��Ŀ����
ij�о���ѧϰС����ʵ���Һϳ���һ������A��
��1����������A����Է�������������100��A��C��H�����������ֱ�Ϊ��w��C��=69.76%��w��H��=11.63%������ȫȼ�պ����ֻ��CO2��H2O����A��Ħ������Ϊ ��
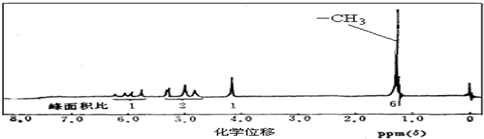
��2��A�ĺ˴Ź�����������ͼ��ʾ����A���Ժͽ����Ʒ�Ӧ����H2����������Cu�������±�������������ʾ���ǻ���̼̼˫�������Ľṹ���ȶ���
�����������Ϣд��A�Ľṹ��ʽ ��
��3��A��ij��ͬ���칹��B�����в���֧�����ܷ���������Ӧ����д��B����������Ӧ�Ļ�ѧ����ʽ ��
��1����������A����Է�������������100��A��C��H�����������ֱ�Ϊ��w��C��=69.76%��w��H��=11.63%������ȫȼ�պ����ֻ��CO2��H2O����A��Ħ������Ϊ
��2��A�ĺ˴Ź�����������ͼ��ʾ����A���Ժͽ����Ʒ�Ӧ����H2����������Cu�������±�������������ʾ���ǻ���̼̼˫�������Ľṹ���ȶ���

�����������Ϣд��A�Ľṹ��ʽ
��3��A��ij��ͬ���칹��B�����в���֧�����ܷ���������Ӧ����д��B����������Ӧ�Ļ�ѧ����ʽ
���㣺�л���ʵ��ʽ�ͷ���ʽ��ȷ��,�����л�������Ľṹ
ר�⣺�л���ѧ����
��������1��A������w��C��=69.76%��w��H��=11.63%����O����������Ϊ1-69.76%-11.63%=18.61%����Է�������������100����Oԭ�������Ŀ=
=1.2���ʺ�1��Oԭ�ӣ���A����Է�������=
=86��
��2��������Է���������̼��������������A��̼��ԭ�Ӹ�����д������ʽ������A�ĺ˴Ź������ף���A���Ժͽ����Ʒ�Ӧ����H2����������Cu�������±�������ȷ���ṹʽ��
��3�����������Ϣ��֪B�Ľṹ��ʽ������д��B����������Ӧ����ʽ��
| 100��18.61% |
| 16 |
| 16 |
| 0.1861 |
��2��������Է���������̼��������������A��̼��ԭ�Ӹ�����д������ʽ������A�ĺ˴Ź������ף���A���Ժͽ����Ʒ�Ӧ����H2����������Cu�������±�������ȷ���ṹʽ��
��3�����������Ϣ��֪B�Ľṹ��ʽ������д��B����������Ӧ����ʽ��
���
�⣺��1��A������w��C��=69.76%��w��H��=11.63%����O����������Ϊ1-69.76%-11.63%=18.61%����Է�������������100����Oԭ�������Ŀ=
=1.2���ʺ�1��Oԭ�ӣ���A����Է�������=
=86��
�ʴ�Ϊ��86g/mol��
��2����A������Cԭ����Ŀ=
=5��Hԭ����Ŀ=
=10�������ʽΪC5H10O��
��A�ĺ˴Ź�������֪���������4����ԭ�ӣ��������Ϊ1��2��1��6����2��-CH3����������Ʒ�Ӧ����H2��˵����-OH�����ܱ���������˵��-OH����̼ԭ������H����-OH����˫��̼��������A�Ľṹ��ʽΪ�� ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��3��A��ij��ͬ���칹��B�����в���֧�����ܷ���������Ӧ����B�Ľṹ�к�ȩ������B�Ľṹ��ʽΪ��CH3CH2CH2CH2CHO��
��Ӧ����ʽΪ��CH3CH2CH2CH2CHO+2Ag��NH3��2OH
CH3CH2CH2CH2COONH4+2Ag+3NH3+H2O��
�ʴ�Ϊ��CH3CH2CH2CH2CHO+2Ag��NH3��2OH
CH3CH2CH2CH2COONH4+2Ag+3NH3+H2O��
| 100��18.61% |
| 16 |
| 16 |
| 0.1861 |
�ʴ�Ϊ��86g/mol��
��2����A������Cԭ����Ŀ=
| 86��69.76% |
| 12 |
| 86��11.63% |
| 1 |
��A�ĺ˴Ź�������֪���������4����ԭ�ӣ��������Ϊ1��2��1��6����2��-CH3����������Ʒ�Ӧ����H2��˵����-OH�����ܱ���������˵��-OH����̼ԭ������H����-OH����˫��̼��������A�Ľṹ��ʽΪ��
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����3��A��ij��ͬ���칹��B�����в���֧�����ܷ���������Ӧ����B�Ľṹ�к�ȩ������B�Ľṹ��ʽΪ��CH3CH2CH2CH2CHO��
��Ӧ����ʽΪ��CH3CH2CH2CH2CHO+2Ag��NH3��2OH
| ˮԡ |
�ʴ�Ϊ��CH3CH2CH2CH2CHO+2Ag��NH3��2OH
| ˮԡ |
���������⿼�����л������ʽ���ƶϡ��ṹ��ʽ��ͬ���칹�����д����Ŀ�Ѷ��еȣ�ע��˴Ź�������Ӧ�ã�

��ϰ��ϵ�д�
 ������ĩ��ϰ��ѵ��ϵ�д�
������ĩ��ϰ��ѵ��ϵ�д� С��ʿ��ĩ����100��ϵ�д�
С��ʿ��ĩ����100��ϵ�д�
�����Ŀ
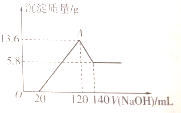 ��һ��������þ���Ͻ�Ͷ�뵽100mLһ�����ʵ���Ũ�ȵ�ij�����У���ַ�Ӧ����Ӧ�����Һ����μ���һ�����ʵ���Ũ�ȵ�NaOH��Һ�����ɳ���������������NaOH��Һ�������ϵ��ͼ��ʾ���Լ��㣺
��һ��������þ���Ͻ�Ͷ�뵽100mLһ�����ʵ���Ũ�ȵ�ij�����У���ַ�Ӧ����Ӧ�����Һ����μ���һ�����ʵ���Ũ�ȵ�NaOH��Һ�����ɳ���������������NaOH��Һ�������ϵ��ͼ��ʾ���Լ��㣺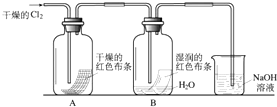 Ϊ��̽��HClO��Ư���ԣ�ijͬѧ��������µ�ʵ�飮
Ϊ��̽��HClO��Ư���ԣ�ijͬѧ��������µ�ʵ�飮