��Ŀ����
���й������ʵ���Ũ�ȱ�����ȷ����
A��0.2mol��L��1 Na2SO4��Һ�к���Na+��SO42�������ʵ���Ϊ0.6mol
B��50mL 2mol��L��1��NaCl��Һ��100 mL L 0.5mol��L��1MgCl2��Һ�У�Cl�������ʵ���Ũ�����
C����1Lˮ����22.4L�Ȼ��⣨����£����������Ũ����1mol��L��1
D��10g 98%�����ᣨ�ܶ�Ϊ1.84g��cm��3����10mL 18.4mol��L��1�������Ũ������ͬ��
��ϰ��ϵ�д�
 ̽���빮�̺��Ͽ�ѧ����������ϵ�д�
̽���빮�̺��Ͽ�ѧ����������ϵ�д�
�����Ŀ


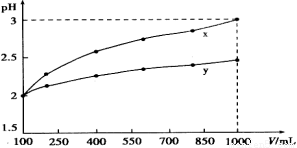

 �ټ�������ϱ���H��_____________��
�ټ�������ϱ���H��_____________��