��Ŀ����
��1��ij���廯��A��̼���⡢������Ԫ����ɣ��������ܶ�����ͬ������H2�ܶȵ�69������֪0.1 molA����������ȫȼ�յ�30.8gCO2��0.3mol H2O���� ��A�ķ���ʽ��____________��
A������NaHCO3��Һ��Ӧ���䱽������������λ��ȡ��������A�к��������ŵ�����������������
��֪��![]()
��2��A������![]() ��Ӧ�Ƶ��л���B������ʽΪC9H9O4����FeCl3��Һ������ɫ�����÷�Ӧ�Ļ�ѧ����ʽ��____________________________________
��Ӧ�Ƶ��л���B������ʽΪC9H9O4����FeCl3��Һ������ɫ�����÷�Ӧ�Ļ�ѧ����ʽ��____________________________________
��3����֪����������Ϣ������ʽΪC3H4�������Է�����ͼת����
![]()
![]()
����E��F�������ԡ�F�������л���B���Ҷ�����ͬ������Ӧ�õ��л���G����Gͨ���Ӿ۷�Ӧ���Խ�һ���ϳɷ���ʽΪ��C15H?15O6��2��ҩ��H��
��д��F��ͬ���칹��ṹ��ʽ��������a��������״���ࡢb��������ÿ��̼ԭ���϶�������ԭ��___________________________________________
��C��D�Ļ�ѧ����ʽ��_________________________________
��H�Ľṹ��ʽ��_______________________________
��1��C3H6O3 ������3�֣��� �Ȼ����ǻ�������2�֣� ��2��������2�֣�
![]()
��3����HCOOCH=CHCH3���� HCOOCH2CH=CH2������4�֣�
��(CH3)2CBr��CH2Br+2NaOH������ (CH3)2C(OH)��CH2��OH+2NaBr������2�֣�
��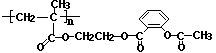

 ����ȫ���ִʾ��ƪ��ϵ�д�
����ȫ���ִʾ��ƪ��ϵ�д� �����߿����ϵ�д�
�����߿����ϵ�д�