��Ŀ����
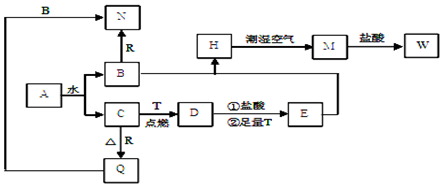
13��ijʵ��С���������Ư���Ե�ʵ��̽����Ӧ��ͼ1��ʾ�ķ���ʵ�飬��������A����Ҫ�ɷ��������������ǿ�����ˮ�������ش��������⣺
��1�������о���ʵ�飩����ҪĿ����̽��Cl2�ڳ�ʪ����ʱ�Ƿ����Ư���ԣ�
��2��Ũ�������������������A�е�ˮ���������о�Ŀ��ֱ����ص�ʵ��������B�ڵ���ɫ��������ɫ��C�ڵ���ɫ������ɫ��
��3����ʯ����Һ�ε�������ˮ�е�����ʯ����Һ�ȱ�����ɫ��
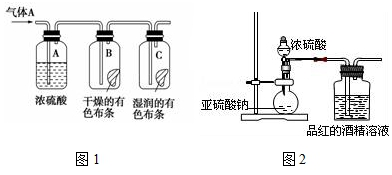
��Ϊ̽��SO2��Ư����SO2ֱ����������ɫ���ʣ�����SO2��ˮ��Ӧ�IJ���H2SO3�����ã������ͼ2װ�ý���ʵ�飺��ͼ2�������������������Ϊ70%��
��1��Ϊ̽�������SO2 �ܲ���ʹƷ����ɫ����ָ����Ƶ�ʵ��װ��ͼ���������ԵIJ����е�һ���ٶ�������û�и����ȱ�ٶ�������ĸ���װ�ã�����û��β������װ��
��2�������ĺ��װ�ã�ʵ���п���SO2�Դ�Լÿ��3�����ݵ��ٶ�ͨ��Ʒ��ľƾ���Һʱ������һСʱ��Ʒ���Բ���ɫ����˵��Ʒ����ɫ��ԭ����SO2ֱ�ӵ��£����ԣ�SO2 ͨ��Ʒ��ˮ��Һ������Ʒ����ɫ�Ŀ�����H2SO3�����ѧʽ��
���� ��1���������Ա�ʵ��������ɫ������ʪ�����ɫ������������
��2��Ũ���������ˮ�Կ�����������ݸ��������������ʹ�������ɫ������ɫ��ʪ�����������ʹ��ɫ������ɫ��������
��3����������ˮ��Ӧ��Cl2+H2O=HCl+HClO��HClO���������ԣ�ǿ�����ԣ�Ư�����Լ����ȶ�����������
��1������ʵ��Ŀ�ļ�������������ʷ�������ȡ����������Ҷ��������ж���Ӧ��β������װ�ã�
��2�����ݶ��������ˮ��Ӧ�ķ���ʽ�жϣ�
��� �⣺��1����ʵ��ͨ���۲����������ʪ�����ֱ����ɫ������Ư�����ã����о������������û��Ư���ԣ�
�ʴ�Ϊ��̽��Cl2�ڳ�ʪ����ʱ�Ƿ����Ư���ԣ�
��2��Ũ������������������ˮ�ԣ���������������A�е�ˮ����������ͨ��Aƿ��Ũ������Bƿ�и���IJ�������ɫ��˵����������û��Ư���ԣ�Cƿ��������ˮ������ˮ����ɫ������ɫ��˵��ʪ��������ɫ������Ư�����ã�
��ΪB�ڵ���ɫ��������ɫ��C�ڵ���ɫ������ɫ��
�ʴ�Ϊ����������A�е�ˮ������B�ڵ���ɫ��������ɫ��C�ڵ���ɫ������ɫ��
��3��������ˮ����H+��HClO��HClO����ǿ�����ԣ�Ư���ԣ��ʿ�ʹ��ɫʯ����Һ�ȱ�����ɫ���ʴ�Ϊ��ʯ����Һ�ȱ�����ɫ��
��1����ʵ����̽�������SO2�ܲ���ʹƷ����ɫ�����Զ�������ͨ��Ʒ����ҺǰҪ���и�����������ж������Զ������岻��ֱ���ſգ���Ҫ��β������װ�ã�
�ʴ�Ϊ���ٶ�������û�и����ȱ�ٶ�������ĸ���װ�ã�����û��β������װ�ã�
��2�����������ˮ��Ӧ����������SO2+H2O?H2SO3��������Ʒ����ɫ�Ŀ�����H2SO3���ʴ�Ϊ��H2SO3��
���� ���⿼�����������ʼ�������ˮ��Ӧ�������Ư���ԣ���������������Լ�ʹƷ����ɫ��ԭ����Ŀ�ѶȲ���

| A�� | CH3CH2CH3 | B�� | CH3CH2CH2CH3 | C�� | CH3COCH2CH3 | D�� | 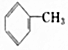 |
 �����Dz�ͬ�¶���ˮ�����ӻ������ݣ�
�����Dz�ͬ�¶���ˮ�����ӻ������ݣ�| �¶�/�� | 25 | t1 | t2 |
| ˮ�����ӻ� | 1��10-14 | a | 1��10-12 |
��1����25��t1��t2����a�������������������=����1��10-14
��2����t2�¶��²��ij��ҺpH=7������Һ�Լ��ᡱ��������С����ԣ�
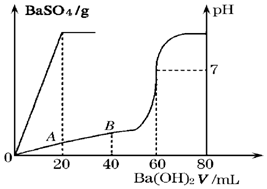
��3��25��ʱ�������������Ļ��Һ20mL������0.05mol•L-1��Ba��OH��2��Һʱ������BaSO4����������Һ��pH������ͼ��ʾ�ı仯��
���ԭ���Һ���������������ʵ���Ũ�ȷֱ��ǣ�c��H2SO4��=0.05 mol•L-1��c��HCl��=0.2 mol•L-1
����A��ʱ��pH=1 ������Һ��Ϻ�������䣩
��1����PM2.5����������ˮ�����Ƴɴ���������
����ø���������ˮ���������ӵĻ�ѧ��ּ���ƽ��Ũ�������
| ���� | K+ | Na+ | NH4+ | SO42- | NO3- | Cl- |
| Ũ��mol/L | 4��10-6 | 6��10-6 | 2��10-5 | 4��10-5 | 3��10-5 | 2��10-5 |
��2������β����NOx��CO�����ɣ�
����֪����������NO�ķ�ӦΪ��N2��g��+O2��g��?2NO��g����H��0���£������ܱ������У�����˵������˵���÷�Ӧ�ﵽ��ѧƽ��״̬����D
A�����������ܶȲ��ٱ仯 B����������ƽ����Է����������ٱ仯
C��N2��O2��NO�����ʵ���֮��Ϊ1��1��2 D�������İٷֺ������ٱ仯
������ȼ�Ͳ���ȫȼ��ʱ����CO���������밴���з�Ӧ��ȥCO��2CO��g��=2C��s��+O2��g������֪�÷�Ӧ�ġ�H��0����������ܷ�ʵ�ֲ��ܣ���ܡ����ܡ���
��3������β����������Ҫԭ����2NO��g��+2CO��g��$\frac{\underline{\;����\;}}{\;}$2CO2��g��+N2��g������H��0�����÷�Ӧ�ھ��ȡ����ݵ��ܱ���ϵ�н��У�����ʾ��ͼ��ȷ����˵����Ӧ�ڽ��е�t1ʱ�̴ﵽƽ��״̬����BD������ţ�������ͼ��v����K��n��w�ֱ��ʾ����Ӧ���ʡ�ƽ�ⳣ�������ʵ���������������
��4�������ŷŵĵ������úȼ�ղ����Ķ��������ǵ������������ġ�������ס�֮һ������̿�ɴ���������Ⱦ��NO����5L�ܱ������м���NO�ͻ���̿�����������ʣ���һ����������������E��F�����¶ȷֱ���T1���T2��ʱ����ø�����ƽ��ʱ���ʵ�����n/mol�������
| ���� �¶�/�� | ����̿ | NO | E | F |
| ��ʼ | 3.000 | 0.10 | 0 | 0 |
| T1 | 2.960 | 0.020 | 0.040 | 0.040 |
| T2 | 2.975 | 0.050 | 0.025 | 0.025 |
����T1��T2����÷�Ӧ�ġ�H_��0�����������������=������
��������ӦT1��ʱ�ﵽ��ѧƽ�����ͨ��0.1molNO���壬��ﵽ�»�ѧƽ��ʱNO��ת����Ϊ80%��
| A�� | ����Ҫʹ�ô�����ɳ����ʹ����½�ӵĺ�ɳ������ĺ�ɳ | |
| B�� | ���������ε�Ư���뺬����Ľ������ʹ�û�����ж����� | |
| C�� | Ư�ۺ�����������������ˮ�Ĵ��������ߵ����ú�ԭ������ͬ | |
| D�� | ��ɫ��ѧ�ĺ����Ǵ�Դͷ�ϼ��ٺ��������������Ի�������Ⱦ |
��1����֪����C��s��+O2��g��=CO2��g����H1=-393.5kJ•mol-1
��C��s��+CO2��g��=2CO��g����H2=+172.5kJ•mol-1
��4Fe��s��+3O3��g��=2Fe2O3 ��s����H3=-1651.0kJ•mol-1
CO��ԭ���������Ȼ�ѧ����ʽΪFe2O3��s��+3CO��g���T2Fe��s��+3CO2��g����H=-23.5kJ��mol-1��
��2����¯���������ĸ�¯���к���CO��H2��CO2�����壬����CO��H2�ڴ��������ºϳɼ״����Ǽ�����Ⱦ����Լ��Դ��һ���¾ٴ룬��Ӧԭ�����£�CO��g��+2H2��g��?CH3OH��g����H���������ͬ�����������ܱ������зֱ����1molCO��2moL H2�����ƽ��������CH3OH����������ڲ�ͬѹǿ�����¶ȵı仯��ͼ1��
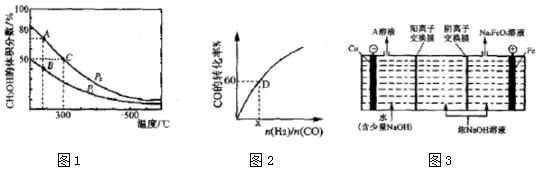
����ͼ1A��B��C�����У�ѡ�����������Ӧ���ĵ㣮
| ��Ӧ����v | ƽ�ⳣ��K | ƽ��ת���ʦ� |
��һ���¶��£�CO��ת��������ʼͶ�ϱ�[$\frac{n��{H}_{2}��}{n��CO��}$]�ı仯��ϵ��ͼ2��ʾ�����D��������ת����Ϊ40%����x=3��
��3�����Ȼ�����һ����Ҫ�Ļ��������������ʴ��·�壮ij��ʴ��Һ�к��� 0.5mol•L-l Fe3+ ��0.26mol•L-1 �� Cu2+����ʹ Fe3+��ȫ����[c��Fe3+����4��10-5 mol•L-1]��Cu2+�����������������Һ pH �ķ�ΧΪ3��pH��5��[KSp��Cu��OH��2=2.6��10-19��Ksp��Fe��OH��3=4��10-38��
��4��������أ�K2FeO4����һ�����͡���Ч�������ɫˮ����������ҵ��ͨ�����Ũ NaOH��Һ���Ʊ�Na2FeO4��Ȼ��ת��ΪK2FeO4�����ԭ����ͼ3��ʾ��
��A��Һ�����ʵijɷ�ΪNaOH���ѧʽ���������ĵ缫��ӦʽΪFe-6e-+8OH-=FeO42-+4H2O��