ŀ�Ѷ��еȣ���ȷ�ⶨ���̼�������Ӧԭ��Ϊ���ؼ�����������ڿ���ѧ����ʵ�������ͷ�������������������ѧ�����Ӧ�û���֪ʶ��������

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
���з�Ӧ�У�������������ԭ��Ӧ���ǣ�������
| A��3Cl2+6KOH�T5KCl+KClO3+3H2O |
| B��2NO2ʮ2NaOH�TNaNO3+NaNO2+H2O |
| C��SnCl4+2H2O�TSnO2+4HCl |
| D��3CCl4+K2Cr2O7�T2CrO2Cl2+3COCl2+2KCl |
���л�ѧ����ʽ�����ӷ���ʽ�У���д��ȷ���ǣ�������
| A����������Һ�м����������CH3COONa+H+-��CH3COOH+Na+ | |||
B����ȩ��������Ӧ��CH3CHO+2Ag��NH3��2OH
| |||
| C����������Һ��ͨ������������̼��2C6H5O-+CO2+H2O��C6H5OH+2CO32- | |||
D����ϩ�ۺϣ�nCH2=CHCH3
 |
����ʽΪC10H14�����ڱ���ͬϵ��ұ����ϵ�һ�ȴ���ֻ��2�ֵ��л����У������������칹��������������3���⣩��������
| A��5�� | B��6�� | C��7�� | D��8�� |
���䡰������ʱ���£�N2H4����Ϊ�����������ȼ�ϣ�NO2Ϊ����������Ӧ����N2��ˮ��������֪��N2��g��+2O2��g��=2NO2��g������H=+67.7kJ/mol��N2H4��g��+O2��g��=N2��g��+2H2O��g������H=-534kJ/mol
���й����º�NO2��Ӧ���Ȼ�ѧ����ʽ�У���ȷ���ǣ�������
���й����º�NO2��Ӧ���Ȼ�ѧ����ʽ�У���ȷ���ǣ�������
| A��2N2H4��g��+2NO2��g��=3N2��g��+4H2O��l������H=-1135.7kJ/mol | ||
| B��2N2H4��g��+2NO2��g��=3N2��g��+4H2O��g������H=-1000.3kJ/mol | ||
C��N2H4��g��+NO2��g��=
| ||
| D��2N2H4��g��+2NO2��g��=3N2��g��+4H2O��g������H=-1135.7kJ/mol |
���й������������У�����ȷ���ǣ�������
| A�����ǵؿ��ﺬ�����Ľ���Ԫ�� |
| B��������ʧȥ���ӣ����ֻ�ԭ�� |
| C���ڿ������þƾ��Ƶ㲻�� |
| D���ڳ����£���������������Ӧ |
 ��֪��A��B��C��D��E��F����Ԫ�أ�ԭ��������������Aԭ�Ӻ�����������״�ĵ����ƣ�������״�ĵ����ƹ���ϵĵ�������ȣ�B�Ƕ�������ԭ�Ӱ뾶����Ԫ�أ�CԪ�ص�ԭ��3p�ܼ��������E���������ڵ縺������Ԫ�أ�F�ǵ�������δ�ɶԵ�������Ԫ�أ��Իش������йص����⣺
��֪��A��B��C��D��E��F����Ԫ�أ�ԭ��������������Aԭ�Ӻ�����������״�ĵ����ƣ�������״�ĵ����ƹ���ϵĵ�������ȣ�B�Ƕ�������ԭ�Ӱ뾶����Ԫ�أ�CԪ�ص�ԭ��3p�ܼ��������E���������ڵ縺������Ԫ�أ�F�ǵ�������δ�ɶԵ�������Ԫ�أ��Իش������йص����⣺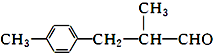
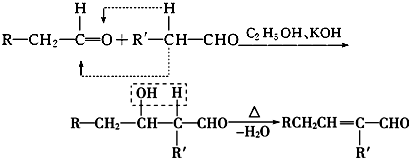 ��R-����������
��R-����������