��Ŀ����
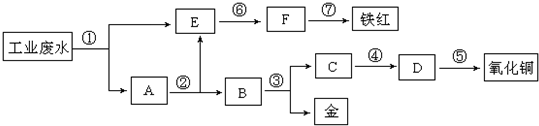
ij���������Թ�ҵ��ˮ�к���һ������Fe3+��Cu2+��Au3+�����ӣ����������ͼ�еĹ������̣����ó��õ��ᡢ���ҵ�����еķ���м���ӷ�ˮ�л��ս𣬲�����һ���������죨Fe2O3��������ͭ������֪ͭ���ܽ���ϡ��������������ͭ��Һ��
����д����հף�
��1��ͼ�б�Ŵ���������Ӧ���ʷֱ��ǣ��ѧʽ���� �� �� �� ��
��2��A������ɷ�Ϊ ���ѧʽ��
��3��д���ݴ���Ӧ�Ļ�ѧ����ʽ ��

����д����հף�
��1��ͼ�б�Ŵ���������Ӧ���ʷֱ��ǣ��ѧʽ����
��2��A������ɷ�Ϊ
��3��д���ݴ���Ӧ�Ļ�ѧ����ʽ
���㣺������ƶ�
ר�⣺�ƶ���
�������ɹ�������ͼʾ֪��EΪ��Ԫ�ص����ӣ���AΪͭ����ĵ��ʣ�ͨ�����˽���Ԫ��������Ա�������ͭ��������������ˢٴ����������Ϊ��м�������ķ�ӦΪFe+2H+�TFe2++H2����2Fe3++Fe�T3Fe2+��Cu2++Fe�TCu+Fe2+��2Au3++3Fe�T2Au+3Fe2+���ڴ�����ϡ�����Գ�ȥͭ�����й�������м���������˺������������Һ�뺬Fe2+��E��Һ���ϣ��۴����ý�������������ʽ�ͭ�����뿪���ܴ����õ���ͭ��������������ת��Ϊ������ͭ�������ݴ������ȷֽ�Ϊ����ͭ���������������ƽ���������ת��Ϊ���������������������ÿ�������ת��Ϊ���������������ߴ��������ȷֽ�Ϊ���죨�����������ݴ˽��
���
�⣺�ɹ�������ͼʾ֪��EΪ��Ԫ�ص����ӣ���AΪͭ����ĵ��ʣ�ͨ�����˽���Ԫ��������Ա�������ͭ��������������ˢٴ����������Ϊ��м�������ķ�ӦΪFe+2H+�TFe2++H2����2Fe3++Fe�T3Fe2+��Cu2++Fe�TCu+Fe2+��2Au3++3Fe�T2Au+3Fe2+���ڴ�����ϡ�����Գ�ȥͭ�����й�������м���������˺������������Һ�뺬Fe2+��E��Һ���ϣ��۴����ý�������������ʽ�ͭ�����뿪���ܴ����õ���ͭ��������������ת��Ϊ������ͭ�������ݴ������ȷֽ�Ϊ����ͭ���������������ƽ���������ת��Ϊ���������������������ÿ�������ת��Ϊ���������������ߴ��������ȷֽ�Ϊ���죨����������
��1��������������֪��������������Ӧ���ʣ���ΪFe����ΪH2SO4����ΪHNO3����ΪNaOH��
�ʴ�Ϊ��Fe��H2SO4��HNO3��NaOH��
��2��������������֪��A������ɷ�Ϊ��Cu��Fe��Au���ʴ�Ϊ��Cu��Fe��Au��
��3���ݴ���Ӧ�Ļ�ѧ����ʽΪ��Cu��OH��2
CuO+H2O���ʴ�Ϊ��Cu��OH��2
CuO+H2O��
��1��������������֪��������������Ӧ���ʣ���ΪFe����ΪH2SO4����ΪHNO3����ΪNaOH��
�ʴ�Ϊ��Fe��H2SO4��HNO3��NaOH��
��2��������������֪��A������ɷ�Ϊ��Cu��Fe��Au���ʴ�Ϊ��Cu��Fe��Au��
��3���ݴ���Ӧ�Ļ�ѧ����ʽΪ��Cu��OH��2
| ||
| ||
���������⿼��������ƶϡ������������仯������ۺ�Ӧ�ã����������ʵķ����ᴿ�Ŀ��飬��Ҫѧ����������̣�����Ԫ�ػ��������ʣ��Ѷ��еȣ�

��ϰ��ϵ�д�
 53���ò�ϵ�д�
53���ò�ϵ�д�
�����Ŀ
��Ȼ��֬�ṹ�ĺ���Ϊ����[HOCH2CH��OH��CH2OH]����һ�����õķ���Ȼ��֬����ṹ�ĺ�����Ϊ���ǣ�C12H22O11�����÷���Ȼ��֬����ֱ���͵IJ��������ᣨC17H33COOH�������Ƿ�Ӧ���ã��䷴Ӧʾ��ͼ��ͼ��ʾ��ע�⣺ͼ�еķ�Ӧʽ������������˵����ȷ���ǣ�������
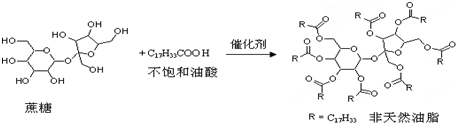

| A��������������֬������ |
| B������Ȼ��֬Ϊ�߷��ӻ����� |
| C���÷���Ȼ��֬��ϡ���Ṳ�ȣ���ˮ������ڳ����¾�Ϊ��̬ |
| D������������ϡ�����������ˮ�⣬���տ����������л������� |
����˵����ȷ���ǣ�������
| A�������22.4L H2O�� 1molO2������������� |
| B��1molˮ�к���2mol���1mol�� |
| C��1mol��̬ˮ��1molҺ̬ˮ�����ķ������� |
| D��3molO2 ��2molH2O����ԭ������� |
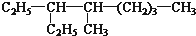
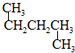 ��
��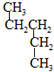
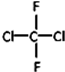 ��
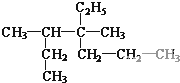
��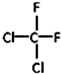 ��CH3CH2CH2CH3��
��CH3CH2CH2CH3�� 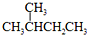
 ��ͼ��ʾ������I�̶�������������������ƶ���M��N�����������������·�Ӧ��A��g��+3B��g��?2C��g��
��ͼ��ʾ������I�̶�������������������ƶ���M��N�����������������·�Ӧ��A��g��+3B��g��?2C��g��