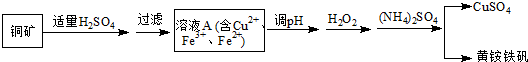��Ŀ����
5����ͼ��Ԫ�����ڱ���һ���֣����еĢ١�����Ԫ�أ��� Ԫ�ط��Ż�ѧʽ��ջش��� ���� | ��A | ��A | ��A | ��A | ��A | ��A | ��A | 0 |
| �� | �� | �� | ||||||
| �� | �� | �� | �� | �� | �� | �� | ||
| �� | �� | �� |
��2���õ���ʽ��ʾ������γɻ�����Ĺ���
 ��
����3����ЩԪ���е�����������Ӧ��ˮ�����У�������ǿ����HClO4��������ǿ����KOH�������Ե�����������Al��OH��3��
��4��д�������������Ʒ�Ӧ�Ļ�ѧ����ʽ��2Al+2NaOH+2H2O=2NaAlO2+3H2����
д���ݵ���������������������Һ��Ӧ�����ӷ���ʽ��Al��OH��3+OH-=AlO2-+2H2O��
���� ��Ԫ�������ڱ���λ�ÿ�֪����ΪN����ΪF����ΪNa����ΪMg����ΪAl����ΪSi����ΪCl����ΪAr����ΪK����ΪBr��
��1��ϡ������Ar�Ļ�ѧ��������ã�
��2��������γɻ���ΪMgF2����þ����������ӹ��ɣ�
��3������Ԫ������������Ӧ��ˮ�����У�������ǿ���Ǹ����ᣬK�Ľ�������ǿ�����������صļ�����ǿ�������Ե���������������������
��4��Al������������Һ��Ӧ����ƫ�������������������������������Ʒ�Ӧ����ƫ��������ˮ��
��� �⣺��Ԫ�������ڱ���λ�ÿ�֪����ΪN����ΪF����ΪNa����ΪMg����ΪAl����ΪSi����ΪCl����ΪAr����ΪK����ΪBr��
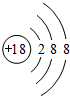
��1��ϡ������Ar�Ļ�ѧ��������ã�ԭ�ӽṹʾ��ͼΪ�� ��
��
�ʴ�Ϊ�� ��
��
��2��������γɻ���ΪMgF2����þ����������ӹ��ɣ��ɵ���ʽ��ʾ���γɹ���Ϊ�� ��
��
�ʴ�Ϊ�� ��
��
��3������Ԫ������������Ӧ��ˮ�����У�������ǿ����HClO4��K�Ľ�������ǿ����KOH�ļ�����ǿ�������Ե�����������Al��OH��3��
�ʴ�Ϊ��HClO4��KOH��Al��OH��3��
��4��Al������������Һ��Ӧ����ƫ����������������Ӧ����ʽΪ��2Al+2NaOH+2H2O=2NaAlO2+3H2����
�����������������Ʒ�Ӧ����ƫ��������ˮ����Ӧ���ӷ���ʽΪ��Al��OH��3+OH-=AlO2-+2H2O��
�ʴ�Ϊ��2Al+2NaOH+2H2O=2NaAlO2+3H2����Al��OH��3+OH-=AlO2-+2H2O��
���� ���⿼��Ԫ�����ڱ���Ԫ�������ɣ��Ƚϻ�������Ҫѧ����������Ԫ�����ڱ��ṹ�����ضԻ�ѧ����Ŀ��飬�����õ���ʽ��ʾ��ѧ�������ʵ��γɹ��̣�

 һ����������ϵ�д�
һ����������ϵ�д�| A�� | Zn�缫�Ϸ����ķ�ӦΪ��2H++2e-�TH2�� | |
| B�� | H+��Zn�缫�ƶ� | |
| C�� | ��Һ��c��H+����С | |
| D�� | ������Cuת�Ƶ�Zn |
| A�� | amol | B�� | bmol | C�� | ��3a-b��mol | D�� | ��a+b��mol |
| A�� | ��ϵͳ��������������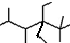 ������ʽ�ṹ����������2��3��5��5-�ļ�-4��4-���һ����� ������ʽ�ṹ����������2��3��5��5-�ļ�-4��4-���һ����� | |
| B�� | ����ϩ�뻯���� ������ʽ�ṹ����Ϊͬϵ�� ������ʽ�ṹ����Ϊͬϵ�� | |
| C�� | ����ʽΪC4H8O2���л��������������¿�ˮ��Ϊ��ʹ����������п��ܵõ��Ĵ������������������γɵ�������16�� | |
| D�� | �����ʵ����ı��ͱ�������ȫȼ����������������� |
| A�� | �������NaAlO2��Һ�� | B�� | CO2����ͨ��Ư����Һ�� | ||
| C�� | NaOH��Һ����AlCl3��Һ�� | D�� | Ba��OH��2��Һ����Al2��SO4��3��Һ�� |
| ������ | K+��Ag+��Mg2+��Ba2+ |
| ������ | NO3- ��CO32- ��SiO32- ��SO42- |
| ��� | ʵ������ | ʵ���� |
| �� | �����Һ�м�������ϡ���� | ������ɫ�������ų���״����0.56L���� |
| �� | ����ķ�Ӧ���Һ���ˣ��Գ���ϴ�ӡ����������أ��������ù������� | ��������Ϊ2.4g |
| �� | ������Һ�еμ�BaCl2��Һ | ���������� |
��1��ʵ�����ȷ��һ�������ڵ�������Ag+��Mg2+��Ba2+��
��2��ʵ��������ɳ��������ӷ���ʽΪSiO32-+2H+=H2SiO3����
��3��ͨ��ʵ���ͱ�Ҫ���㣬��д�±��������ӵ�Ũ�ȣ��ܼ�����ģ���д��������һ�������ڵ������0��������ȷ���Ƿ���ڵ��������������
| ������ | NO3- | CO32- | SiO32- | SO42- |
| c��mol•h-1�� |
| A�� | H | B�� | Na | C�� | Al | D�� | Si |
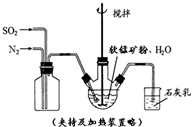 �ߴ�MnCO3���Ʊ������ܴ��Բ��ϵ���Ҫԭ�ϣ�ij��ѧС����ʵ����ģ�������̿���Ҫ�ɷ�MnO2������Ϊ����ͭ�Ļ�����ȣ��Ʊ��ߴ�̼���̣��������£����ֲ����������ԣ���
�ߴ�MnCO3���Ʊ������ܴ��Բ��ϵ���Ҫԭ�ϣ�ij��ѧС����ʵ����ģ�������̿���Ҫ�ɷ�MnO2������Ϊ����ͭ�Ļ�����ȣ��Ʊ��ߴ�̼���̣��������£����ֲ����������ԣ����ٻ�������ƿ�У���ͼ��ͨ�������������С����̡���������Ҫ��Ӧԭ��Ϊ��SO2+H2O=H2SO3��MnO2+H2SO3=MnSO4+H2O��������������SO2�ὫFe3+��ԭΪFe2+��
�����̡����������ƿ�м���һ������MnO2��ĩ��
������Na2CO3��Һ����pHΪ3.5���ң����ˣ�
�ܵ�����ҺpHΪ6.5��7.2������NH4HCO3������ɫ��ζ������ų���ͬʱ��dz��ɫ�ij������ɣ������ˡ�ϴ�ӡ�����õ��ߴ�̼���̣���ش�
��1��ʯ������뷴Ӧ�Ļ�ѧ����ʽΪCa��OH��2+SO2�TCaSO3+H2O��
��2�������̡���Ӧ�������и�����MnS2O6�����ɣ��¶ȶԡ����̡���Ӧ��Ӱ������ͼ��Ϊ����MnS2O6 �����ɣ������̡��������˵��¶���150�棨��150�����ϣ���

��3�������±��ش���м���һ������MnO2��ĩ����Ҫ�����ǽ�Fe2+����ΪFe3+����������SO2������ȥ��
| ���� | Fe��OH��3 | Fe��OH��2 | Mn��OH��2 | Cu��OH��2 |
| ��ʼ����pH | 2.7 | 7.6 | 8.3 | 4.7 |
| ��ȫ����pH | 3.7 | 9.6 | 9.8 | 6.7 |
��5�����м���NH4HCO3������Ӧ�����ӷ���ʽ��Mn2++2HCO3-=MnCO3��+CO2��+H2O��
��6����֪MnCO3������ˮ���Ҵ�����ʪʱ�ױ�����������100�濪ʼ�ֽ⣮����������C2H5OHϴ�ӵ�ԭ���Ƿ�ֹMnCO3��ʪʱ�������������������¶ȵ���100�����ȣ�