��Ŀ����
13����֪�Ҵ��ڼ��ȵ������¿���HBr��Ӧ�õ������飨CH3CH2Br��������ijЩ�������������ʾ��| �ܽ��� | �е� | �ܶȣ�g/mL�� | |
| �Ҵ� | ��ˮ���� | 78.5 | 0.8 |
| ������ | ������ˮ | 38.4 | 1.4 |
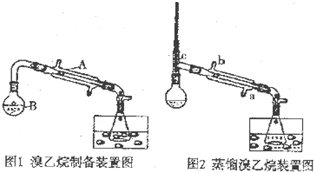
ij�о���ѧϰС���ͬѧ���Ҵ���80%�����ᡢNaBr���尴һ��������Ϻ������ԼΪ140mL������ͼ1�ķ�Ӧ����B�к���м��ȣ�����װ���еļ��ȡ��̶�װ�ü���ʯ��ʡ�ԣ���
��1��������A��������ֱ��������
��2������ˮ���е�Һ����DZ�ˮ��ʹ�ñ�ˮ��Ŀ���ǽ��£�����������Ļӷ���
��3��HBr���Ҵ���Ӧ����Ŀ�����Ļ�ѧ����ʽHBr+CH3CH2OH$\stackrel{��}{��}$CH3CH2Br+H2O��
��4��ͼ1���ռ����IJ�Ʒ�к���һ����������Br2��Ϊ��ȥ�����ʣ���������Һ�м��������ģ������������Լ���ѡȡ����ѡ����ĸ��b��Ȼ���ٽ��з�Һ�������ɣ�
a�����Ȼ�̼����������b��̼������Һ�������� c����ˮCaCl2���������� d��Ũ����
��5��������4���������Ŀ������л�����������ˮ�����ʣ���ͨ�����Ʒ�м���һ������c���������a--d��ѡȡ����ѡ����ĸ����������ú��ˣ�����Һʹ��ͼ2�е�װ�ý����ᴿ����ȴˮӦ��a�ڣ���a��b�����룮
��6����������ʹ�õ���ˮ�Ҵ��������46mL�������õ���������������50mL����������IJ���Ϊ80%��
���� ��1������������������������������A������Ϊֱ�������ܣ�
��2������ˮ���е�Һ����DZ�ˮ��ʹ�ñ�ˮ��Ϊ����������CH3CH2Br�����£�����������Ļӷ���
��3��HBr�����Ҵ�����ȡ����Ӧ�����������ˮ��
��4���������л����壬����ʱע���������������ʽǶȿ��ǣ���ˮ��̼���Ʒ�Ӧ���ģ�����������Һ�����ܣ����÷�Һ�ķ������룻
��5���������л���ˮʱ���ɼ�����ˮCaCl2��ȥ������ʱ��ˮӦ�������ܵ��¶˽��룬�϶˳��������������ɳ��������
��6�������n���Ҵ���=$\frac{46ml��0.8g/ml}{46g/mol}$=0.8mol��n�������飩=$\frac{50ml��1.4g/ml}{109g/mol}$��0.6422mol��Ȼ�����ת���ʵĸ��������
��� �⣺��1��������������������A������������������������ֱ�������ܣ��ʴ�Ϊ��ֱ�������ܣ�
��2��������е�ֻ��38.4�棬�е�ͣ��ӷ�������ˮ���е�Һ����DZ�ˮ��ʹ�ñ�ˮ��Ŀ���ǣ����£�����������Ļӷ���
�ʴ�Ϊ�����£�����������Ļӷ���
��3��HBr�����Ҵ�����ȡ����Ӧ�����������ˮ��HBr+CH3CH2OH$\stackrel{��}{��}$CH3CH2Br+H2O��
�ʴ�Ϊ��HBr+CH3CH2OH$\stackrel{��}{��}$CH3CH2Br+H2O��
��4������Ũ�������ǿ�����ԣ���HBr������Br2�������������л��е����壬��������ȫ��ȥ���ʣ����Ҳ����鷳�����Ȼ�̼�������顢�嵥�ʶ����ܣ��������µ����ʣ���ˮCaCl2����ˮMgSO4�������뵥���巴Ӧ�����ܳ�ȥ�����壬̼������Һ�ܺ��嵥�ʷ�Ӧ���嵥�ʱ���ȥ������������Һ�����ܣ����߷ֲ㣬�÷�Һ�ķ������룬
�ʴ�Ϊ��b�� ��Һ��
��5��������3���������Ŀ������л�����������ˮ����ͨ�����Ʒ�м���һ������ˮCaCl2����ˮMgSO4��Ϊ�˳������������ʱ��ˮӦ�������ܵ��¶�a�ڽ��룬�϶˳���
�ʴ�Ϊ��c��a��
��6��46mL�Ҵ������ʵ���Ϊn���Ҵ���=$\frac{46ml��0.8g/ml}{46g/mol}$=0.8mol����������Ӧ����������0.8moL��ʵ��������������50mL��
n�������飩=$\frac{50ml��1.4g/ml}{109g/mol}$��0.6422mol��
��������IJ���Ϊ $\frac{0.6422mol}{0.8mol}$��100%��80%��
�ʴ�Ϊ��80%��
���� ���⿼���������ʵ������ȡ�����ؿ���ѧ��ʵ�����������������ʵ��ԭ�������ʵ������ǽ���Ĺؼ����ۺ��Խ�ǿ����Ŀ�Ѷ��еȣ�

 �����������һ��һ��ϵ�д�
�����������һ��һ��ϵ�д���ʵ�顿��ͬѧ����ƿ�м�����������˿��ŨH2SO4��Һ����ʼ�����Ա仯���Է�Ӧ����ȣ������ݲ������ռ�����ɫ���壮
���������ϡ�������Ũ�����ڳ����»ᷢ���ۻ�����������������������»ᷴӦ�������д̼�����ζ�Ķ����������壻
�ڶ�������������ʹƷ����Һ��ɫ���ܱ�����������Һ���գ�
��������衿��ͬѧ��Ϊ�ռ����ľ��Ƕ����������壬��ͬѧ��Ϊ��������������Ϊ��ͬѧԤ���������������ǣ����ŷ�Ӧ�Ľ��У�Ũ������ϡ��Fe����ϡ���ᷴӦ����������
�������ɿ��������¼��������
����һ����ɫ������SO2��
���������ɫ������H2��
����������ɫ������SO2 ��H2 �Ļ�����壮
��ʵ����֤���������ʵ����֤����������������±������ݣ�С��ͬѧ��ʵ�����ҵ�������ʵ��̽�����������õ����Լ��У�Ʒ����Һ��NaOH��Һ�����CuO��ĩ����ˮ����ͭ��������ѡ��
| ʵ�鷽���벽�� | ʵ������ͽ��� |
| 1������������ͨ��ʢ������Ʒ����Һ���Թ��ڣ� | ��Ʒ����Һ��ɫ������ɫ��������SO2�� |
| 2��������ͨ��װ����������������Һ��ϴ��ƿ����С�Թ��ռ����壬���ھƾ����ϵ�ȼ | ���ռ����������ȼ�գ�����ɫ��������H2�� �������ʵ���֪������������ |
��1��Ũ������ľ̿���ڼ��������µĻ�ѧ����ʽΪC+2H2SO4��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$2SO2��+CO2��+2H2O��
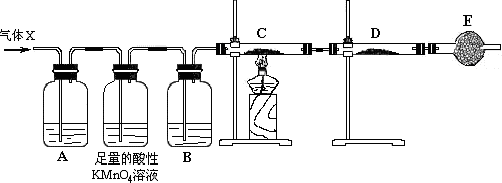
��2����֪KMnO4������Һ��������SO2������ͼ�и�װ�����һ��ʵ�飬��֤������Ӧ�������ĸ��ֲ����Щװ�õ�����˳���������������ҵķ����ǣ���װ�õı�ţ����ܡ��ڡ��١��ۣ�
| ��� | �� | �� | �� | �� |
| װ�� | 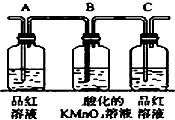 |  |  |  |
��4��װ�â������ӵĹ���ҩƷ����ˮCuSO4�����ɫ�轺������ȷ֤�IJ����ǣ�ˮ������ȷ��װ�â�������װ����λ�õ����������ڲ�������ͨ���٢�ʱ�����ˮ���������Ԣڱ����ڢ٢�֮ǰ��
��5��װ�â�����ʢ��Һ�dz���ʯ��ˮ������֤�IJ�����CO2 ��
| ��ѧ�� | H-Cl | O�TO | Cl-Cl | H-O |
| E/��kJ•mol-1�� | 431 | 397 | a | 465 |
| A�� | 242 | B�� | 303.5 | C�� | 180.5 | D�� | 365 |
| A�� | -441 kJ/mol | B�� | +441 kJ/mol | C�� | -882 kJ/mol | D�� | +882 kJ/mol |
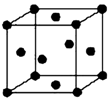 ��֪A��B��C��D��E����Ԫ�ص�ԭ���������ε�����A��B��C��Dλ��ǰ�����ڣ�Aλ�����ڱ���s������ԭ���е��Ӳ�����δ�ɶԵ�������ͬ��B�Ļ�̬ԭ���е���ռ������������ͬ��ԭ�ӹ������ÿ�ֹ���еĵ���������ͬ��Dԭ�ӵĺ���ɶԵ�������δ�ɶԵ������ı�ֵΪ3��2��ͬʱ����A��B��D����Ԫ�صĻ�����M��һ�־�����Ⱦ���壬����������е�ԭ�ӹ�ƽ�森A��B����Ԫ����ɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�����N�dz������л��ܼ���E�С����������֮�ƣ�E4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��
��֪A��B��C��D��E����Ԫ�ص�ԭ���������ε�����A��B��C��Dλ��ǰ�����ڣ�Aλ�����ڱ���s������ԭ���е��Ӳ�����δ�ɶԵ�������ͬ��B�Ļ�̬ԭ���е���ռ������������ͬ��ԭ�ӹ������ÿ�ֹ���еĵ���������ͬ��Dԭ�ӵĺ���ɶԵ�������δ�ɶԵ������ı�ֵΪ3��2��ͬʱ����A��B��D����Ԫ�صĻ�����M��һ�־�����Ⱦ���壬����������е�ԭ�ӹ�ƽ�森A��B����Ԫ����ɵ�ԭ�Ӹ�����Ϊ1��1�Ļ�����N�dz������л��ܼ���E�С����������֮�ƣ�E4+���Ӻ��ԭ�ӵĺ�������Ų���ͬ��