��Ŀ����
12����֪�������Ĵ������£����Ժ�±�ط�Ӧ���ɦ�-±���RCH2COOH+Cl2��RCH��Cl��COOH+HCl�����ʾ���ǿ���Ϊ��ʵ��������л���A��һ��������ת��ΪH�Ĺ�ϵ��

�ش��������⣺
��1��д���л���Ľṹ��ʽ��ACH2=CH2ECH3COOHGCH2��OH��COONaHCH2��OH��COOH
��2��д�����л�ѧ����ʽ��A��BCH2=CH2+HCl$��_{��}^{����}$CH3CH2Cl�� C��D2CH3CH2OH+O2$��_{��}^{����}$2CH3CHO+2H2O��
���� �л���A��Ϊ��ʵ���������AΪCH2=CH2��A���Ȼ���ӳɵ�BΪCH3CH2Cl��B��������ˮ���CΪCH3CH2OH��C������DΪCH3CHO��D������EΪCH3COOH��E���巢��ȡ����FΪBrCH2COOH��F��������ˮ���GΪCH2��OH��COONa��G�ữ��HΪCH2��OH��COOH��H��Ũ����������·�����������I���ݴ˴��⣮
��� �⣺�л���A��Ϊ��ʵ���������AΪCH2=CH2��A���Ȼ���ӳɵ�BΪCH3CH2Cl��B��������ˮ���CΪCH3CH2OH��C������DΪCH3CHO��D������EΪCH3COOH��E���巢��ȡ����FΪBrCH2COOH��F��������ˮ���GΪCH2��OH��COONa��G�ữ��HΪCH2��OH��COOH��H��Ũ����������·�����������I��
��1����������ķ�����֪��AΪCH2=CH2��EΪCH3COOH��GΪCH2��OH��COONa��HΪCH2��OH��COOH��
�ʴ�Ϊ��CH2=CH2��CH3COOH��CH2��OH��COONa��CH2��OH��COOH��
��2����ӦA��B�Ļ�ѧ����ʽΪCH2=CH2+HCl $��_{��}^{����}$ CH3CH2Cl����ӦC��D�Ļ�ѧ����ʽΪ2CH3CH2OH+O2 $��_{��}^{����}$ 2CH3CHO+2H2O��
�ʴ� Ϊ��CH2=CH2+HCl $��_{��}^{����}$ CH3CH2Cl��2CH3CH2OH+O2 $��_{��}^{����}$ 2CH3CHO+2H2O��
���� ���⿼���л��ƶϣ���Ŀ�Ѷ��еȣ���Ҫѧ���Ը������Ϣ�������ã�ȷ��AΪ��ϩ�ǹؼ����Ƕ��л�������֪ʶ���ۺϿ��飬�ܽϺõĿ��鿼������ѧ�����ͷ��������������Ǹ߿��ȵ����ͣ�

 ���Ͱ�ͨ��ĩ���ϵ�д�
���Ͱ�ͨ��ĩ���ϵ�д�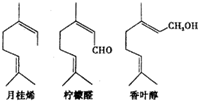
| A�� | �¹�ϩ����������ԭ�ӿ��ܹ�ƽ�� | |
| B�� | ����ȩ����Ҷ����ͬ���칹�� | |
| C�� | ��Ҷ���ܷ����ӳɷ�Ӧ��������Ӧ��������Ӧ | |
| D�� | �����л����һ�ȴ������7�֣������������칹�� |
| A�� | 2�� | B�� | 3�� | C�� | 4�� | D�� | 5�� |
| A�� | ��λʱ��������amol H2��ͬʱ���� 2amol HI | |
| B�� | �����ڵ���ѹǿ����ʱ��仯 | |
| C�� | ������H2��I2���ټ��٣�HI�������� | |
| D�� | ��λʱ����ÿ����amol HI��ͬʱa NA��HI���ӷֽ� |

| A�� | X���⻯���Z���⻯���ۡ��е�� | |
| B�� | Y��ԭ�Ӱ뾶��X��ԭ�Ӱ뾶С | |
| C�� | ����Ԫ�ص��⻯���У�X���⻯���������ѷֽ� | |
| D�� | R�������ڵ�Ԫ������Ϊ32�� |
���ƼغϽ�ͨ��״���³�Һ̬������ԭ�ӷ�Ӧ�ѵĵ��ȼ�
���ƵĻ�ѧ���ʻ��ã��������ƿɱ������л��ܼ�CCl4��
�����ڿ����л�����������ȼ����������ȼ�ո�Ϊ����
��Na2CO3��Һ�ܸ�����Һ��Ӧ�������ܸ�����Һ��Ӧ��
| A�� | �٢ڢ� | B�� | �ڢۢ� | C�� | �٢� | D�� | �� |
| A�� | ��ˮ | B�� | NH4Cl | C�� | NaCl | D�� | HCl |
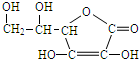
 ��
��