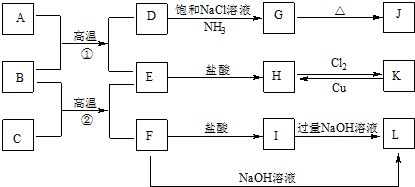
��Ŀ����
5�� 2015��8��12�������ش�ը�¹ʣ���һ�����������ǶԻ�������Ĺ�ע���ݲ�Σ���ֿ��д��д������ơ��ء����ף�P4��������狀��軯�ƣ�NaCN����
2015��8��12�������ش�ը�¹ʣ���һ�����������ǶԻ�������Ĺ�ע���ݲ�Σ���ֿ��д��д������ơ��ء����ף�P4��������狀��軯�ƣ�NaCN������1�������ж��ܺ�������Һ����������ԭ��Ӧ��3P4+10HClO3+18H2O=10HCl+12H3PO4���÷�Ӧ������������H3PO4������1mol P4�μӷ�Ӧת�Ƶ���Ϊ20mol��
��2���軯�ƣ�NaCN����һ�ֻ���ԭ�ϣ����ڻ�����ѧ�ϳɡ���ơ�ұ����л��ϳ�ҽҩ��ũҩ�����������ȷ��森
����֪��25��ʱ��HCN�ĵ���ƽ�ⳣ��Ka=6.2��10-10��H2CO3��25��ʱ�ĵ���ƽ�ⳣ����Ka1=4.5��10-7��
Ka2=4.7��10-11������ NaCN��Һͨ������CO2��Ӧ����ʽ�ǣ�CO2+NaCN+H2O=NaHCO3+HCN��
�ڳ����£��軯���������������Һ��Ӧ������̼�����ƺ���ʹʪ��ĺ�ɫʯ����ֽ����ɫ�����壬����䶾�ԣ��÷�Ӧ�Ļ�ѧ����ʽ�ǣ�NaCN+H2O2+H2O=NaHCO3+NH3����
�۵�ⷨ���Դ��������еĵ������������ͼʾ���أ���NOx�ڵ����зֽ������Ⱦ��N2��O2��ȥ�����缫���ǹ������������ʣ���һ�������¿����ɴ���O2-������������ӦΪ2NOx+4xe-=N2+2xO2-��
��3���ٴ�ͳ��ҵ�����ð������Ժϳ����أ���Ҫ��Ӧ���£�
2NH3��g��+CO2��g��=NH2CO2NH4��s����H=-159.47kJ•mol-1
NH2CO2NH4��s��=CO��NH2��2��s��+H2O��g����H=+72.49kJ•mol-1
��Ӧ2NH3��g��++CO2��g��=CO��NH2����s��+H2O��g���ġ�H=-86.98 kJ•mol-1
�ڹ�ҵ���ð����Ʊ����ᣬ��һ�����������ۼ��뵽100mLijŨ�ȵ�ϡ�����г�ַ�Ӧ��������ʣ��m1g���ۣ���ʱ���ռ���NO����448mL����״������Ȼ��������������м���ϡ���������ٲ���NOΪֹ������ʣ������m2g����m1-m2Ϊ5.04����������Ϊͭ������Ӧ������
���� ��1��Ԫ�ػ��ϼ����߷���������Ӧ�õ������������PԪ�ػ��ϼ۱仯����ת�Ƶ������ʵ�����
��2�����ɵ���ƽ�ⳣ����֪�����ԣ�H2CO3��HCN��HCO3-����NaCN��Һͨ������CO2����NaHCO3��HCN��
�ڳ����£��軯���������������Һ��Ӧ������̼�����ƺ���ʹʪ��ĺ�ɫʯ����ֽ����ɫ�����壬������Ϊ��������ƽ��д����ʽ��
�۵�������������ԭ��Ӧ��NOx��������õ�������N2��O2-��
��3������֪����.2NH3��g��+CO2��g���TNH2CO2NH4��s����H=-159.47kJ•mol-1
��NH2CO2NH4��s���TCO��NH2��2��s��+H2O��g����H=+72.49kJ•mol-1
���ݸ�˹���ɣ���+��ɵã�2NH3��g��+CO2��g���TCO��NH2����s��+H2O��g����
�����ۼ��뵽100mLijŨ�ȵ�ϡ�����У���ַ�Ӧ��������ʣ��m1g���ۣ�����Fe��NO3��2��NO����0.02mol�����ݵ���ת���غ����μӷ�Ӧ��Fe�����ʵ������ٸ���Feԭ���غ������Һ��n[Fe��NO3��2]��������Һ������ϡ���������ٷ�ӦΪֹ������ʣ��Fe��m2g��Fe��NO3��2��NO3-������Ӧ3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O����m1-m2��Ϊ����Һ��NO3-��Ӧ��Fe��������
��� �⣺��1����Ӧ��PԪ�ػ��ϼ����ߵõ�H3PO4����������ΪH3PO4������1mol P4�μӷ�Ӧת�Ƶ���Ϊ1mol��4��5=20mol���ʴ�Ϊ��H3PO4��20��
��2�����ɵ���ƽ�ⳣ����֪�����ԣ�H2CO3��HCN��HCO3-����NaCN��Һͨ������CO2����NaHCO3��HCN����Ӧ����ʽΪ��CO2+NaCN+H2O=NaHCO3+HCN��
�ʴ�Ϊ��CO2+NaCN+H2O=NaHCO3+HCN��
�ڳ����£��軯���������������Һ��Ӧ������̼�����ƺ���ʹʪ��ĺ�ɫʯ����ֽ����ɫ�����壬������Ϊ��������Ӧ����ʽΪ��NaCN+H2O2+H2O=NaHCO3+NH3����
�ʴ�Ϊ��NaCN+H2O2+H2O=NaHCO3+NH3����
�۵�������������ԭ��Ӧ��NOx��������õ�������N2��O2-�������缫��ӦʽΪ��2NOx+4xe-=N2+2xO2-��
�ʴ�Ϊ��2NOx+4xe-=N2+2xO2-��
��3������֪����.2NH3��g��+CO2��g���TNH2CO2NH4��s����H=-159.47kJ•mol-1
��NH2CO2NH4��s���TCO��NH2��2��s��+H2O��g����H=+72.49kJ•mol-1
���ݸ�˹���ɣ���+��ɵã�2NH3��g��+CO2��g���TCO��NH2����s��+H2O��g������H=-159.47kJ•mol-1+72.49kJ•mol-1=-86.98kJ•mol-1��
�ʴ�Ϊ��-86.98��
�����ۼ��뵽100mLijŨ�ȵ�ϡ�����У���ַ�Ӧ��������ʣ��m1g���ۣ�����Fe��NO3��2��NO���壬NO�����ʵ���Ϊ$\frac{0.448L}{22.4L/mol}$=0.02mol�����ݵ���ת���غ��֪���μӷ�Ӧ��Fe�����ʵ���Ϊ$\frac{0.02mol����5-2��}{2}$=0.03mol������Feԭ���غ��֪����Һ��n[Fe��NO3��2]=0.03mol����Һ��n��NO3-��=0.06mol��
������Һ������ϡ���������ٷ�ӦΪֹ��Fe��NO3��2��NO3-������Ӧ3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O������ʣ��ͭ��m2g����m1-m2��Ϊ����Һ��NO3-��Ӧ��ͭ�����������ٴη�Ӧ��ͭ�����ʵ���Ϊxmol����
3Fe+8H++2NO3-�T3Fe2++2NO��+4H2O
3 2
xmol 0.06mol
����x=0.09mol
�����ٴη�Ӧ��Fe������Ϊ0.09mol��56g/mol=5.04g������m1-m2��=5.04g��
�ʴ�Ϊ��5.04��
���� ���⿼��������ԭ��Ӧ������������㡢��ѧ����ʽ��д�����ԭ������Ӧ�ȼ���ȣ���3���йؼ����жϣ�m1-m2��Ϊ����Һ��NO3-��Ӧ��Fe��������ע����ݵ���ת���غ㡢Ԫ���غ���㣬����ݷ���ʽ���㣬��ԱȽ��鷳��ע���غ�˼����������ԭ��Ӧ������Ӧ�ã������������ȫ��Ӧ���������ӷ���ʽ���㣮

| ���� | Fe3+ | Cu2+ | SO42- | Na+ | H+ |
| Ũ��/��mol/L�� | 2��10-3 | 1��10-3 | 1��10-2 | 2��10-3 |
��2��Ϊ�˼�����Ⱦ�����Ϊ��������ʦ��������������̣�����ͭ���̷���FeSO4•7H2O����
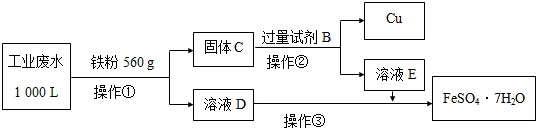
�ٹ�ҵ��ˮ�м������ۺ�Ӧ�����ӷ���ʽΪ��Fe+Cu2+�TFe2++Cu��Fe+2Fe3+�T3Fe2+��Fe+2H+�TFe2++H2����
���Լ�B��H2SO4��
�۲�����������Ũ������ȴ�ᾧ�����ˣ������������ˮϴ�Ӿ��壬�ڵ����¸��
��ͨ��������ҺD��Fe3+���жϷ�ˮ��Fe3+�Ƿ������ʵ�鷽����ȡ����D����Һ���Թ��У��μ�1��KSCN��Һ�������ֺ�ɫ��˵����ˮ��Fe3+������
�ݻ�õ�FeSO4•7H2O���ܱձ��棬ԭ����FeSO4�л�ԭ�ԣ��ױ���������FeSO4•7H2Oʧȥ�ᾧˮ����
������1000L��ˮ���������ɻ�� FeSO4•7H2O�����ʵ�����12mol��
| A�� | 22.2% | B�� | 30.0% | C�� | 75.7% | D�� | 80.6% |
| A�� | 18g������ຬ���������ĿΪ4NA | |
| B�� | 0.1L3mol•L-1��NH4NO3��Һ�к��е�NH4+��ĿΪ0.3NA | |
| C�� | 7.8gNa2O2����������������Ϊ0.4NA | |
| D�� | 4.5gSiO2�����к��еĹ�������ĿΪ0.3NA |
| ѡ�� | ���� | ���� | ���� |
| A | KI������Һ�е�����ˮ����ͨ��SO2 | �ȱ���ɫ����ɫ | SO2��Ư���� |
| B | �ýྻ��˿պȡ��ɫ��Һ������ɫ��Ӧ | ����ʻ�ɫ | ��ɫ��ҺΪ������Һ |
| C | ���ɫZnS�����ϵμ�0.1mol/LCuSO4��Һ | ������Ϊ��ɫ | Ksp��ZnS����Ksp��CuS�� |
| D | �����½�AlƬ����Ũ������ | �����Ա仯 | Al��Ũ�����Ӧ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | �������������θ������ҩ�� | |
| B�� | �������������ڳ��������桢����Ũ���� | |
| C�� | �������������������ά��ʯӢ���� | |
| D�� | �������ƿ�����Ư���ͺ�������еĹ����� |
| A�� | Na3PO4�T3Na++P5++4O2- | B�� | H2SO4�TH++SO42- | ||
| C�� | NH3•H2O?NH4++O2-+H+ | D�� | NH4NO3�TNH4++NO3- |
| A�� | Na | B�� | Mg | C�� | Al | D�� | һ���� |