��Ŀ����
ʵ������Na2CO3����500ml 0.1mol?L-1��Na2CO3��Һ���ش��������⣺
��1��Ӧ��������ƽ��ȡ̼���ƾ��� g��
��2������ʱӦѡ�� mL������ƿ��������ƽ���ձ���ҩ�ף�����Ҫ�õ��������� ��
��3������ƿ�ϱ��У����¶ȡ���Ũ�ȡ�����������ѹǿ���ݿ̶��ߡ�����ʽ���ʽ���������е� ���������ַ��ţ�
��4������ƿʹ��ǰ��Ӧ���еIJ����� ��
��5����ʵ��ʱ���������������ʹ��Һ��Ũ��ƫ�ߡ�ƫ�ͻ���Ӱ�죮
A����ˮ����ʱ�����̶��ߣ�
B�����ǽ�ϴ��Һת������ƿ��
C������ƿϴ�Ӻ��ڱ���ˮ���δ�����ﴦ����
D������ʱ���ӿ̶��ߣ� ��
��1��Ӧ��������ƽ��ȡ̼���ƾ���
��2������ʱӦѡ��
��3������ƿ�ϱ��У����¶ȡ���Ũ�ȡ�����������ѹǿ���ݿ̶��ߡ�����ʽ���ʽ���������е�
��4������ƿʹ��ǰ��Ӧ���еIJ�����
��5����ʵ��ʱ���������������ʹ��Һ��Ũ��ƫ�ߡ�ƫ�ͻ���Ӱ�죮
A����ˮ����ʱ�����̶��ߣ�
B�����ǽ�ϴ��Һת������ƿ��
C������ƿϴ�Ӻ��ڱ���ˮ���δ�����ﴦ����
D������ʱ���ӿ̶��ߣ�
���㣺����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺
��������1������n=cV��m=nM�������Ҫ����ع����������
��2������������Һ�����ѡ����ʵ�����ƿ���������Ʋ���ȷ����Ҫ��������
��3����������ƿ�Ĺ����жϽ��
��4�����Ǵ����ӵ�������ʹ��ǰ����Ҫ����Ƿ�©ˮ��
��5�������������������ʵ����ʵ�������Һ�����������C=
������������
��2������������Һ�����ѡ����ʵ�����ƿ���������Ʋ���ȷ����Ҫ��������
��3����������ƿ�Ĺ����жϽ��
��4�����Ǵ����ӵ�������ʹ��ǰ����Ҫ����Ƿ�©ˮ��
��5�������������������ʵ����ʵ�������Һ�����������C=
| n |
| V |
���
�⣺��1��500mL 0.1mol?L-1Na2CO3��Һ�к�������ص����ʵ���Ϊ��0.1mol?L-1��0.5L=0.05mol����Ҫ����ص�����Ϊ��106g/mol��0.05mol=5.3g��
�ʴ�Ϊ��5.3g��
��2����Na2CO3����500ml 0.1mol?L-1��Na2CO3��Һ�IJ���Ϊ�����㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵȣ��õ��������У�������ƽ���ձ���ҩ�ף�500ml����ƿ������������ͷ�ιܣ�
�ʴ�Ϊ��500ml����ƿ������������ͷ�ιܣ�
��3������ƿ�ϱ��У������¶ȡ����������ݿ̶��ߣ�
�ʴ�Ϊ���٢ۢݣ�
��4������ƿ�����ӣ�ʹ�ù�������Ҫ���µߵ�ҡ�ȣ�����ʹ��ǰ�������Ƿ�©ˮ��
�ʴ�Ϊ������Ƿ�©ˮ��
��5��A����ˮ����ʱ�����̶��ߣ�������Һ�����ƫ����Һ��Ũ��ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
B�����ǽ�ϴ��Һת������ƿ���������ʵ����ʵ���ƫС����Һ��Ũ��ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
C������ƿϴ�Ӻ��ڱ���ˮ���δ�����ﴦ���������ʵ����ʵ�������Һ��������������Ӱ�죬��Һ��Ũ�Ȳ��䣬
�ʴ�Ϊ����Ӱ�죻
D������ʱ���ӿ̶��ߣ�������Һ�����ƫС����Һ�����ʵ���Ũ��ƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
�ʴ�Ϊ��5.3g��
��2����Na2CO3����500ml 0.1mol?L-1��Na2CO3��Һ�IJ���Ϊ�����㡢�������ܽ⡢��Һ��ϴ�ӡ����ݡ�ҡ�ȵȣ��õ��������У�������ƽ���ձ���ҩ�ף�500ml����ƿ������������ͷ�ιܣ�
�ʴ�Ϊ��500ml����ƿ������������ͷ�ιܣ�
��3������ƿ�ϱ��У������¶ȡ����������ݿ̶��ߣ�
�ʴ�Ϊ���٢ۢݣ�
��4������ƿ�����ӣ�ʹ�ù�������Ҫ���µߵ�ҡ�ȣ�����ʹ��ǰ�������Ƿ�©ˮ��
�ʴ�Ϊ������Ƿ�©ˮ��
��5��A����ˮ����ʱ�����̶��ߣ�������Һ�����ƫ����Һ��Ũ��ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
B�����ǽ�ϴ��Һת������ƿ���������ʵ����ʵ���ƫС����Һ��Ũ��ƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
C������ƿϴ�Ӻ��ڱ���ˮ���δ�����ﴦ���������ʵ����ʵ�������Һ��������������Ӱ�죬��Һ��Ũ�Ȳ��䣬
�ʴ�Ϊ����Ӱ�죻
D������ʱ���ӿ̶��ߣ�������Һ�����ƫС����Һ�����ʵ���Ũ��ƫ�ߣ�
�ʴ�Ϊ��ƫ�ߣ�
���������⿼��������һ�����ʵ���Ũ�ȵ���Һ��������Ŀ�ѶȲ���ע����������һ�����ʵ���Ũ�ȵ���Һ�������ܹ��������Ʋ���ѡ��ʹ�õ�������

��ϰ��ϵ�д�
�����Ŀ
���з��ӵ�����ԭ���γ�sp2�ӻ�������ǣ�������
| A��H2O |
| B��NH3 |
| C��C2H4 |
| D��BeCl2 |
�����£�99mL0.1mol/L�������101mL0.05mol/L����������Һ��Ϻ���Һ��c��H+��Ϊ��������
�������ǻ��ʱ������仯��
�������ǻ��ʱ������仯��
| A��0.5����10-8+10-10��mol/L |
| B����10-8+10-10��mol/L |
| C����1��10-14-5��10-5��mol/L |
| D��1��10-11 mol/L |



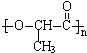 �����γɸø߾���ĵ���ṹ��ʽΪ
�����γɸø߾���ĵ���ṹ��ʽΪ