��Ŀ����
��������ƣ�Na2S2O3�������������ƺ����ͨ�����Ϸ�Ӧ�Ƶã�Na2SO3+S
Na2S2O3����������Һ����������ΪNa2S2O3?5H2O��Na2S2O3?5H2O��40��45���ۻ���48��ֽ⣻Na2S2O3 ������ˮ���������Ҵ�����ˮ���й����ʵ��ܽ��������ͼ1��ʾ��
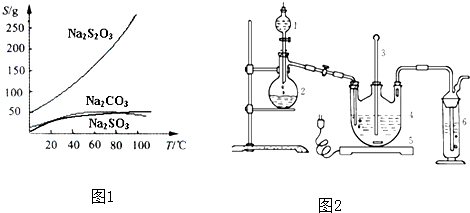
���ְ����·����Ʊ�Na2S2O3?5H2O��
�����ƺ�̼���ư���ӦҪ�����һ������������ƿ�У�ע��150mL����ˮʹ���ܽ⣬�ڷ�Һ©���У�ע��Ũ���ᣬ��װ��2�м����������ƹ��壬��������ͼ��װ��װ�ã���Һ©��������ע��Ũ����ʹ��Ӧ�����Ķ�����������Ͼ��ȵ�ͨ��Na2S��Na2CO3�Ļ����Һ�У����ô������������������ȣ���Ӧԭ��Ϊ��
��Na2CO3+SO2�TNa2SO3+CO2
��Na2S+SO2+H2O�TNa2SO3+H2S
��2H2S+SO2�T3S��+2H2O
��Na2SO3+S
Na2S2O3
�ܷ�ӦΪ��2Na2S+Na2CO3+4SO2�T3Na2S2O3+CO2
��1������2������Ϊ ��
��2��װ��6�пɷ��� ��
A��BaCl2��Һ B��ŨH2SO4 C������KMnO4��Һ D��NaOH��Һ����Na2S2O3?5H2O��
�ȵķ�Ӧ���Һ
��Һ
�־���
��3��Ϊ���ٲ�Ʒ����ʧ��������Ϊ ���������dz���ϴ�Ӹ������ϴ�Ӳ������� �����Լ�����ϴ�Ӽ���
��4������Ũ����Һֱ����Һ����ɫ����Ϊֹ������ʱ�����¶Ȳ��˹��ߵ�ԭ���� ��
��Na2S2O3���ʵļ��鼰��Ӧ��
��1����������������ˮ�еμ�Na2S2O3��Һ����ˮ��ɫ��dz��д���÷�Ӧ�����ӷ���ʽ ��
��2������Na2S2O3��Һ�ⶨ��ˮ��Ba2+Ũ�ȣ��������£�ȡ��ˮ25.00mL�������ʵ�����ȼ�������K2Cr2O7��Һ����BaCrO4���������ˡ�ϴ�Ӻ�������ϡ�����ܽ⣬��ʱCrO42-ȫ��ת��ΪCr2O72-���ټӹ���KI��Һ����ַ�Ӧ��û����ҺV mL������ƽ���ֳ�4�ȷݣ����������Һ��ָʾ������0.0010mol?L-1 ��Na2S2O3��Һ���еζ�����Ӧ��ȫʱ��������ݼ�¼���±���ʾ��
���ַ�Ӧ���ӷ���ʽΪ����Cr2O72-+6I-+14H+�T2Cr3++3I2+7H2O ��2S2O32-+I2�TS4O62-+2I-
���жϴﵽ�ζ��յ�������� ����ˮ��Ba2+�����ʵ���Ũ��Ϊ ��
| ||

���ְ����·����Ʊ�Na2S2O3?5H2O��
�����ƺ�̼���ư���ӦҪ�����һ������������ƿ�У�ע��150mL����ˮʹ���ܽ⣬�ڷ�Һ©���У�ע��Ũ���ᣬ��װ��2�м����������ƹ��壬��������ͼ��װ��װ�ã���Һ©��������ע��Ũ����ʹ��Ӧ�����Ķ�����������Ͼ��ȵ�ͨ��Na2S��Na2CO3�Ļ����Һ�У����ô������������������ȣ���Ӧԭ��Ϊ��
��Na2CO3+SO2�TNa2SO3+CO2
��Na2S+SO2+H2O�TNa2SO3+H2S
��2H2S+SO2�T3S��+2H2O
��Na2SO3+S
| ||
�ܷ�ӦΪ��2Na2S+Na2CO3+4SO2�T3Na2S2O3+CO2
��1������2������Ϊ
��2��װ��6�пɷ���
A��BaCl2��Һ B��ŨH2SO4 C������KMnO4��Һ D��NaOH��Һ����Na2S2O3?5H2O��
�ȵķ�Ӧ���Һ
| ����̿��ɫ |
| ������ |
| ������ |
| ����Ũ�� |
| ��ȴ���� |
| ������ |
��3��Ϊ���ٲ�Ʒ����ʧ��������Ϊ
��4������Ũ����Һֱ����Һ����ɫ����Ϊֹ������ʱ�����¶Ȳ��˹��ߵ�ԭ����
��Na2S2O3���ʵļ��鼰��Ӧ��
��1����������������ˮ�еμ�Na2S2O3��Һ����ˮ��ɫ��dz��д���÷�Ӧ�����ӷ���ʽ
��2������Na2S2O3��Һ�ⶨ��ˮ��Ba2+Ũ�ȣ��������£�ȡ��ˮ25.00mL�������ʵ�����ȼ�������K2Cr2O7��Һ����BaCrO4���������ˡ�ϴ�Ӻ�������ϡ�����ܽ⣬��ʱCrO42-ȫ��ת��ΪCr2O72-���ټӹ���KI��Һ����ַ�Ӧ��û����ҺV mL������ƽ���ֳ�4�ȷݣ����������Һ��ָʾ������0.0010mol?L-1 ��Na2S2O3��Һ���еζ�����Ӧ��ȫʱ��������ݼ�¼���±���ʾ��
| ��� | 1 | 2 | 3 | 4 |
| ����Na2S2O3����Һ�����/mL | 18.02 | 17.98 | 18.00 | 20.03 |
���жϴﵽ�ζ��յ��������
���㣺�Ʊ�ʵ�鷽�������
ר�⣺ʵ�������
��������1����������2�Ĺ��켰ʹ�÷���д�������ƣ�
��2������װ��6��ʵ���е�����ѡ���Լ���
��3�������ٵ�Ŀ���Ƿ��������������ɫ���ʵĻ���̿����ò���Ϊ���ˣ����ڳ�������Һ����������Na2S2O3?5H2O��Ϊ�˱����Ʒ��ʧ����Ҫ���ȹ��ˣ�Na2S2O3������ˮ���������Ҵ���Ϊ�˼�����ʧ���������Ҵ�Ϊϴ�Ӽ���
��4�����ݡ�Na2S2O3?5H2O��40��45���ۻ���48��ֽ⡱��������ʱҪ�����¶Ȳ��˹��ߵ�ԭ��
��1��Na2S2O3��������ˮ��Ӧ��������ǿ���������������������Ԫ�ر�����Ϊ���������������Ԫ�ر���ԭΪCI-�����ӷ���ʽΪS2O32-+4Cl2+5H2O=2SO42-+8Cl-+10H+��
��2�����ɵĵⵥ���������۱���������������Ƶζ�ʱ������ɫ��ȥ����Ӳ��仯��˵����Ӧ�ﵽ�յ㣻���ݷ�Ӧ����ʽ�и����ʼ�����֮��Ĺ�ϵ���㣮
����n=cV�����1.28g��Ʒ���ĵĸ�����ص����ʵ��������ݸ��ݷ�Ӧ�������Ʒ�к���Na2S2O3?5H2O�����ʵ������ٸ������������ı���ʽ�������Ʒ��Na2S2O3?5H2O������������
��2������װ��6��ʵ���е�����ѡ���Լ���
��3�������ٵ�Ŀ���Ƿ��������������ɫ���ʵĻ���̿����ò���Ϊ���ˣ����ڳ�������Һ����������Na2S2O3?5H2O��Ϊ�˱����Ʒ��ʧ����Ҫ���ȹ��ˣ�Na2S2O3������ˮ���������Ҵ���Ϊ�˼�����ʧ���������Ҵ�Ϊϴ�Ӽ���
��4�����ݡ�Na2S2O3?5H2O��40��45���ۻ���48��ֽ⡱��������ʱҪ�����¶Ȳ��˹��ߵ�ԭ��
��1��Na2S2O3��������ˮ��Ӧ��������ǿ���������������������Ԫ�ر�����Ϊ���������������Ԫ�ر���ԭΪCI-�����ӷ���ʽΪS2O32-+4Cl2+5H2O=2SO42-+8Cl-+10H+��
��2�����ɵĵⵥ���������۱���������������Ƶζ�ʱ������ɫ��ȥ����Ӳ��仯��˵����Ӧ�ﵽ�յ㣻���ݷ�Ӧ����ʽ�и����ʼ�����֮��Ĺ�ϵ���㣮
����n=cV�����1.28g��Ʒ���ĵĸ�����ص����ʵ��������ݸ��ݷ�Ӧ�������Ʒ�к���Na2S2O3?5H2O�����ʵ������ٸ������������ı���ʽ�������Ʒ��Na2S2O3?5H2O������������
���
�⣺��1������ͼʾװ�ÿ�֪������2������Ϊ������ƿ���ʴ�Ϊ��������ƿ��
��2��װ��6��β������װ����Ҫ���ն���������Ⱦ�����壬ѡ��������KMnO4��Һ���������ԣ��������������������������գ�����������Һ�Ͷ�������Ӧ�����������ƺ�ˮ�������ն�������Ũ���ᡢ�Ȼ������������Ӧ���������ն�����������CD��ȷ��
�ʴ�Ϊ��CD��
��3����������Һ����������ΪNa2S2O3?5H2O��Na2S2O3?5H2O��40��45���ۻ���Ϊ�˱�������Na2S2O3?5H2O���²��ʽ��ͣ����Բ����ٹ��˳�����̿ʱ��Ҫ���ȹ��ˣ�
ϴ�Ӿ���ʱΪ���پ�����ʧ������Na2S2O3?5H2O���ܽ⣬����Na2S2O3������ˮ���������Ҵ�������ѡ���Ҵ�ϴ�ӣ���ϴ�Ӻ��Ҵ��ӷ����������µ����ʣ�
�ʴ�Ϊ�����ȹ��ˣ��Ҵ���
��4������Na2S2O3?5H2O��40��45���ۻ���48��ֽ⣬��������ʱ�¶ȹ��ᵼ�������ľ���ֽ⣬�����˲��ʣ�
�ʴ�Ϊ���¶ȹ��ᵼ�������ľ���ֽ⣻
��1������������ԭ��Ӧ��ʵ�ʵ�ʧ�����غ㣬S2O32-��2SO42-��8e-��Cl2��2CI-��2e-������4molCl2����1molS2O32-���õ�8molCl-��2molSO42-�����ݵ���غ㣬��������Ӧ��10molH+������ԭ���غ㷴Ӧ����Ӧ��5molH2O�����ӷ���ʽΪS2O32-+4Cl2+5H2O=2SO42-+8Cl-+10H+���ʴ�Ϊ��S2O32-+4Cl2+5H2O=2SO42-+8Cl-+10H+��
��2�����ݱ궨��ԭ����֪�����ɵĵⵥ���������۱���������������Ƶζ�����ɫ��ȥ����Ӳ��仯��˵����Ӧ�ﵽ�յ㣬���Եζ��յ������Ϊ���������һ��Na2S2O3����Һ����ɫ��ʧ���Ұ��������ɫ���ı䣻
��Һ�з����ķ�ӦΪ��2Ba2++Cr2O72-=2BaCrO4+2H+�� Cr2O72-+6I-+14H+=2Cr3++3I2+7H2O�� I2+2S2O32-=2I-+S4O62-��
���ݷ���ʽ�г������ʵĹ�ϵΪBa2+��BaCrO4��
Cr2O72-��
I2��3S2O32-
1mol 3mol
��n��Ba2+��=3n��S2O32-��=
��0.0010mol?L-1��18.00��10-3 L=6��10-6mol
����c��Ba2+���T
=0.00024mol?L-1��
�ʴ�Ϊ���������һ��Na2S2O3����Һ����ɫ��ʧ���Ұ��������ɫ���ı䣻 0.00024mol?L-1��
��2��װ��6��β������װ����Ҫ���ն���������Ⱦ�����壬ѡ��������KMnO4��Һ���������ԣ��������������������������գ�����������Һ�Ͷ�������Ӧ�����������ƺ�ˮ�������ն�������Ũ���ᡢ�Ȼ������������Ӧ���������ն�����������CD��ȷ��
�ʴ�Ϊ��CD��
��3����������Һ����������ΪNa2S2O3?5H2O��Na2S2O3?5H2O��40��45���ۻ���Ϊ�˱�������Na2S2O3?5H2O���²��ʽ��ͣ����Բ����ٹ��˳�����̿ʱ��Ҫ���ȹ��ˣ�
ϴ�Ӿ���ʱΪ���پ�����ʧ������Na2S2O3?5H2O���ܽ⣬����Na2S2O3������ˮ���������Ҵ�������ѡ���Ҵ�ϴ�ӣ���ϴ�Ӻ��Ҵ��ӷ����������µ����ʣ�
�ʴ�Ϊ�����ȹ��ˣ��Ҵ���
��4������Na2S2O3?5H2O��40��45���ۻ���48��ֽ⣬��������ʱ�¶ȹ��ᵼ�������ľ���ֽ⣬�����˲��ʣ�
�ʴ�Ϊ���¶ȹ��ᵼ�������ľ���ֽ⣻
��1������������ԭ��Ӧ��ʵ�ʵ�ʧ�����غ㣬S2O32-��2SO42-��8e-��Cl2��2CI-��2e-������4molCl2����1molS2O32-���õ�8molCl-��2molSO42-�����ݵ���غ㣬��������Ӧ��10molH+������ԭ���غ㷴Ӧ����Ӧ��5molH2O�����ӷ���ʽΪS2O32-+4Cl2+5H2O=2SO42-+8Cl-+10H+���ʴ�Ϊ��S2O32-+4Cl2+5H2O=2SO42-+8Cl-+10H+��
��2�����ݱ궨��ԭ����֪�����ɵĵⵥ���������۱���������������Ƶζ�����ɫ��ȥ����Ӳ��仯��˵����Ӧ�ﵽ�յ㣬���Եζ��յ������Ϊ���������һ��Na2S2O3����Һ����ɫ��ʧ���Ұ��������ɫ���ı䣻
��Һ�з����ķ�ӦΪ��2Ba2++Cr2O72-=2BaCrO4+2H+�� Cr2O72-+6I-+14H+=2Cr3++3I2+7H2O�� I2+2S2O32-=2I-+S4O62-��
���ݷ���ʽ�г������ʵĹ�ϵΪBa2+��BaCrO4��
| 1 |
| 2 |
| 3 |
| 2 |
1mol 3mol
��n��Ba2+��=3n��S2O32-��=
| 1 |
| 3 |
����c��Ba2+���T
| 6��10 -6mol |
| 25��10 -3L |
�ʴ�Ϊ���������һ��Na2S2O3����Һ����ɫ��ʧ���Ұ��������ɫ���ı䣻 0.00024mol?L-1��
����������ͨ��Na2S2O3?5H2O���Ʊ�����������������ʵ�鷽����Ʒ�����Ϊ�߿��ĸ�Ƶ�⣬�Ѷ��еȣ���ȷ���������Ϣ��ȷ�Ʊ�ԭ��Ϊ��������Ĺؼ��������ֿ�����ѧ���ķ�����������������ѧʵ����������һ�������ϸߵ���Ŀ��

��ϰ��ϵ�д�
�����Ŀ
��ѧ��Ӧ�У���ʱ���ڡ�һ�����ʹ�������һ�������Բ�����ȫ��Ӧ������������з�Ӧ����������������ǣ�������
| A��������Ũ������MnO2��Ӧ |
| B��������Zn��18 mol/L�����ᷴӦ |
| C��������Fe��ϡ���ᷴӦ |
| D��������H2��N2�ڴ��������·�Ӧ |
�����£�������Һ��һ���ܴ���������������ǣ�������
| A��ʹpH��ֽ�ʺ�ɫ����Һ��Na+��[Al��OH��4]-��SO42-��Cl- |
| B����������Һ��Cr2O72-��SO42-��K+��NO3- |
| C��pH=0����Һ��Na+��C2O42-��MnO4-��SO42- |
| D��ˮ���������c��H+��=1��10-12mol?L-1����Һ��HCO3-��NH4+��C1-��Ca2+ |
һ�����Ļ���������ܱ������з�����Ӧ m A ��g��+n B ��g��?p C ��g���ﵽƽ����¶Ȳ��䣬�����������С��ԭ����
���ﵽƽ��ʱ��C��Ũ��Ϊԭ����2.5����������˵������ȷ���ǣ�������
| 1 |
| 2 |
| A��m+n��p |
| B��C������������� |
| C��ƽ��������Ӧ�����ƶ� |
| D��A ��ת���ʽ��� |
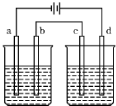 ��ͼ���ʵ��װ�ã����Ƕ��Ե缫������ֱͨ����һ��ʱ����������Һ��pH�������ҳ�����Һ��pH�½�������a��b���缫�ŵ����ӵ����ʵ�����ȣ���b��c���缫�ŵ����ӵ����ʵ���Ҳ��ȣ�����ҺӦѡ���±��еģ�������
��ͼ���ʵ��װ�ã����Ƕ��Ե缫������ֱͨ����һ��ʱ����������Һ��pH�������ҳ�����Һ��pH�½�������a��b���缫�ŵ����ӵ����ʵ�����ȣ���b��c���缫�ŵ����ӵ����ʵ���Ҳ��ȣ�����ҺӦѡ���±��еģ�������