��Ŀ����
��25�桢101 kPa�£�1 g�״�ȼ������CO2��Һ̬ˮʱ����22.68 kJ,�����Ȼ�ѧ����ʽ��ȷ���ǣ� ��
A��CH3OH��l��+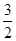 O2��g��=CO2��g��+2H2O��l������H=+725.8 kJ/mol
O2��g��=CO2��g��+2H2O��l������H=+725.8 kJ/mol
B��2CH3OH��l��+3O2��g��=2CO2��g��+4H2O��l������H=��1452 kJ/mol
C��2CH3OH��l��+3O2��g��=2CO2��g��+4H2O��l������H=��725.8 kJ/mol
D��2CH3OH��l��+3O2��g��=2CO2��g��+4H2O��l������H=+1452 kJ/mol
16��B
��������
����������״�ȼ���Ƿ��ȷ�Ӧ����H��0��A��D����ȷ�����������֪��1mol�״���ȫȼ�շų���������22.68kJ��32��725.8kJ������ѡ��C����ȷ����ѡB��
���㣺�����Ȼ�ѧ����ʽ���ж�

��10�֣�����̼ѭ�����Ѿ������˹�������ӣ��Իش��������⣺
��1��ú��������Һ���������ȼ�ϵ������ʡ�
��֪25�棬101 ʱ��
ʱ��

����25�棬101 ʱ��
ʱ�� .
.
��2����¯������CO�������Ҫ��;֮һ���������ӦΪ��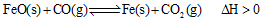 ����֪��1100��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��K=0.263��
����֪��1100��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��K=0.263��
���¶����ߣ���ѧƽ���ƶ���ﵽ�µ� ƽ�⣬��ʱƽ�ⳣ��Kֵ �����������С�����䡱����
ƽ�⣬��ʱƽ�ⳣ��Kֵ �����������С�����䡱����
��1100��ʱ��ø�¯ʱ�� ������������£��÷�Ӧ�Ƿ��ڻ�ѧƽ��״̬�� (��ǡ���)�����ж������� ��
������������£��÷�Ӧ�Ƿ��ڻ�ѧƽ��״̬�� (��ǡ���)�����ж������� ��
��Ŀǰ��ҵ�Ͽ��� ������ȼ�ϼ״�
������ȼ�ϼ״� ���йط�ӦΪ��
���йط�ӦΪ�� ���������Ϊ1L���ܱ������У�����1mol
���������Ϊ1L���ܱ������У�����1mol ��3mol
��3mol ����Ӧ�����в��
����Ӧ�����в�� ��
�� (g)��Ũ����ʱ��ı仯��ͼ��ʾ��
(g)��Ũ����ʱ��ı仯��ͼ��ʾ��
�ٴӷ�Ӧ��ʼ��ƽ�⣬������ƽ����Ӧ���� ��
��
�����д�ʩ��ʹ �������
�������  ������ţ���
������ţ���
| A�������¶� |
B���ٳ��� |
C���ٳ��� |
D���� ��g������ϵ�з��� ��g������ϵ�з��� |
 ��
�� Fe(s)��CO2(g) ��H>0����֪��1100��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��K=0.263��
Fe(s)��CO2(g) ��H>0����֪��1100��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��K=0.263��
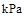 ʱ��
ʱ��

 .
. ����֪��1100��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��K=0.263��
����֪��1100��ʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��K=0.263�� ������������£��÷�Ӧ�Ƿ��ڻ�ѧƽ��״̬�� (��ǡ���)�����ж�������
��
������������£��÷�Ӧ�Ƿ��ڻ�ѧƽ��״̬�� (��ǡ���)�����ж�������
�� ������ȼ�ϼ״����йط�ӦΪ��
������ȼ�ϼ״����йط�ӦΪ�� ���������Ϊ1L���ܱ������У�����1mol
���������Ϊ1L���ܱ������У�����1mol ����Ӧ�����в��
����Ӧ�����в�� (g)��Ũ����ʱ��ı仯��ͼ��ʾ��
(g)��Ũ����ʱ��ı仯��ͼ��ʾ��
 ��
�� ������� ������ţ���
������� ������ţ��� ��g������ϵ�з���
E.����He(g),ʹ��ϵѹǿ����
��g������ϵ�з���
E.����He(g),ʹ��ϵѹǿ����