��Ŀ����
16��Ԫ�������ɽ�ʾ��Ԫ�ؼ�ĵݱ���ɣ����еݱ������ȷ���ǣ�������| A�� | ��A��Ԫ���⻯��е㣺SiH4��CH4�����Ԣ�A��Ԫ���⻯��е㣺PH3��NH3 | |
| B�� | �ڶ�����Ԫ���⻯���ȶ��ԣ�HF��H2O����������Ԫ���⻯���ȶ��ԣ�HCl��H2S | |
| C�� | ��A��Ԫ�صķǽ����ԣ�F��Cl�����Ԣ�A��Ԫ���⻯������ԣ�HF��HCl | |
| D�� | þ�������ã���ҵ���õ���������������������Թ�ҵ��Ҳ�õ����������þ��þ |
���� A���������Ӽ京������е�ߣ�
B���ǽ�����Խǿ����Ӧ�⻯��Խ�ȶ���
C���������÷ǽ����ԱȽ϶�Ӧ�⻯������ԣ�
D������þ���۵�ߣ�Ӧѡ��������Ȼ�þұ��Mg��
��� �⣺A���������Ӽ京������е�ߣ����A��Ԫ���⻯��е㣺PH3��NH3����A����
B���ǽ�����Խǿ����Ӧ�⻯��Խ�ȶ�����ڶ�����Ԫ���⻯���ȶ��ԣ�HF��H2O����������Ԫ���⻯���ȶ��ԣ�HCl��H2S����B��ȷ��
C����A��Ԫ�صķǽ����ԣ�F��Cl��Ԫ���⻯������ԣ�HF��HCl�����߲����ֵݱ���ɣ���C����
D������þ���۵�ߣ�Ӧѡ��������Ȼ�þұ��Mg��ҵ���õ��������������������D����
��ѡB��
���� ���⿼��Ԫ�����ڱ��������ɣ�Ϊ��Ƶ���㣬����Ԫ�ص�λ�á����ʡ�Ԫ��������Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬ע�������֪ʶ��Ӧ�ã���Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
11����NA��ʾ�����ӵ�������ֵ������������ȷ���ǣ�������
| A�� | 1molCl2��Fe����ȫ��Ӧʱת�Ƶ�����Ϊ2NA | |
| B�� | ����������ˮ��Ӧʱ��ÿ����1molO2��ת��4mol���� | |
| C�� | ��״���£�11.2L�������к��з�ԭ�ӵ���ĿΪ0.5NA | |
| D�� | 0.1 mol•L-1NaHSO4��Һ�У���������Ŀ֮��Ϊ0.3NA |
1��X��Y��Z��WΪ����������Ԫ�أ�Xԭ��M����1�����ӣ�Yԭ������������ΪΪ���ڲ��������2����ZԪ�ص�����ϼ�Ϊ������ϼ۾���ֵ��3����Z��Wͬ���ڣ�W�� ԭ�Ӱ뾶С��Z������������ȷ���ǣ�������
| A�� | ԭ�Ӱ뾶�ɴ�С��˳��Z��X��Y | |
| B�� | Ԫ�طǽ�������ǿ������˳��W��Z��Y | |
| C�� | ����̬�⻯����ȶ���ǿ��˳��Z��W | |
| D�� | W�ֱ���X��Y�γɵĻ����ﻯѧ��������ͬ |
8��ij��̬��������̬��ϩ����ɵĻ��������ͬ��ͬѹ�¶�����������ܶ�Ϊ13��ȡ��״���´˻������4.48Lͨ��������ˮ�У���ˮ����2.8g�����������ǣ�������
| A�� | ����ͱ�ϩ | B�� | �����2-��ϩ | ||
| C�� | �����2-����ϩ | D�� | ��ϩ��1-��ϩ |
5������${\;}_{92}^{238}$U��������������֮���ǣ�������
| A�� | 54 | B�� | 92 | C�� | 146 | D�� | 238 |
6���ճ������������ĺܶ����ⶼ�漰����ѧ֪ʶ�������й�������ȷ���ǣ�������
| A�� | ���챦ʯ��Ҫ�ɷ������������մɣ�����ɸ��Ҫ�ɷ����л���߷��ӻ����� | |
| B�� | ʯ��ԭ�͵���ˮ����ɫ���������ײ��ϡ����ϡ����ϳ���ά��������̶����õ����� | |
| C�� | ̼��Ʒֽ⡢��������������Ȼ�粒��巴Ӧ��������������������Ӧ�������ȷ�Ӧ | |
| D�� | �绯���ۻ�����������ɫ��Ӧ���ǻ�ѧ�仯 |
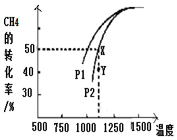 �о�����������Ⱦ��������ǻ�����һ����Ҫ������
�о�����������Ⱦ��������ǻ�����һ����Ҫ������