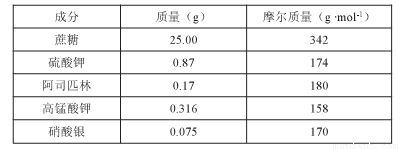
��Ŀ����
��1��ij�¶ȣ�t �棩ʱ��ˮ��KW��1��10��13������¶ȣ�����ڡ�����С�ڡ����ڡ���__ __________25 �棬��������____________�������¶���pH��11��NaOH��Һa L��pH��1��H2SO4����Һb L��ϣ������û��ҺΪ���ԣ���a��b��____________��
��2����֪25 ��ʱ0.1 mol/L������Һ�ﵽƽ�⣨HAc H����Ac����ʱ��pHԼΪ3������ڴ���Һ�м���������pHΪ5�����ᣬ�����ƽ��____________�ƶ�������������ҡ�������������ԭ������Һ�м������������ƾ��壬�������ܽ����Һ��pHֵ____________�����������С�����䡱����
H����Ac����ʱ��pHԼΪ3������ڴ���Һ�м���������pHΪ5�����ᣬ�����ƽ��____________�ƶ�������������ҡ�������������ԭ������Һ�м������������ƾ��壬�������ܽ����Һ��pHֵ____________�����������С�����䡱����
���и������ܴ������棬��������Ӧ�Լ���ᷢ����ѧ�仯���ҷ�����Ӧ�����ӷ���ʽ��д��ȷ���� �� ��
ѡ�� | | �����Լ� | ���ӷ���ʽ |
A | NH | ����Ba��OH��2��Һ | 2NH = BaSO4����NH3��H2O |
B | Mg2����HCO | ����NaOH��Һ | Mg2����2HCO = MgCO |
C | H����Na����NO | Fe�� | Fe��2H��= Fe2����H2�� |
D | Ca2����NH3��H2O��Cl�� | ͨ�����CO2 | NH3��H2O��CO2 = NH |
��Դ��ȱ������������ٵ��ش����⣬���û�ѧ��Ӧ��ʵ�ֶ�����ʽ�������ת������ش��������⣺
(1������̬��̬ԭ���γ�1mol��ѧ���ͷŵ���������м��ܡ��ӻ�ѧ���ĽǶȷ�������ѧ��Ӧ�Ĺ��̾��Ǿɼ����Ѻ��¼����γɹ��̡���֪��ӦN2(g)��3H2(g) 2NH3(g) ��H����93 kJ��mol��1��
2NH3(g) ��H����93 kJ��mol��1��
�Ը��ݱ������м������ݣ�����a ����ֵΪ kJ��mol��
��ѧ�� | H��H | N��H | N��N |
����/kJ��mol��1 | 436 | a | 945 |
��2���״���һ�ֿ�������Դ�����й㷺�Ŀ�����Ӧ��ǰ������֪�ڳ�ѹ�������±仯��
�� 2CH3OH(l)��3O2(g)��2CO2(g)��4H2O(g) ��H ��a kJ��mol
�� H2O(g)��H2O(l) ��H ��b kJ��mol
д��Һ̬�״���ȫȼ�����ɶ�����̼��Һ̬ˮ���Ȼ�ѧ����ʽ�� ��
(3����֪��HCN(aq����NaOH��aq����Ӧ���ʱ��H��-12.1kJ��mol��1; HCl(aq)��NaOH(aq)��Ӧ���ʱ䦤H����55.6 kJ��mol��1����HCN��ˮ��Һ�е�����ʱ䦤H���� ��
(4)��֪��H2(g)+1/2O2(g)=H2O(1) ��H��-285.83kJ��mol��1
CO(g)+1/2O2(g)=CO2(g) ��H��-282.9kJ��mol��1;
��������һ����̼�Ļ��������ȫȼ�տ�����5.4g H2O(l)�����ų�114.3kJ����������������CO�����ʵ���Ϊ (������������ΪС��)


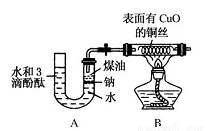
 �������У���Ͳ����������ҩ�ס��ձ���������ƽ�� �� �����ں�������д��ȱ���������ƣ���
�������У���Ͳ����������ҩ�ס��ձ���������ƽ�� �� �����ں�������д��ȱ���������ƣ��� 2
2 SO3��g�� ��H����197 kJ��mol��1����ͬ�¡�ͬ����������ܱ������зֱ�������壺���ף�2 mol SO2��1 mol O2�����ң�1 mol SO2��0.5 mol O2�������� 2 mol SO3�����¡������·�Ӧ��ƽ��ʱ�����й�ϵһ����ȷ����
SO3��g�� ��H����197 kJ��mol��1����ͬ�¡�ͬ����������ܱ������зֱ�������壺���ף�2 mol SO2��1 mol O2�����ң�1 mol SO2��0.5 mol O2�������� 2 mol SO3�����¡������·�Ӧ��ƽ��ʱ�����й�ϵһ����ȷ����
 ����
���� ��Mg 2����SO
��Mg 2����SO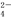
 ��Cl��
��Cl��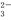 ��2H2O
��2H2O