��Ŀ����
A��B��C��D��Ϊ��ѧ��ѧ�г��������ʣ�����֮���ת����ϵ����ͼ�����ֲ�������ȥ����
�Իش�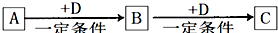
��1����D�Ǿ��������Եĵ��ʣ������ڶ����ڵ��������Ԫ��AΪ ����Ԫ�ط��ţ���
��2����D�ǽ������ʣ�D�ڳ�ʪ�Ŀ���������������ʴ��C��Һ�ڱ���ʱӦ�����������D��ֹ����ʣ�������D��C��Һ�ڿ����б��ʵ����ӷ���ʽΪ ����D���Ȼ����ˮ��Һ���ɲ����ղ����� ��
��3����A��B��C��Ϊ��������Ҿ����ؿ��к�����ߵĽ���Ԫ��E������Һ��A��C��Ӧ����B����д��Bת��ΪC�����п��ܵ����ӷ���ʽ ��
��4�����ڣ�1�����Ƴ���A������ڣ�3����E���ʵĻ����11.9gͶ��һ������ˮ�г�ַ�Ӧ��A��E��û��ʣ�࣬���ռ�����״���µ�����vL����������Һ����μ���Ũ��Ϊ2mol?L-1��H2SO4��Һ����100mLʱ��ɫ�����ﵽ���������v= ��
�Իش�
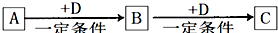
��1����D�Ǿ��������Եĵ��ʣ������ڶ����ڵ��������Ԫ��AΪ
��2����D�ǽ������ʣ�D�ڳ�ʪ�Ŀ���������������ʴ��C��Һ�ڱ���ʱӦ�����������D��ֹ����ʣ�������D��C��Һ�ڿ����б��ʵ����ӷ���ʽΪ
��3����A��B��C��Ϊ��������Ҿ����ؿ��к�����ߵĽ���Ԫ��E������Һ��A��C��Ӧ����B����д��Bת��ΪC�����п��ܵ����ӷ���ʽ
��4�����ڣ�1�����Ƴ���A������ڣ�3����E���ʵĻ����11.9gͶ��һ������ˮ�г�ַ�Ӧ��A��E��û��ʣ�࣬���ռ�����״���µ�����vL����������Һ����μ���Ũ��Ϊ2mol?L-1��H2SO4��Һ����100mLʱ��ɫ�����ﵽ���������v=
���㣺������ƶ�
ר�⣺�ƶ���
��������1��D�Ǿ��������Եĵ��ʣ�AԪ�����ڶ������������Ԫ�أ����ݷ�Ӧ��ϵ��֪AΪNa��BΪNa2O��DΪO2��CΪNa2O2��
��2����D�ǽ������ʣ�D�ڳ�ʪ�Ŀ���������������ʴ��C��Һ�ڱ���ʱӦ�����������D��ֹ����ʣ���DΪFe��BΪ���Σ�CΪ�����Σ�
��3����A��B��CΪ��ͬһ�ֽ���Ԫ�ص������������Һ��A��C��Ӧ����B��Ӧ����AlԪ�أ�����֪AΪƫ�����Ρ�DΪǿ�ᡢBΪ����������CΪ���Σ�����AΪ���Ρ�DΪǿ�BΪ����������CΪƫ�����Σ���
��4����Na������Al���ʵĻ����Ͷ��һ������ˮ�г�ַ�Ӧ��A��E��û��ʣ�࣬�õ�������Ϊ��������������Һ����μ���H2SO4��Һ������ɫ�����ﵽ���������ʱ��Һ������ΪNa2SO4����������غ��֪n��Na2SO4��=n��H2SO4�����ٸ���NaԪ���غ����m��Na������������m��Al�������ݵ���ת���غ�������������������
��2����D�ǽ������ʣ�D�ڳ�ʪ�Ŀ���������������ʴ��C��Һ�ڱ���ʱӦ�����������D��ֹ����ʣ���DΪFe��BΪ���Σ�CΪ�����Σ�
��3����A��B��CΪ��ͬһ�ֽ���Ԫ�ص������������Һ��A��C��Ӧ����B��Ӧ����AlԪ�أ�����֪AΪƫ�����Ρ�DΪǿ�ᡢBΪ����������CΪ���Σ�����AΪ���Ρ�DΪǿ�BΪ����������CΪƫ�����Σ���
��4����Na������Al���ʵĻ����Ͷ��һ������ˮ�г�ַ�Ӧ��A��E��û��ʣ�࣬�õ�������Ϊ��������������Һ����μ���H2SO4��Һ������ɫ�����ﵽ���������ʱ��Һ������ΪNa2SO4����������غ��֪n��Na2SO4��=n��H2SO4�����ٸ���NaԪ���غ����m��Na������������m��Al�������ݵ���ת���غ�������������������
���
�⣺��1��D�Ǿ��������Եĵ��ʣ�AԪ�����ڶ������������Ԫ�أ����ݷ�Ӧ��ϵ��֪AΪNa��BΪNa2O��DΪO2��CΪNa2O2���ʴ�Ϊ��Na��
��2����D�ǽ������ʣ�D�ڳ�ʪ�Ŀ���������������ʴ��C��Һ�ڱ���ʱӦ�����������D��ֹ����ʣ���DΪFe��BΪ���Σ�CΪ�����Σ�
������Fe������������Һ�������������ӱ������е������������������Ӷ����ʣ����ӷ���ʽΪ��4Fe2++O2+4H+=4Fe3++2H2O����Fe���Ȼ����ˮ��Һ���ɣ�HCl�ӷ���ˮ�⳹���У����յõ����������������ղ�����������ֽ�õ�Fe2O3��
�ʴ�Ϊ��4Fe2++O2+4H+=4Fe3++2H2O��Fe2O3��
��3����A��B��CΪ��ͬһ�ֽ���Ԫ�ص������������Һ��A��C��Ӧ����B��Ӧ����AlԪ�أ�����֪AΪƫ�����Ρ�DΪǿ�ᡢBΪ����������CΪ���Σ�����AΪ���Ρ�DΪǿ�BΪ����������CΪƫ�����Σ�����Bת��ΪC�����п��ܵ����ӷ���ʽΪ��Al��OH��3+3H+=Al3++3H2O ��Al��OH��3+OH-=AlO2-+2H2O��
�ʴ�Ϊ��Al��OH��3+3H+=Al3++3H2O ��Al��OH��3+OH-=AlO2-+2H2O��
��4����Na������Al���ʵĻ����Ͷ��һ������ˮ�г�ַ�Ӧ��A��E��û��ʣ�࣬�õ�������Ϊ��������������Һ����μ���H2SO4��Һ������ɫ�����ﵽ���������ʱ��Һ������ΪNa2SO4����������غ��֪n��Na2SO4��=n��H2SO4��=0.1L��2mol/L=0.2mol����n��Na��=2n��Na2SO4��=0.4mol����m��Na��=0.4mol��23g/mol=9.2g����m��Al��=11.9g-9.2g=2.7g����n��Al��=
=0.1mol�����ݵ���ת���غ��֪���������������ʵ���=
=0.35mol�����������������=0.35mol��22.4L/mol=7.84L��
�ʴ�Ϊ��7.84L��
��2����D�ǽ������ʣ�D�ڳ�ʪ�Ŀ���������������ʴ��C��Һ�ڱ���ʱӦ�����������D��ֹ����ʣ���DΪFe��BΪ���Σ�CΪ�����Σ�
������Fe������������Һ�������������ӱ������е������������������Ӷ����ʣ����ӷ���ʽΪ��4Fe2++O2+4H+=4Fe3++2H2O����Fe���Ȼ����ˮ��Һ���ɣ�HCl�ӷ���ˮ�⳹���У����յõ����������������ղ�����������ֽ�õ�Fe2O3��
�ʴ�Ϊ��4Fe2++O2+4H+=4Fe3++2H2O��Fe2O3��
��3����A��B��CΪ��ͬһ�ֽ���Ԫ�ص������������Һ��A��C��Ӧ����B��Ӧ����AlԪ�أ�����֪AΪƫ�����Ρ�DΪǿ�ᡢBΪ����������CΪ���Σ�����AΪ���Ρ�DΪǿ�BΪ����������CΪƫ�����Σ�����Bת��ΪC�����п��ܵ����ӷ���ʽΪ��Al��OH��3+3H+=Al3++3H2O ��Al��OH��3+OH-=AlO2-+2H2O��
�ʴ�Ϊ��Al��OH��3+3H+=Al3++3H2O ��Al��OH��3+OH-=AlO2-+2H2O��
��4����Na������Al���ʵĻ����Ͷ��һ������ˮ�г�ַ�Ӧ��A��E��û��ʣ�࣬�õ�������Ϊ��������������Һ����μ���H2SO4��Һ������ɫ�����ﵽ���������ʱ��Һ������ΪNa2SO4����������غ��֪n��Na2SO4��=n��H2SO4��=0.1L��2mol/L=0.2mol����n��Na��=2n��Na2SO4��=0.4mol����m��Na��=0.4mol��23g/mol=9.2g����m��Al��=11.9g-9.2g=2.7g����n��Al��=
| 2.7g |
| 27g/mol |
| 0.4mol��1+0.1mol��3 |
| 2 |
�ʴ�Ϊ��7.84L��
���������⿼�������ƶϣ����ڿ�������Ŀ����Ҫѧ����������Ԫ�ػ�����֪ʶ����4��ע�������غ�˼�����Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
��1gN2�к���x�����ӣ����ӵ������ǣ�������
A��
| ||
B��
| ||
| C��14x mol-1 | ||
| D��28x mol-1 |
�����и������ʰ�������������������˳�����У�������ȷ���ǣ�������
| A��Ư�� NaOH SO2 |
| B����ˮ ���� SO2 |
| C������ CuSO4 CaO |
| D���ƾ� Na2O CO |

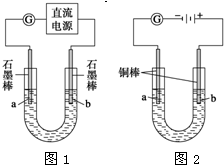
 ��
�� ��11��
��11��
 �������������ɫ�ͳ����µ�Ksp���±���ʾ��
�������������ɫ�ͳ����µ�Ksp���±���ʾ��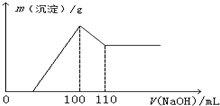 ijѧ��������ȤС��Ϊ�о�ijþ���Ͻ�ijɷ֣�����ƽ��ȡ0.51g�úϽ�����100mL���ᣬ�γ���ɫ��Һ��Ȼ����μ���1mol?L-1 NaOH��Һ�����ɳ���������NaOH��Һ����仯��ͼ��ʾ����
ijѧ��������ȤС��Ϊ�о�ijþ���Ͻ�ijɷ֣�����ƽ��ȡ0.51g�úϽ�����100mL���ᣬ�γ���ɫ��Һ��Ȼ����μ���1mol?L-1 NaOH��Һ�����ɳ���������NaOH��Һ����仯��ͼ��ʾ����