��Ŀ����
14�������Ȼ�ѧ����ʽS��l��+O2��g���TSO2��g����H=-293.23kJ•mol-1����������˵������ȷ���ǣ�������| A�� | S��s��+O2��g���TSO2��g������Ӧ�ų�����������293.23 kJ•mol-1 | |
| B�� | S��g��+O2��g���TSO2��g������Ӧ�ų�������С��293.23 kJ•mol-1 | |
| C�� | 1 mol SO2�ļ����ܺʹ���1 mol���1 mol�����ļ���֮�� | |
| D�� | 1 mol SO2�ļ����ܺ�С��1 mol���1 mol�����ļ���֮�� |
���� ����������Һ̬ת��ɹ�̬Ҫ�ų���������Һ̬ת�����̬Ҫ���������Լ����ȷ�Ӧ�оɼ��������յ�����С���¼��γ����ų����������ַ�����
��� �⣺A����������Һ̬ת��ɹ�̬Ҫ�ų�����������1mol����S����ȼ�շų�������С��297.23kJ•mol-1����A����
B����������Һ̬ת�����̬Ҫ��������������1mol����S����ȼ�շų�����������297.23kJ•mol-1����B����
C������ȷ�Ӧ�оɼ��������յ�����С���¼��γ����ų���������1molSO2�ļ����ܺʹ���1molS��1molO2�ļ����ܺͣ���C��ȷ��
D������ȷ�Ӧ�оɼ��������յ�����С���¼��γ����ų���������1molSO2�ļ����ܺʹ���1molS��1molO2�ļ����ܺͣ���D����
��ѡC��
���� ������Ҫ�����˻�ѧ��Ӧ�з�Ӧ�ȵļ��㣬��Ҫע�����������̬֮���ת��Ҳ���������ı仯��

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
9����25�棬101kPa�£�1molH2���ȼ������Һ̬ˮ����286kJ������˵���в���ȷ���ǣ�������
| A�� | ��ͬ�����£�2mol H2��ȫȼ��ʱҺ̬ˮ����572kJ | |
| B�� | ��ͬ�����£�2mol H2O��ȫ�ֽ���������572kJ | |
| C�� | ��ͬ�����£�1mol H2���ȼ��������̬ˮ���ȴ���286kJ | |
| D�� | �γ�1mol H2O�Ļ�ѧ�����ͷŵ��������ڶ���lmol H2��0.5mol O2�Ļ�ѧ�������յ������� |
4���������ӷ���ʽ��д��ȷ���ǣ�������
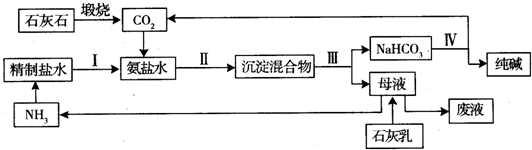
| A�� | ����������Һ��ͨ�����������̼��OH-+CO2�THCO3- | |
| B�� | ̼�������ϡ���CaCO3+2H+�TCa2++H2O+CO2�� | |
| C�� | ��̼�����ϡ��Һ�м�������ռ���Һ��NH4++OH-�TNH3H2O | |
| D�� | ����ˮ�ķ�Ӧ��Na+H2O�TNa++OH-+H2�� |
 ��
��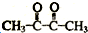 ��A��B�ķ�Ӧ����Ϊ������Ӧ��
��A��B�ķ�Ӧ����Ϊ������Ӧ��