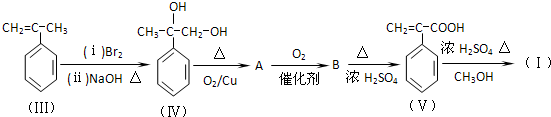
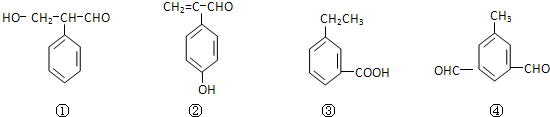
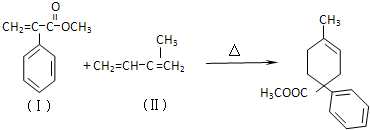��Ŀ����
ʯ�ͻ�������Ҫԭ��CxHy���Ժϳɺܶ��л������������CxHy�ϳ�����E��J������ͼ��

��֪�������з�Ӧ��R��R���������
��J�ķ���ʽΪC4H4O4����һ�ֻ�״�����
��1����CxHy��ͬϵ���У�����̼ԭ��һ����ƽ����̼ԭ�������ķ��ӵ������� ��
��2��H�ķ���ʽ�� ��
��3������˵����ȷ���� ��
a��CxHy�ͱ�����ʹ��ˮ��ɫ��ԭ����ͬ
b����Ӧ�ںͷ�Ӧ�ܵķ�Ӧ���;�Ϊ�ӳɷ�Ӧ
c��C����Na��NaOH��NaHCO3��Ӧ
d��E��һ��ˮ���Ժܺõĸ߷��ӻ�����
e��J�����Ի���Ի����о���ˮ��
��4��K��J��ͬ���칹�壬��1mol K��������NaHCO3��Һ��Ӧ�ɷų�2mol CO2���壬��д��
һ�ַ�������K�Ľṹ��ʽ ��
��5��д����Ӧ�ݵĻ�ѧ����ʽ ��
��6��D�ж���ͬ���칹�壬��D������ͬ�����ŵĻ��� �֣���˳���칹�壩�����к˴Ź���
������3�����շ壬���ܷ���������Ӧ�Ľṹ��ʽ�� ��

��֪�������з�Ӧ��R��R���������

��J�ķ���ʽΪC4H4O4����һ�ֻ�״�����
��1����CxHy��ͬϵ���У�����̼ԭ��һ����ƽ����̼ԭ�������ķ��ӵ�������
��2��H�ķ���ʽ��
��3������˵����ȷ����
a��CxHy�ͱ�����ʹ��ˮ��ɫ��ԭ����ͬ
b����Ӧ�ںͷ�Ӧ�ܵķ�Ӧ���;�Ϊ�ӳɷ�Ӧ
c��C����Na��NaOH��NaHCO3��Ӧ
d��E��һ��ˮ���Ժܺõĸ߷��ӻ�����
e��J�����Ի���Ի����о���ˮ��
��4��K��J��ͬ���칹�壬��1mol K��������NaHCO3��Һ��Ӧ�ɷų�2mol CO2���壬��д��
һ�ַ�������K�Ľṹ��ʽ
��5��д����Ӧ�ݵĻ�ѧ����ʽ
��6��D�ж���ͬ���칹�壬��D������ͬ�����ŵĻ���
������3�����շ壬���ܷ���������Ӧ�Ľṹ��ʽ��
���㣺�л���ĺϳ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
�������������и�����ת����ϵ��J�ķ���ʽΪC4H4O4����һ�ֻ�״���������I��Ũ���������¼��ȵõ������Կ�����֪JΪ��������JΪ �������йط�Ӧ����������֪IΪHOCH2COOH��HΪBrCH2COOH��GΪBrCH2CHO��FΪBrCH2CH2OH��CxHy��HBrO�ӳɵ�F������CxHyΪCH2=CH2��������AΪCH3CHO��CH3CHO��������BΪCH3COOH��A��B������Ϣ�еļӳɷ�Ӧ��CΪCH3COOCH��OH��CH3��C��Ũ������������ˮ��DΪCH3COOCH=CH2��D�����Ӿ۷�Ӧ�õ�E���ݴ˴��⣮
�������йط�Ӧ����������֪IΪHOCH2COOH��HΪBrCH2COOH��GΪBrCH2CHO��FΪBrCH2CH2OH��CxHy��HBrO�ӳɵ�F������CxHyΪCH2=CH2��������AΪCH3CHO��CH3CHO��������BΪCH3COOH��A��B������Ϣ�еļӳɷ�Ӧ��CΪCH3COOCH��OH��CH3��C��Ũ������������ˮ��DΪCH3COOCH=CH2��D�����Ӿ۷�Ӧ�õ�E���ݴ˴��⣮
 �������йط�Ӧ����������֪IΪHOCH2COOH��HΪBrCH2COOH��GΪBrCH2CHO��FΪBrCH2CH2OH��CxHy��HBrO�ӳɵ�F������CxHyΪCH2=CH2��������AΪCH3CHO��CH3CHO��������BΪCH3COOH��A��B������Ϣ�еļӳɷ�Ӧ��CΪCH3COOCH��OH��CH3��C��Ũ������������ˮ��DΪCH3COOCH=CH2��D�����Ӿ۷�Ӧ�õ�E���ݴ˴��⣮
�������йط�Ӧ����������֪IΪHOCH2COOH��HΪBrCH2COOH��GΪBrCH2CHO��FΪBrCH2CH2OH��CxHy��HBrO�ӳɵ�F������CxHyΪCH2=CH2��������AΪCH3CHO��CH3CHO��������BΪCH3COOH��A��B������Ϣ�еļӳɷ�Ӧ��CΪCH3COOCH��OH��CH3��C��Ũ������������ˮ��DΪCH3COOCH=CH2��D�����Ӿ۷�Ӧ�õ�E���ݴ˴��⣮���
�⣺�������и�����ת����ϵ��J�ķ���ʽΪC4H4O4����һ�ֻ�״���������I��Ũ���������¼��ȵõ������Կ�����֪JΪ��������JΪ �������йط�Ӧ����������֪IΪHOCH2COOH��HΪBrCH2COOH��GΪBrCH2CHO��FΪBrCH2CH2OH��CxHy��HBrO�ӳɵ�F������CxHyΪCH2=CH2��������AΪCH3CHO��CH3CHO��������BΪCH3COOH��A��B������Ϣ�еļӳɷ�Ӧ��CΪCH3COOCH��OH��CH3��C��Ũ������������ˮ��DΪCH3COOCH=CH2��D�����Ӿ۷�Ӧ�õ�E��
�������йط�Ӧ����������֪IΪHOCH2COOH��HΪBrCH2COOH��GΪBrCH2CHO��FΪBrCH2CH2OH��CxHy��HBrO�ӳɵ�F������CxHyΪCH2=CH2��������AΪCH3CHO��CH3CHO��������BΪCH3COOH��A��B������Ϣ�еļӳɷ�Ӧ��CΪCH3COOCH��OH��CH3��C��Ũ������������ˮ��DΪCH3COOCH=CH2��D�����Ӿ۷�Ӧ�õ�E��
��1����C2H4��ͬϵ���У�����̼ԭ��һ����ƽ����̼ԭ�������ķ����ǽ���ϩ�е���ԭ�Ӷ�����̼�Ľṹ��C6H12����������2��3-����-2-��ϩ��
�ʴ�Ϊ��2��3-����-2-��ϩ�� ������������
��2��HΪBrCH2COOH��H�ķ���ʽ��C2H3O2Br��
�ʴ�Ϊ��C2H3O2Br��
��3��a��CxHy�ͱ�����ʹ��ˮ��ɫ��ԭ������ͬ��ǰ���Ǽӳɣ���������ȡ����a����
b����Ӧ�ںͷ�Ӧ�ܵķ�Ӧ���;�Ϊ�ӳɷ�Ӧ����b��ȷ��
c��C�����������ǻ���������NaOH��NaHCO3��Ӧ����c����
d��E������������ˮ���Խϲ��d����
e��J���������������Ի���Ի����о���ˮ�⣬��e��ȷ��
�ʴ�Ϊ��be��
��4��JΪ ��K��J��ͬ���칹�壬��1 mol K��������NaHCO3��Һ��Ӧ�ɷų�2mol CO2���壬˵��K���������Ȼ������Է�������K�Ľṹ��ʽΪCH2=C��COOH��2����HOOCCH=CHCOOH��
��K��J��ͬ���칹�壬��1 mol K��������NaHCO3��Һ��Ӧ�ɷų�2mol CO2���壬˵��K���������Ȼ������Է�������K�Ľṹ��ʽΪCH2=C��COOH��2����HOOCCH=CHCOOH��
�ʴ�Ϊ��CH2=C��COOH��2����HOOCCH=CHCOOH��
��5����Ӧ�ݵĻ�ѧ����ʽΪ ��
��
�ʴ�Ϊ�� ��
��
��6��DΪCH3COOCH=CH2��D�ж���ͬ���칹�壬��D������ͬ�����ŵĻ���CH2=CHCOOCH3��HCOOCH=CHCH3��˳�������֣���HCOOCH2CH=CH2��HCOOC��CH3��=CH2������5 �֣���˳���칹�壩�����к˴Ź���������3�����շ壬���ܷ���������Ӧ�Ľṹ��ʽΪHCOOC��CH3��=CH2��
�ʴ�Ϊ��5��HCOOC��CH3��=CH2��
 �������йط�Ӧ����������֪IΪHOCH2COOH��HΪBrCH2COOH��GΪBrCH2CHO��FΪBrCH2CH2OH��CxHy��HBrO�ӳɵ�F������CxHyΪCH2=CH2��������AΪCH3CHO��CH3CHO��������BΪCH3COOH��A��B������Ϣ�еļӳɷ�Ӧ��CΪCH3COOCH��OH��CH3��C��Ũ������������ˮ��DΪCH3COOCH=CH2��D�����Ӿ۷�Ӧ�õ�E��
�������йط�Ӧ����������֪IΪHOCH2COOH��HΪBrCH2COOH��GΪBrCH2CHO��FΪBrCH2CH2OH��CxHy��HBrO�ӳɵ�F������CxHyΪCH2=CH2��������AΪCH3CHO��CH3CHO��������BΪCH3COOH��A��B������Ϣ�еļӳɷ�Ӧ��CΪCH3COOCH��OH��CH3��C��Ũ������������ˮ��DΪCH3COOCH=CH2��D�����Ӿ۷�Ӧ�õ�E����1����C2H4��ͬϵ���У�����̼ԭ��һ����ƽ����̼ԭ�������ķ����ǽ���ϩ�е���ԭ�Ӷ�����̼�Ľṹ��C6H12����������2��3-����-2-��ϩ��
�ʴ�Ϊ��2��3-����-2-��ϩ�� ������������
��2��HΪBrCH2COOH��H�ķ���ʽ��C2H3O2Br��
�ʴ�Ϊ��C2H3O2Br��
��3��a��CxHy�ͱ�����ʹ��ˮ��ɫ��ԭ������ͬ��ǰ���Ǽӳɣ���������ȡ����a����
b����Ӧ�ںͷ�Ӧ�ܵķ�Ӧ���;�Ϊ�ӳɷ�Ӧ����b��ȷ��
c��C�����������ǻ���������NaOH��NaHCO3��Ӧ����c����
d��E������������ˮ���Խϲ��d����
e��J���������������Ի���Ի����о���ˮ�⣬��e��ȷ��
�ʴ�Ϊ��be��
��4��JΪ
 ��K��J��ͬ���칹�壬��1 mol K��������NaHCO3��Һ��Ӧ�ɷų�2mol CO2���壬˵��K���������Ȼ������Է�������K�Ľṹ��ʽΪCH2=C��COOH��2����HOOCCH=CHCOOH��
��K��J��ͬ���칹�壬��1 mol K��������NaHCO3��Һ��Ӧ�ɷų�2mol CO2���壬˵��K���������Ȼ������Է�������K�Ľṹ��ʽΪCH2=C��COOH��2����HOOCCH=CHCOOH���ʴ�Ϊ��CH2=C��COOH��2����HOOCCH=CHCOOH��
��5����Ӧ�ݵĻ�ѧ����ʽΪ
 ��
���ʴ�Ϊ��
 ��
����6��DΪCH3COOCH=CH2��D�ж���ͬ���칹�壬��D������ͬ�����ŵĻ���CH2=CHCOOCH3��HCOOCH=CHCH3��˳�������֣���HCOOCH2CH=CH2��HCOOC��CH3��=CH2������5 �֣���˳���칹�壩�����к˴Ź���������3�����շ壬���ܷ���������Ӧ�Ľṹ��ʽΪHCOOC��CH3��=CH2��
�ʴ�Ϊ��5��HCOOC��CH3��=CH2��
���������⿼���л�����ƶ���ϳɣ���ϸ������Ϣ��������úϳ�·�������ʹ����ż�̼���ı仯�ƶϣ���Ŀ�Ѷ��еȣ�ע���л�����֪ʶ��������ã�

��ϰ��ϵ�д�
 ���ѵ����Ԫ��ĩ���100��ϵ�д�
���ѵ����Ԫ��ĩ���100��ϵ�д� ��˼άС�ھ�100����ҵ��ϵ�д�
��˼άС�ھ�100����ҵ��ϵ�д� ��ʦָ��һ��ͨϵ�д�
��ʦָ��һ��ͨϵ�д�
�����Ŀ
�����ij���ᣨH2CrO4����Һ������Ũ�ȣ�mol?L-1����pH�Ĺ�ϵ������˵������ȷ���ǣ�������
| pH | c��CrO42-�� | c��HCrO4-�� | c�� Cr2O72-�� | c��H2CrO4�� |
| 4 | 0.0003 | 0.1040 | 0.4480 | 0 |
| 6 | 0.0319 | 0.0999 | 0.4370 | 0 |
| 7 | 0.2745 | 0.0860 | 0.3195 | 0 |
| 9 | 0.9960 | 0.0031 | 0.0004 | 0 |
| A������ĵ�һ����������ȫ���� |
| B������ĵڶ��������Dz��ֵ��룬�Ҵ������ֵ��뷽ʽ |
| C��pHԽ�������CrO42-Ũ��Խ�� |
| D����Һ�д���2HCrO4-?Cr2O72-+H2O |
�����£�0.1mol/L������Һ��pH=3�������¶���0.2mol/L������Һ�� 0.2mol/L��������Һ�������Ϻ��Һ��pH=4.7������˵����ȷ���ǣ�������
| A�����¶��£�0.0lmol/L������Һ��pH=4 |
| B�����¶��£���0.1mol/L�����0.01 mol/L����ֱ���ȫ�к͵����0.1 mol/L��NaOH��Һ������������������Ϊ1��10 |
| C�����¶��£�0.01mol/L������Һ����ˮ�������c��H+��=10-11mol/L |
| D�����¶��£�0.2mol/L������Һ��0.2mol/L��������Һ�������Ϻ��Һ��c��CH3COO-����c��CH3COOH����c��Na+����c��H+����c��OH-�� |