��Ŀ����
������������һ������Ư�ס���������Ư�ס�����Ч������H2O2�������ᣬij��ҵ�ù��������صIJ��ֲ��������±���
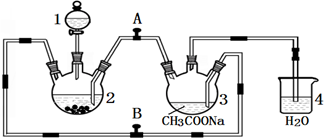
�ϳɹ��������ص����̼���Ӧ����ʾ��ͼ���£�
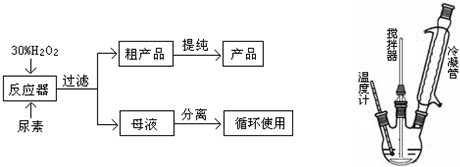
��ش��������⣺
��1��д���ϳɹ��������صĻ�ѧ����ʽ�� ��
��2����Ӧ��������������ˮ�� ����ϡ����¡��������룻��Ӧ���ļ��ȷ�ʽ�� ��
��ֱ�Ӽ��� ��ˮԡ���� ������ԡ���� �ܱ�ԡ
��3��������ѡ�õIJ����Dz������������ʻ����ʲ��ϵ�ԭ���� ��
��4����ĸҺ�з����H2O2�����أ����õ��Ǽ�ѹ��������ȴ�ᾧ�ķ�������ԭ���� ��
��5�������������ĸߵ�ֱ�Ӿ�����Ʒ���������ϸ��Ʒ�л������ĺ�����16%���൱�����к�H2O234%����Ϊ��ȷ�����ò�Ʒ�ϸ�����ʼ�Ա��ȡ������Ʒ2.000g���ܽ���ˮ����250mL����ƿ�ж��ݣ�ȷ��ȡ����25.00mL��Һ����ƿ�У�����1mL 6mol/L H2SO4��Ȼ����0.1000mol/L KMnO4����Һ�ζ���Ʒ�е�H2O2��KMnO4��Һ�������ط�Ӧ�������������һ��ʱ����Һ��dz��ɫ�Ұ�����ڲ���ɫ�����εζ�ƽ������KMnO4��Һ6.000mL��
����ɲ���ƽ���л�ѧ����ʽ��
MnO4-+ H2O2+ H+= Mn2++ H2O+
��KMnO4��ҺӦʢ���ڵζ����У����ζ�ǰ�ζ��ܼ��촦�����ݣ��ζ�����ʧ����ʹ��õĻ��������� ���ƫ�ߡ�����ƫ�͡����䡱����
�۸��ݵζ����ȷ����Ʒ���� ����ϸ��ϸ�������������������Ϊ ��
| ����ʽ | ��� | �ȷֽ��¶� | �۵� | ˮ���ԣ�20�棩 |
| CO��NH2��2?H2O2 | ��ɫ���� | 45�� | 75-85�� | 500g?L-1 |

��ش��������⣺
��1��д���ϳɹ��������صĻ�ѧ����ʽ��
��2����Ӧ��������������ˮ��
��ֱ�Ӽ��� ��ˮԡ���� ������ԡ���� �ܱ�ԡ
��3��������ѡ�õIJ����Dz������������ʻ����ʲ��ϵ�ԭ����
��4����ĸҺ�з����H2O2�����أ����õ��Ǽ�ѹ��������ȴ�ᾧ�ķ�������ԭ����
��5�������������ĸߵ�ֱ�Ӿ�����Ʒ���������ϸ��Ʒ�л������ĺ�����16%���൱�����к�H2O234%����Ϊ��ȷ�����ò�Ʒ�ϸ�����ʼ�Ա��ȡ������Ʒ2.000g���ܽ���ˮ����250mL����ƿ�ж��ݣ�ȷ��ȡ����25.00mL��Һ����ƿ�У�����1mL 6mol/L H2SO4��Ȼ����0.1000mol/L KMnO4����Һ�ζ���Ʒ�е�H2O2��KMnO4��Һ�������ط�Ӧ�������������һ��ʱ����Һ��dz��ɫ�Ұ�����ڲ���ɫ�����εζ�ƽ������KMnO4��Һ6.000mL��
����ɲ���ƽ���л�ѧ����ʽ��
��KMnO4��ҺӦʢ���ڵζ����У����ζ�ǰ�ζ��ܼ��촦�����ݣ��ζ�����ʧ����ʹ��õĻ���������
�۸��ݵζ����ȷ����Ʒ����
���㣺�Ʊ�ʵ�鷽�������,������ԭ��Ӧ����ʽ����ƽ,�к͵ζ�
ר�⣺
��������1�����̷�����֪���ϳɹ��������������ù�����������ػ��Ϸ�Ӧ���ɹ��������أ�
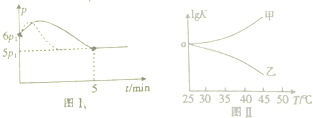
��2������װ��ͼ����������ˮӦ�ô��¿����룬CO��NH2��2?H2O2���Ʊ��¶Ƚϵͣ�Ӧ����ˮԡ���ȣ�
��3������������Fe��Al��Ӧ�������������ӣ�
��4�������������ֽ⣻��ѹ�������õ�H2O2��Ȼ��ᾧ�õ����أ�
��5������֪������KMnO4��Һ����Ӧ���������ⱻ���������Һ�������������̡���������ϵ����غ㡢ԭ���غ���ƽ��д��Ӧ�����Ļ�ѧ����ʽ��
��KMnO4��Һ����ǿ�����ԣ�ʢ������ʽ�ζ����У����ζ�ǰ�ζ��ܼ��촦�����ݣ��ζ�����ʧ�����ı���Һ������ⶨ���ƫ�ߣ�
�۸��ݸ�����ص����ʵ������H2O2��CO��NH2��2?H2O2�����ʵ��������������������������
��2������װ��ͼ����������ˮӦ�ô��¿����룬CO��NH2��2?H2O2���Ʊ��¶Ƚϵͣ�Ӧ����ˮԡ���ȣ�
��3������������Fe��Al��Ӧ�������������ӣ�
��4�������������ֽ⣻��ѹ�������õ�H2O2��Ȼ��ᾧ�õ����أ�
��5������֪������KMnO4��Һ����Ӧ���������ⱻ���������Һ�������������̡���������ϵ����غ㡢ԭ���غ���ƽ��д��Ӧ�����Ļ�ѧ����ʽ��
��KMnO4��Һ����ǿ�����ԣ�ʢ������ʽ�ζ����У����ζ�ǰ�ζ��ܼ��촦�����ݣ��ζ�����ʧ�����ı���Һ������ⶨ���ƫ�ߣ�
�۸��ݸ�����ص����ʵ������H2O2��CO��NH2��2?H2O2�����ʵ��������������������������
���
�⣺��1�����̷�����֪���ϳɹ��������������ù�����������ػ��Ϸ�Ӧ���ɹ��������أ���Ӧ�Ļ�ѧ����ʽΪ��CO��NH2��2+H2O2=CO��NH2��2?H2O2��
�ʴ�Ϊ��CO��NH2��2+H2O2=CO��NH2��2?H2O2��
��2������װ��ͼ��֪����XΪ������ƿ������ˮӦ�ô��¿����룬��������Ч���ã�������ˮ���¿����룻CO��NH2��2?H2O2���Ʊ��¶Ƚϵͣ�CO��NH2��2?H2O2��45��ʱ�����ֽ⣬���Բ����þƾ���ֱ�Ӽ��ȣ�Ӧ����ˮԡ���ȣ�
�ʴ�Ϊ���£��ڣ�
��3������������Fe��Al��Ӧ�������������ӣ����������ӻ���ٹ�������ķֽ⣬���Խ��������������ʡ����ʲ��ϵģ�
�ʴ�Ϊ�������ױ����������⣩��ʴ��
��4���������������¶ȸ���45��ʱ�ᷢ���ֽ⣬���ü�ѹ����ʱ��Һ���ڽϵ��¶��¼��ɻӷ������Է�ֹ���������ط����ֽ⣻ĸҺ����ѭ��ʹ�����ԭ�������ʣ�ĸҺ�к���H2O2�����أ���ѹ�������õ�H2O2��Ȼ��ᾧ�õ����أ�
�ʴ�Ϊ�����������¶ȣ���ֹ��������ֽ⣻
��5������֪������KMnO4��Һ����Ӧ���������ⱻ���������Һ�������������̡���������ϵ����غ㡢ԭ���غ���ƽ��д��Ӧ�����Ļ�ѧ����ʽΪ��5H2O2+2KMnO4+3H2SO4=8H2O+2MnSO4+K2SO4+5O2����
�ʴ�Ϊ��2��5��6��2��8��5O2��
��KMnO4��Һ����ǿ�����ԣ�ʢ������ʽ�ζ����У����ζ�ǰ�ζ��ܼ��촦�����ݣ��ζ�����ʧ����ζ�ʱ���ĵĸ���������ƫ������ص����ʵ���ƫ�����������CO��NH2��2?H2O2������ƫ��õĹ��������غ���ƫ�ߣ�
�ʴ�Ϊ����ʽ��ƫ�ߣ�
����֪������KMnO4��Һ����Ӧ�������ķ�ӦΪ5H2O2+2KMnO4+3H2SO4=8H2O+2MnSO4+K2SO4+5O2����
5H2O2����������2KMnO4
5 2
nmol 0.1000mol?L-1��0.006L
��n=0.0015mol��
250ml��Һ��CO��NH2��2?H2O2Ϊ0.0015mol��
=0.015mol
����CO��NH2��2?H2O2Ϊ0.015mol���������������Ϊ0.015mol��34g/mol=0.51g
���Բ�Ʒ��H2O2����������=
��100%=25.5%��
����������������=
=12%�����ϸ�
�ʴ�Ϊ�����ϸ�12%��
�ʴ�Ϊ��CO��NH2��2+H2O2=CO��NH2��2?H2O2��
��2������װ��ͼ��֪����XΪ������ƿ������ˮӦ�ô��¿����룬��������Ч���ã�������ˮ���¿����룻CO��NH2��2?H2O2���Ʊ��¶Ƚϵͣ�CO��NH2��2?H2O2��45��ʱ�����ֽ⣬���Բ����þƾ���ֱ�Ӽ��ȣ�Ӧ����ˮԡ���ȣ�
�ʴ�Ϊ���£��ڣ�
��3������������Fe��Al��Ӧ�������������ӣ����������ӻ���ٹ�������ķֽ⣬���Խ��������������ʡ����ʲ��ϵģ�
�ʴ�Ϊ�������ױ����������⣩��ʴ��
��4���������������¶ȸ���45��ʱ�ᷢ���ֽ⣬���ü�ѹ����ʱ��Һ���ڽϵ��¶��¼��ɻӷ������Է�ֹ���������ط����ֽ⣻ĸҺ����ѭ��ʹ�����ԭ�������ʣ�ĸҺ�к���H2O2�����أ���ѹ�������õ�H2O2��Ȼ��ᾧ�õ����أ�
�ʴ�Ϊ�����������¶ȣ���ֹ��������ֽ⣻
��5������֪������KMnO4��Һ����Ӧ���������ⱻ���������Һ�������������̡���������ϵ����غ㡢ԭ���غ���ƽ��д��Ӧ�����Ļ�ѧ����ʽΪ��5H2O2+2KMnO4+3H2SO4=8H2O+2MnSO4+K2SO4+5O2����
�ʴ�Ϊ��2��5��6��2��8��5O2��
��KMnO4��Һ����ǿ�����ԣ�ʢ������ʽ�ζ����У����ζ�ǰ�ζ��ܼ��촦�����ݣ��ζ�����ʧ����ζ�ʱ���ĵĸ���������ƫ������ص����ʵ���ƫ�����������CO��NH2��2?H2O2������ƫ��õĹ��������غ���ƫ�ߣ�
�ʴ�Ϊ����ʽ��ƫ�ߣ�
����֪������KMnO4��Һ����Ӧ�������ķ�ӦΪ5H2O2+2KMnO4+3H2SO4=8H2O+2MnSO4+K2SO4+5O2����
5H2O2����������2KMnO4
5 2
nmol 0.1000mol?L-1��0.006L
��n=0.0015mol��
250ml��Һ��CO��NH2��2?H2O2Ϊ0.0015mol��
| 250 |
| 25 |
����CO��NH2��2?H2O2Ϊ0.015mol���������������Ϊ0.015mol��34g/mol=0.51g
���Բ�Ʒ��H2O2����������=
| 0.51g |
| 2.00g |
����������������=
| 16%��25.5% |
| 34% |
�ʴ�Ϊ�����ϸ�12%��
���������⿼���������Ʊ�ʵ����̷����жϣ�װ��ͼ������Ӧ�ã��ζ�ʵ����̺ͼ���ķ���Ӧ�ã����ջ����ǹؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 ����������������ϵ�д�
����������������ϵ�д�
�����Ŀ
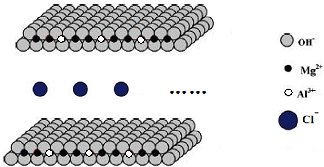
���������У����������ƶ���Cl-���ǣ�������
| A��KCl���� |
| B��HCl���� |
| C��KClO3��Һ |
| D��MgCl2��Һ |
��NAΪ����٤��������ֵ������˵��ȷ���ǣ�������
| A�������£�0.2molFe������ϡ���ᷴӦ��Ӧ�����ɵ�H2������ĿΪ0.3NA |
| B��78g���������к���������Ϊ2 NA |
| C��C2H4��N2��ɵ�42g���������ԭ�ӵĸ���Ϊ3 NA |
| D��5NH4 NO3�T2HNO3+4N2��+9H2O��Ӧ�У�����28gN2 ʱ��ת�Ƶĵ�����ĿΪ3.75NA |
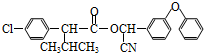 ����
����
 ����һȡ�������̼̼������X���п��ܵĽṹ����
����һȡ�������̼̼������X���п��ܵĽṹ����

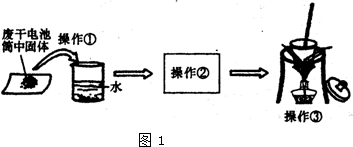 �ճ�������ʹ�õ�п�̸ɵ�أ���������пƬ��������ʯī��������Ǻ�ʯī֮�����������̺�̼���Լ��Ȼ�п���Ȼ�淋ĺ�״�ij�о���ѧϰС��Ըɵ�ص�������ʽ�������ͼ1ʵ�飮
�ճ�������ʹ�õ�п�̸ɵ�أ���������пƬ��������ʯī��������Ǻ�ʯī֮�����������̺�̼���Լ��Ȼ�п���Ȼ�淋ĺ�״�ij�о���ѧϰС��Ըɵ�ص�������ʽ�������ͼ1ʵ�飮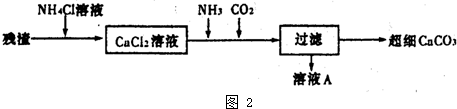
 þ���仯�����ڹ�ҵ���й㷺��Ӧ�ã�
þ���仯�����ڹ�ҵ���й㷺��Ӧ�ã�