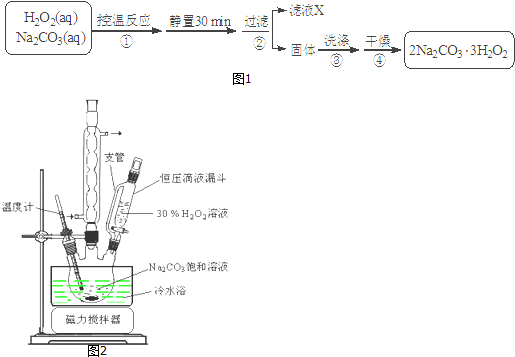
��Ŀ����
20�����ڱ��еڢ�A��Ԫ�ض�Ϊ����Ԫ�أ���1������Ϊ�ڢ�AԪ�أ���ȴ��ڢ�A����Ԫ�����ʸ����������ߵ��Ȼ��ﶼΪ���ۻ���������γɹ��۷��ӣ�

���ڶ���Ȼ����У���ԭ�ӵĹ���ӻ�����Ϊsp3�ӻ���AlCl3����Cl-�������[AlCl4]�ĽṹʽΪ��������λ�����á�������ʾ��
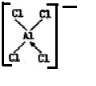 ��[AlCl4]�����幹������Ϊ���������Σ�
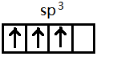
��[AlCl4]�����幹������Ϊ���������Σ�����[AlCl4]�У���ԭ�Ӽ۵������ӻ�����е��Ų�ͼΪ
 ��������Ԫ�����������ԭ����Be��Al�ĵ縺�������
��������Ԫ�����������ԭ����Be��Al�ĵ縺���������2��þ����ͬΪ��������Ԫ�أ���þ�ĵ�����ΪI1��I2��I3�������ĵ�����ΪI1�䡢I2�䡢I3�䡭�����й���þ���������ܵ���������ȷ����B
A��I1��I2��I3��I4 B��I3+I2��I2+I1 C��I2-I1�䣼I2-I1 D��I2�䣾I2��I1�䣼I1
��3��̼���ε������Ӳ�ͬ���ȷֽ���¶Ȳ�ͬ��MgCO3��CaCO3��SrCO3��BaCO3�����ȶ�����ǿ������˳��ΪBaCO3��SrCO3��CaCO3��MgCO3����ԭ����̼���ηֽ�ı���ΪCO32-����CO2��O2-��O2-����������ӽ�ϵĹ��̣���MgO������������ȶ�����MgCO3���ֽ⣨�����Ӿ����������ӵİ뾶ԽС�����̼����е�������Խ���ף�
��4�����ǹ��ɿ������ʯ����ҪԪ�أ��ҽ���MgSiO3��CaSiO3��SrSiO3��BaSiO3�������Ⱥ�˳��ΪMgSiO3��CaSiO3��SrSiO3��BaSiO3
��5��þ����Ķѻ���ʽΪABA�ͣ��侧���ṹ��ͼ2��ʾ���öѻ���ʽ�Ŀռ�������Ϊ$\frac{\sqrt{2}��}{6}$���ú��е�ʽ�ӱ�ʾ��
���� ��1���ٸ����Ȼ���Ľṹͼ��֪����ԭ����Χ���ĸ����ۼ����ṩ�ĸ����ڳɼ��Ĺ����[AlCl4]�Ľṹ�У�ÿ����ԭ�Ӻ��ĸ���ԭ���γɹ��ۼ���������һ�����õ��Ӷ�����ԭ���ṩ�γɵ���λ���������ӻ�����������ж�[AlCl4]�����幹�ͣ�
����[AlCl4]�У���ԭ�Ӽ۵���Ϊsp3�ӻ����ݴ���д�Ų�ͼ����ЩԪ�صĻ�ѧ���ʺ��������ڱ������Ϸ������·���һ����Ԫ�ص縺��������������ƣ��ݴ˷���������Ԫ�����������ԭ��
��2����̬ԭ�ӻ���̬����ʧȥһ����������Ҫ����С������ΪԪ�صĵ����ܣ�ͬ�����������Ԫ�صĵ�һ�����ܳ��������ƣ�����A�������Ϊns�ܼ�����2�����ӣ�Ϊȫ��ȷ��״̬�������ϵͣ���һ�����ܸ���ͬ��������Ԫ�أ�һ��ʧȥһ������Ϊ��һ�����ܣ��Դ����ƣ�
��3��̼���ηֽ�ı���ΪCO32-����CO2��O2-�����Ӱ뾶ԽС�����Ӽ�Խǿ��������Խ�����ȶ���Խ�ã�
��4�������۵�ϸ��������ҽ���ȴ�������Ƚᾧ��������Խ�����Ӽ�Խǿ���۵�Խ�ߣ�
��5��þ����Ķѻ���ʽΪABA�ͣ������������ܶѻ�������ʽ�Ŀռ�������Ϊ=$\frac{V��}{V����}$���ݴ˼���ռ������ʣ�
��� �⣺��1��?�����Ȼ���Ľṹͼ��֪����ԭ����Χ���ĸ����ۼ�����������������λ����������Ҫ����sp3�ӻ����Ӷ��ṩ�ĸ����ڳɼ��Ĺ������ԭ����������ֻ��3�����ӣ��γ�3�����ۼ���[AlCl4]�Ľṹ�У�ÿ����ԭ�Ӻ��ĸ���ԭ���γɹ��ۼ���������һ�����õ��Ӷ�����ԭ���ṩ�γɵ���λ�����ṹʽΪ�� ������ԭ��Al�ļ۲���Ӷ���4���ӻ�����Ϊsp3�����幹��Ϊ�����������Σ�
������ԭ��Al�ļ۲���Ӷ���4���ӻ�����Ϊsp3�����幹��Ϊ�����������Σ�
�ʴ�Ϊ��sp3��  �����������Σ�
������������
?����λ�����ڱ��е������ڵ�IIIA�����������Ų�ʽΪ1s22s22p63s23p1��[Ne]3s23p1����[AlCl4]�У��ṹʽΪ�� ������ԭ��Al�ļ۲���Ӷ���4���ӻ�����Ϊsp3�����Լ۵������ӻ�����е��Ų�ͼΪ
������ԭ��Al�ļ۲���Ӷ���4���ӻ�����Ϊsp3�����Լ۵������ӻ�����е��Ų�ͼΪ ��Al��Be�����ڱ��д��Խ��ߣ��縺��������������ƣ���ǿ�ᡢǿ�����Ӧ��
��Al��Be�����ڱ��д��Խ��ߣ��縺��������������ƣ���ǿ�ᡢǿ�����Ӧ��
�ʴ�Ϊ�� �� Be��Al�ĵ縺�������
�� Be��Al�ĵ縺�������
��2��A��þ�ĵ�����ΪI1��I2��I3����ʧȥ��һ�����ӱȵڶ��������������ͣ��Դ����ƣ�����I1��I2��I3��I4����A��ȷ��
B����I3+I2��I2+I1 ��֪I3��I1����ʧȥ��һ�����ӱȵ�����������������ì�ܣ���B����
C��ͬ���ڴ����ҵ�һ��������������þ�������⣬������þ�ļ۵����Ų���3s2��3p���ȫ�ս��ȶ���������3s23p1����ȫ����ȫ�գ����������һ����������ȶ��������ĵ�һ�����ܱ�þС������I2-I1�䣼I2-I1����C��ȷ��
D�����ĵ�һ�����ܱ�þС������I1�䣼I1��3p���ȫ�ս��ȶ�������I2�䣾I2����D��ȷ��
�ʴ�Ϊ��B��
��3��̼���ηֽ�ı���ΪCO32-����CO2��O2-��O2-����������ӽ�ϵĹ��̣�MgO��CaO��SrO��BaO����������ͬ��þ���Ӱ뾶��С��MgO������������ȶ�����MgCO3���ֽ⣨�����Ӿ����������ӵİ뾶ԽС�����̼����е�������Խ���ף�������MgCO3��CaCO3��SrCO3��BaCO3�����ȶ�����ǿ������˳��ΪBaCO3��SrCO3��CaCO3��MgCO3��
�ʴ�Ϊ��BaCO3��SrCO3��CaCO3��MgCO3��̼���ηֽ�ı���ΪCO32-����CO2��O2-��O2-����������ӽ�ϵĹ��̣���MgO������������ȶ�����MgCO3���ֽ⣨�����Ӿ����������ӵİ뾶ԽС�����̼����е�������Խ���ף���
��4��MgSiO3��CaSiO3��SrSiO3��BaSiO3��Ϊ���Ӿ��壬��������ͬ����ΪSiO32-�������Ӱ뾶��Mg 2+��Ca2+��Sr2+��Ba2+�����Ծ�����MgSiO3��CaSiO3��SrSiO3��BaSiO3�������۵�ϸ��������ҽ���ȴ�������Ƚᾧ�������ҽ���MgSiO3��CaSiO3��SrSiO3��BaSiO3�������Ⱥ�˳��Ϊ��MgSiO3��CaSiO3��SrSiO3��BaSiO3��
�ʴ�Ϊ��MgSiO3��CaSiO3��SrSiO3��BaSiO3��
��5��þ����Ķѻ���ʽΪABA�ͣ������������ܶѻ�����ƽ�������θ��߳�Ϊa=2r����ͼ��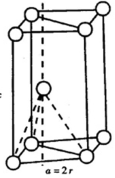 ��ƽ���ı��ε����s=a��asin60��=$\frac{\sqrt{3}}{2}$a2��h=2���߳�Ϊa��������ĸ�=2��$\frac{\sqrt{6}}{3}$a=$\frac{2\sqrt{6}}{3}$a��V����=Sh=$\frac{\sqrt{3}}{2}$a2��$\frac{2\sqrt{6}}{3}$a=$\sqrt{2}$a3=8$\sqrt{2}$r3��V��=2��$\frac{4}{3}$���С�r3���öѻ���ʽ�Ŀռ�������Ϊ=$\frac{V��}{V����}$=$\frac{\sqrt{2}��}{6}$��
��ƽ���ı��ε����s=a��asin60��=$\frac{\sqrt{3}}{2}$a2��h=2���߳�Ϊa��������ĸ�=2��$\frac{\sqrt{6}}{3}$a=$\frac{2\sqrt{6}}{3}$a��V����=Sh=$\frac{\sqrt{3}}{2}$a2��$\frac{2\sqrt{6}}{3}$a=$\sqrt{2}$a3=8$\sqrt{2}$r3��V��=2��$\frac{4}{3}$���С�r3���öѻ���ʽ�Ŀռ�������Ϊ=$\frac{V��}{V����}$=$\frac{\sqrt{2}��}{6}$��
�ʴ�Ϊ��$\frac{\sqrt{2}��}{6}$��
���� ���⿼���˽�������ṹ����Ŀ�Ѷ��еȣ�ע�����յ��;��徧���ṹ����ȷ�ѻ���ʽ�Ŀռ������ʵĺ����ǽ��5���ؼ���Ϊѧϰ���ѵ㣬�ӻ����������ʽṹѧϰ���ص㣮

 100�ִ�����ĩ���ϵ�д�
100�ִ�����ĩ���ϵ�д� ��У���˿��ֿ���ϵ�д�
��У���˿��ֿ���ϵ�д� ������֪����1molN-N����167kJ����������1molN��N�ų�942kJ����������������Ϣ�����ݣ�����˵����ȷ���ǣ�������
������֪����1molN-N����167kJ����������1molN��N�ų�942kJ����������������Ϣ�����ݣ�����˵����ȷ���ǣ�������| A�� | 1molN4������ȫת��ΪN2���ų�882kJ���� | |
| B�� | 14N2��15N2��Ϊͬλ�أ�N4��N2��Ϊͬ�������� | |
| C�� | N4����һ�����͵Ļ����� | |
| D�� | 14N��15N��ѧ���ʲ���ͬ |
| A�� | �ı��¶Ⱥ�ʹ�ô������ܸı仯ѧ��Ӧ���� | |
| B�� | ���ö����ЧӦ��������FeCl3��Һ��Fe��OH��3���� | |
| C�� | ���ݡ�G=��H-T��S�Ʋ⣺��H��0�Ļ�ѧ��Ӧ�������Է����� | |
| D�� | ��AlCl3��Һ���ɲ����գ����ù���ΪAl2O3 |
| A�� | ������ǻ벻�£�Ҫ��������˼� | B�� | ���������Ϊͭ | ||
| C�� | ���ϵ���˿����������ɻ���ʼ�� | D�� | ˮ��ʯ�� |
| A�� | ������������Ȼ�л��߷��ӻ����û�е����ʾ�û������ | |
| B�� | HCHO��Һ��NH4��2SO4��Һ����ʹ�����ʱ��� | |
| C�� | ijЩ�����ʸ�Ũ�������û��� | |
| D�� | ���Բ��ö���������������ķ������롢�ᴿ������ |
| A�� | ������̼ ̼���� | B�� | ������ �Ҵ� | ||
| C�� | ������ �� | D�� | ���� ���� |
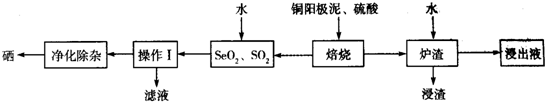

 ��Q3Y2��ˮ�ɾ��ҷ�Ӧ���������������壬��Ӧ�Ļ�ѧ����ʽΪMg3N2+6H2O=3Mg��OH��2+2NH3����
��Q3Y2��ˮ�ɾ��ҷ�Ӧ���������������壬��Ӧ�Ļ�ѧ����ʽΪMg3N2+6H2O=3Mg��OH��2+2NH3����