��Ŀ����
6��ij����Na2O���ʵ�Na2O2�����������ʵ��ⶨ����Ʒ�Ĵ��ȣ��ɹ�ѡ���װ�ã�

�ɹ�ѡ���ҩƷ��CaCO3���壬6mol/L���ᣬ����ˮ
��ش��������⣺
��1��������װ�ÿ�����װһ����IJⶨ����������Ʒ���ȵ�ʵ��װ�ã����ѡ������������A������ĸ����
A���٢ܢ�B���٢ڢܢ�C���ݢޢ�D���٢ۢݢ�
��2��ʵ��ѡ���ҩƷ������ˮ����ѡ��װ�õ�����˳��Ӧ�ǣ�����ӿڵ���ĸ�����ӽ���ʡ�ԣ���G���ӣ�A����B���ӣ�F��������ĸ��
��3��д��ʵ����Na2O2��Na2O�ֱ�����Ӧ�����ӷ���ʽ��2Na2O2+2H2O�T4Na++4OH-+O2����2Na2O+2H2O�T4Na++4OH-
��4��������ʵ���������Һ���Ƴ�Ũ��Ϊ1.0mol/L����Һ��
����400ml����Һ��ͨ��0.3molCO2����������Һ��HCO3-��CO32-�����ʵ���Ũ��֮��ԼΪ
A.1��3B.1��2��������������C.2��1��������������D.3��1
�ڽ�������Һ�����ᾧ�õ�Na2CO3��NaHCO3��������ܲⶨ�������Na2CO3������������ABD��
A��ȡa�˻�����ּ��ȣ��ڸ���������ȴ�����£�����b��
B��ȡa�˻����������ϡ�����ַ�Ӧ�����ȡ����ɡ����գ���b�˹���
C��ȡa�˻����������ϡ�����ַ�Ӧ���ݳ������ü�ʯ�����գ�����b��
D��ȡa�˻����������Ba��OH��2��Һ��ַ�Ӧ�����ˡ�ϴ�ӡ���ɣ���b�˹��壮
��5�����²�ú�����õ��Ծ��������ƺ�����ߣ���ʢ�ŵ�Ҳ�ǹ������ƣ�д�������Ծ����Ծȹ��ܵĻ�ѧ��Ӧ����ʽ��2Na2O2+2CO2�T2Na2CO3+O2��
���� ��1�����ù���������ˮ��Ӧ����������Ȼ����ˮ�������ⶨ�������������������Na2O2�����Ĵ��ȣ�
��2���ɣ�1��������֪����Ҫ��ҩƷΪ����ˮ��
װ�õ���װ˳����������ˮ��Ӧ������ˮ�������ⶨ���������������ʢˮ���Լ�ƿ����һ��Ҫ�̽�����������ѹǿԭ����ˮ�ų�����Ͳ��ˮ������������������������
��3�������������Ƿ�Ӧ����������������������������ˮ��Ӧ�����������ƣ�
��4������̼�����ơ�̼���Ƶ����ʵ����ֱ�Ϊxmol��ymol�������������غ㡢̼Ԫ���غ�ⷽ�̼���x��y��ֵ��
��A������̼�����Ʒֽ�¹����������ᣬ���ݲ��������Լ���̼�����Ƶ��������ⶨ�������Na2CO3����������
B�����չ���ΪNaCl��������̼�����ơ�̼���Ƶ����ʵ����ֱ�Ϊxmol��ymol�����ݶ�������֮�ͼ��������غ��з��̿��Լ���x��y��ֵ��
C����������Ϊ���ɵĶ�����̼��������Һ�д�����ˮ����������ȷ��������̼��������
D�����չ���Ϊ̼�ᱵ��������̼�����ơ�̼���Ƶ����ʵ����ֱ�Ϊxmol��ymol�����ݶ�������֮�ͼ�̼�ᱵ�������з��̿��Լ���x��y��ֵ��
��5�����������������̼��Ӧ����̼������������
��� �⣺��1�������ù���������ˮ��Ӧ����������Ȼ����ˮ�������ⶨ�������������������Na2O2�����Ĵ��ȣ���ѡ��ݢ٢ܣ�
�ʴ�Ϊ��A��
��2���ɣ�1��������֪����Ҫ��ҩƷΪ����ˮ��
װ�õ���װ˳����������ˮ��Ӧ���������ɵ������е�ˮ����������ˮ�������ⶨ���������������ʢˮ���Լ�ƿ����һ��Ҫ�̽�����������ѹǿԭ����ˮ�ų�����Ͳ��ˮ������������������������������˳��Ϊ����G���ӣ�A����B���ӣ�F����
�ʴ�Ϊ������ˮ����G���ӣ�A����B���ӣ�F����
��3��Na2O2���Ƿ�Ӧ�����ӷ���ʽΪ2Na2O2+2H2O�T4Na++4OH-+O2����Na2O��ˮ��Ӧ�����ӷ���ʽΪ2Na2O+2H2O�T4Na++4OH-��
�ʴ�Ϊ��2Na2O2+2H2O�T4Na++4OH-+O2����2Na2O+2H2O�T4Na++4OH-��
��4������̼�����ơ�̼���Ƶ����ʵ����ֱ�Ϊxmol��ymol�������������غ㡢̼Ԫ���غ㣬��$\left\{\begin{array}{l}{x+2y=0.4��1}\\{x+y=0.3}\end{array}\right.$
���x=0.2��y=0.1��
��������Һ��HCO3-��CO32-�����ʵ���Ũ��֮��Լ0.2mol��0.1mol=2��1��
�ʴ�Ϊ��C��
��A��ȡa�˻�����ּ��ȣ��ڸ���������ȴ�����£�����b�ˣ�����̼�����Ʒֽ�¹����������ᣬ���ݲ��������Լ���̼�����Ƶ����������Բⶨ�������Na2CO3������������A��ȷ��
B��ȡa�˻����������ϡ�����ַ�Ӧ�����ȡ����ɡ����գ���b�˹��壬���չ���ΪNaCl��������̼�����ơ�̼���Ƶ����ʵ����ֱ�Ϊxmol��ymol�����ݶ�������֮�ͼ��������غ��з��̿��Լ���x��y��ֵ����������m=nM����̼���Ƶ����������Բⶨ�������Na2CO3������������B��ȷ��
C��ȡa�˻����������ϡ�����ַ�Ӧ���ݳ������ü�ʯ�����գ�����b�ˣ���������Ϊ���ɵĶ�����̼��������Һ�д�����ˮ����������ȷ��������̼�����������ܲⶨ�������Na2CO3������������C����
D��ȡa�˻����������Ba��OH��2��Һ��ַ�Ӧ�����ˡ�ϴ�ӡ���ɣ���b�˹��壮���չ���Ϊ̼�ᱵ��������̼�����ơ�̼���Ƶ����ʵ����ֱ�Ϊxmol��ymol�����ݶ�������֮�ͼ�̼�ᱵ�������з��̿��Լ���x��y��ֵ����������m=nM����̼���Ƶ����������Բⶨ�������Na2CO3������������D��ȷ��
�ʴ�Ϊ��ABD��
��5�����������������̼��Ӧ����̼��������������Ӧ����ʽΪ��2Na2O2+2CO2�T2 Na2CO3+O2��
�ʴ�Ϊ��2Na2O2+2CO2�T2 Na2CO3+O2��
���� ���⿼��ѧ����ʵ��ԭ�������⡢ʵ�鷽�������ۡ���ѧ����ȣ��ۺ��Խϴ��Ѷ��еȣ����ʵ��ԭ���ǽ���Ĺؼ�����Ҫѧ���߱���ʵ�Ļ���֪ʶ���ۺ�����֪ʶ�������⡢��������������

 ��1����Ԫ�¿�������ĩϵ�д�
��1����Ԫ�¿�������ĩϵ�д�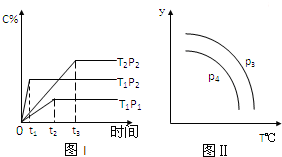
| A�� | ��P3��P4����y���ʾB�İٷֺ��� | |
| B�� | ��P3��P4����y���ʾ��������ƽ��Ħ������ | |
| C�� | ��P3��P4����y���ʾB��������� | |
| D�� | ��P3��P4����y���ʾ���������ܶ� |
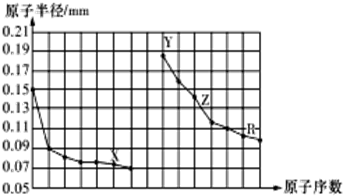
| A�� | ͬ����Ԫ���У�X�⻯��ķе���� | |
| B�� | �����Ӱ뾶��X��Y��Z | |
| C�� | Y��Z��R��Ӧ������������ˮ�����֮����Է�����Ӧ | |
| D�� | ������ڵ�X��Z���ɵĻ�������Եõ�����Z |
| A�� |  ��ȡ���ռ����﴿����NH3 | |
| B�� |  ��װ�ö��������������ͱ���̼������Һ | |
| C�� |  �����Ʊ����ռ�����NO2 | |
| D�� |  �Ƚ�MnO2��Cl2��S�������� |
| A�� | ֻ����ˮ�Ϲ��� | B�� | ֻ������ѧ���� | ||
| C�� | ˮ�Ϲ��̷��ȴ�����ɢ�������� | D�� | ��ɢ�������ȴ���ˮ�Ϲ��̷��� |
| A�� | ����ĵ���ʽ�� | B�� | ��ϩ�ļ���ʽ�� | ||
| C�� | ��ϩ�Ľṹ��ʽ��CH2CH2 | D�� | �Ҵ��Ľṹʽ�� |
| A�� | �������1�����ӵ�ԭ�Ӷ��Ǽ���� | |
| B�� | �������5�����ӵ�ԭ�Ӷ��Ƿǽ���ԭ�� | |
| C�� | ϡ������ԭ������㶼�ﵽ8�����ȶ��ṹ | |
| D�� | ����Ԫ��ԭ�ӵ�����㶼������2�����ӣ����ǽ���Ԫ�� |