��Ŀ����
����˵������ȷ����
[ ]
A����֪�����ۻ���Ϊ6.0 kJ��mol-1�������������Ϊ20 kJ��mol-1������1 mol������2 mol ��������ۻ�����ȫ�����ƻ���������������ֻ���ƻ�����15�������
B����֪һ���¶��£�������Һ�����ʵ���Ũ��Ϊc�������Ϊ����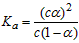 �����������������ƹ��壬��CH3COOH
�����������������ƹ��壬��CH3COOH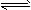 CH3COO-��H+�����ƶ�������С��Ka��С
CH3COO-��H+�����ƶ�������С��Ka��С
C��ʵ���û�����(l)������ϩ(l)�ͱ�(l)�ı�ȼ���ȷֱ�Ϊ��3916 kJ��mol-1����3747 kJ��mol-1��
��3265 kJ��mol-1������֤���ڱ������в����ڶ�����̼̼˫��
D����֪��Fe2O3(s)��3C(ʯī)==2Fe(s)��3CO(g)����H����489.0 kJ��mol-1��
CO(g)��O2(g)==CO2(g)����H����283.0 kJ��mol-1�� C(ʯī)��O2(g)==CO2(g)����H����393.5 kJ��mol-1���� 4Fe(s)��3O2(g)==2Fe2O3(s)����H����1641.0 kJ��mol-1
B����֪һ���¶��£�������Һ�����ʵ���Ũ��Ϊc�������Ϊ����
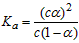 �����������������ƹ��壬��CH3COOH
�����������������ƹ��壬��CH3COOH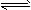 CH3COO-��H+�����ƶ�������С��Ka��С
CH3COO-��H+�����ƶ�������С��Ka��С C��ʵ���û�����(l)������ϩ(l)�ͱ�(l)�ı�ȼ���ȷֱ�Ϊ��3916 kJ��mol-1����3747 kJ��mol-1��
��3265 kJ��mol-1������֤���ڱ������в����ڶ�����̼̼˫��
D����֪��Fe2O3(s)��3C(ʯī)==2Fe(s)��3CO(g)����H����489.0 kJ��mol-1��
CO(g)��O2(g)==CO2(g)����H����283.0 kJ��mol-1�� C(ʯī)��O2(g)==CO2(g)����H����393.5 kJ��mol-1���� 4Fe(s)��3O2(g)==2Fe2O3(s)����H����1641.0 kJ��mol-1
B

��ϰ��ϵ�д�
�����Ŀ
 �ס������ɶ�����Ԫ����ɵij������ʻ���������ѧ��ѧ�����������ʻ�����������ͼ�ת����ϵ������˵������ȷ���ǣ�������
�ס������ɶ�����Ԫ����ɵij������ʻ���������ѧ��ѧ�����������ʻ�����������ͼ�ת����ϵ������˵������ȷ���ǣ�������