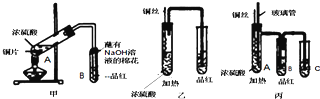
��Ŀ����
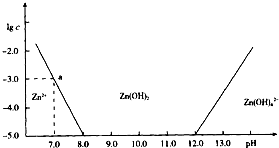 ��֪Zn2+��Ӧ�����ɰ�ɫ���������������ɫ�����ܽ�����Zn��OH��42-����ͼ����Zn2+����Һ����μ�������������Һ�ı仯����ʾ��ͼ��������Ϊ��Һ��pH��������ΪZn2+���ӻ�Zn��OH��42-�������ʵ���Ũ�ȵĶ���ֵ���ش��������⣮
��֪Zn2+��Ӧ�����ɰ�ɫ���������������ɫ�����ܽ�����Zn��OH��42-����ͼ����Zn2+����Һ����μ�������������Һ�ı仯����ʾ��ͼ��������Ϊ��Һ��pH��������ΪZn2+���ӻ�Zn��OH��42-�������ʵ���Ũ�ȵĶ���ֵ���ش��������⣮��1����ZnCl2��Һ�м�������������������Һ����Ӧ�����ӷ���ʽ�ɱ�ʾΪ��
��2����ͼ�����ݼ���ɵ�Zn��OH��2���ܶȻ���Ksp���T
��3��ij��Һ�к�Zn2+���ӣ�Ϊ��ȡZn2+���ӿ��Կ�����Һ��pHֵ�ķ�Χ��
��4����֪��ZnCl2��Һ�м�������Ǧ�����Ǧ��Һ�����Ƶ�PbCl2��ɫ���壬25��ʱ��PbCl2�����������е��ܽ�����£�
| c��HCl����mol?L-1�� | 0.50 | 1.00 | 2.04 | 2.90 | 4.02 | 5.16 | 5.78 |
| c��PbCl2����mol?L-1����10-3 | 5.10 | 4.91 | 5.21 | 5.90 | 7.48 | 10.81 | 14.01 |
A����������Ũ�ȵ�����PbCl2������ܽ���ȱ�С���ֱ��
B��PbCl2������0.50mol?L-1�����е��ܽ��С���ڴ�ˮ�е��ܽ��
C��PbCl2����Ũ���ᷢ��ij�ֻ�ѧ��Ӧ
D��PbCl2���岻�����ܽ��ڱ���ʳ��ˮ�У�
���㣺���ܵ���ʵ��ܽ�ƽ�⼰����ת���ı���
ר�⣺����ƽ������Һ��pHר��
��������1����ͼ���֪��Һ�ļ��Խ�ǿʱ��п�Ĵ�����ʽΪ[Zn��OH��4]2-�����Ԫ���غ���д���̣�
��2������ͼ�����ݣ�����Ksp=c��Zn2+����c2��OH-�����㣻
��3��Ϊ��ȡZn2+�ɽ�Zn2+ת��ΪZn��OH��2����������ͼ��ȷ����ʱ������Һ��pH��Χ��
��4��A�����ݱ��е����ݷ�����
B��PbCl2��������ܽ�ƽ�������
C��PbCl2��Ũ�������ܽ������˵������������
D��PbCl2����Cl-�γ������ӣ�
��2������ͼ�����ݣ�����Ksp=c��Zn2+����c2��OH-�����㣻
��3��Ϊ��ȡZn2+�ɽ�Zn2+ת��ΪZn��OH��2����������ͼ��ȷ����ʱ������Һ��pH��Χ��
��4��A�����ݱ��е����ݷ�����
B��PbCl2��������ܽ�ƽ�������
C��PbCl2��Ũ�������ܽ������˵������������
D��PbCl2����Cl-�γ������ӣ�
���
�⣺��1����ͼ���֪��Һ�ļ��Խ�ǿʱ��п�Ĵ�����ʽΪ[Zn��OH��4]2-�������䷴Ӧ�����ӷ���ʽΪ��Zn2++4OH-�T[Zn��OH��4]2-��
�ʴ�Ϊ��Zn2++4OH-�T[Zn��OH��4]2-��
��2������Һ��pH=7.0ʱ��c��Zn2+��=10-3mol?L-1��Ksp=c��Zn2+����c2��OH-��=10-3?��10-7��2=10-17��
�ʴ�Ϊ��10-17��
��3������ͼ��֪����Һ��pH��ΧΪ8��pH��12ʱ��Zn2+�ɽ�Zn2+ת��ΪZn��OH��2��������Һ��п����Ũ��С��10-5 mol?L-1��
���Խ�����Ϊп���Ӳ����ڣ�����ij��Һ�к�Zn2+���ӣ�Ϊ��ȡZn2+���ӿ��Կ�����Һ��pHֵ�ķ�Χ��PHΪ8-12֮�䣬
�ʴ�Ϊ��PHΪ8-12֮�䣻
��4��A�����ݱ��е����ݷ�����֪����������Ũ�ȵ�����PbCl2������ܽ���ȱ�С���ֱ��A��ȷ��
B��PbCl2������ڳ����ܽ�ƽ�⣬������Cl-ƽ�����ƣ��ܽ�ȼ�С����B��ȷ��
C��PbCl2��Ũ�������ܽ������˵����������������PbCl2����Ũ���ᷴӦ����һ���ѵ���������ӣ���C��ȷ��
D��PbCl2����Cl-�γ������ӣ�����PbCl2��������ڱ���ʳ��ˮ����D����
�ʴ�Ϊ��D��
�ʴ�Ϊ��Zn2++4OH-�T[Zn��OH��4]2-��
��2������Һ��pH=7.0ʱ��c��Zn2+��=10-3mol?L-1��Ksp=c��Zn2+����c2��OH-��=10-3?��10-7��2=10-17��
�ʴ�Ϊ��10-17��
��3������ͼ��֪����Һ��pH��ΧΪ8��pH��12ʱ��Zn2+�ɽ�Zn2+ת��ΪZn��OH��2��������Һ��п����Ũ��С��10-5 mol?L-1��
���Խ�����Ϊп���Ӳ����ڣ�����ij��Һ�к�Zn2+���ӣ�Ϊ��ȡZn2+���ӿ��Կ�����Һ��pHֵ�ķ�Χ��PHΪ8-12֮�䣬
�ʴ�Ϊ��PHΪ8-12֮�䣻
��4��A�����ݱ��е����ݷ�����֪����������Ũ�ȵ�����PbCl2������ܽ���ȱ�С���ֱ��A��ȷ��
B��PbCl2������ڳ����ܽ�ƽ�⣬������Cl-ƽ�����ƣ��ܽ�ȼ�С����B��ȷ��
C��PbCl2��Ũ�������ܽ������˵����������������PbCl2����Ũ���ᷴӦ����һ���ѵ���������ӣ���C��ȷ��
D��PbCl2����Cl-�γ������ӣ�����PbCl2��������ڱ���ʳ��ˮ����D����
�ʴ�Ϊ��D��
���������⿼�������ܵ���ʵ��ܽ�ƽ�⣬ע���ͼ��ͱ����е����ݵķ�������Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �����ƻ���ĩ��̶�100��ϵ�д�
�����ƻ���ĩ��̶�100��ϵ�д� �ܿ���ȫ��100��ϵ�д�
�ܿ���ȫ��100��ϵ�д�
�����Ŀ
��NA��ʾ����٤��������ֵ������������ȷ���ǣ�������
| A��22.4L������£�NO��0.5molO2��ϳ�ַ�Ӧ�����õ�NA��NO2���� |
| B���κη�Ӧ�У�����1molO2��ת��4NA������ |
| C��100g17%�İ�ˮ����Һ�к��а�������NA |
| D��7.1gCl2����������������Һ��Ӧת�Ƶ�����Ϊ0.1NA |
ʵ���е����в�����ȷ���ǣ�������
| A�����Թ�ȡ���Լ�ƿ�е�Na2CO3��Һ������ȡ�����࣬�ְѹ������Լ������Լ�ƿ�� |
| B��Ba��NO3��2 ����ˮ���ɽ�����Ba��NO3��2�ķ�Һ����ˮ���У�����ˮ������ˮ�� |
| C������������ʹNaCl����Һ������ʱ��Ӧ����������NaCl ��Һȫ���������� |
| D����Ũ��������һ�����ʵ���Ũ�ȵ�ϡ����ʱ��Ũ��������ˮ��Ӧ��ȴ��������ת�Ƶ�����ƿ�� |
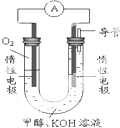 �״���CH3OH����һ�ֿ�������Դ�����й㷺�Ŀ�����Ӧ��ǰ����
�״���CH3OH����һ�ֿ�������Դ�����й㷺�Ŀ�����Ӧ��ǰ����