��Ŀ����
20�� 50mL 0.50mol•L-1������50mL 0.55mol•L-1 NaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ�
50mL 0.50mol•L-1������50mL 0.55mol•L-1 NaOH��Һ����ͼ��ʾ��װ���н����кͷ�Ӧ��ͨ���ⶨ��Ӧ���������ų��������ɼ����к��ȣ��Իش��������⣺
��1����С�ձ���������ֽ���������Ǽ���ʵ������е�������ʧ��
��2�����ձ����粻��Ӳֽ�壬��õ��к�����ֵ��ƫС���ƫ����ƫС������Ӱ�족��������ճ������ʵ�ʸ�ʵ���ڱ��±�����Ч�����ã�
��3��ʵ���и���60mL 0.50mol•L-1������50mL 0.55mol•L-1 NaOH��Һ���з�Ӧ��������ʵ����ȣ����ų�����������ȣ����ȡ���������ȡ������������ɣ���Ӧ�ų����������������Լ�������Ķ����йأ�
��4������ͬŨ�Ⱥ�����İ�ˮ����NaOH��Һ��������ʵ�飬��õ��к��ȵ���ֵ��ƫС����50mL 0.5mo1•L-1NaOH��Һ��������ʵ�飬��õ��к��ȵ���ֵ����Ӱ�죨�ƫ����ƫС������Ӱ�족��
���� ��1���к��Ȳⶨʵ��ɰܵĹؼ��DZ��¹�����
��2�����ձ����粻��Ӳֽ�壬��ʹһ��������ɢʧ���ճ����������Ǿ����õ����±����ڱ��±��н���ʵ�鱣��Ч������ã�
��3����Ӧ�ų����������������Լ�������Ķ����йأ��������к��ȵĸ����ʵ�����ش�
��4������������ʵ������ȷ����������к��ȵĸ��������
��� �⣺��1���к��Ȳⶨʵ��ɰܵĹؼ��DZ��¹�������С�ձ�֮��������ֽ���������Ǽ���ʵ������е�������ʧ��
�ʴ�Ϊ������ʵ������е�������ʧ��
��2�����ձ����粻��Ӳֽ�壬��ʹһ��������ɢʧ����õ��к�����ֵ�����С�����ճ�����ʵ�ʸ�ʵ���ڱ��±���Ч�����ã�
�ʴ�Ϊ��ƫС�����±���
��3����Ӧ�ų����������������Լ�������Ķ����йأ���60mL 0.50mol•L-1������50mL 0.55mol•L-1 NaOH��Һ���з�Ӧ��������ʵ����ȣ�����ˮ�������࣬���ų�������ƫ�ߣ������к��ȵľ���ǿ���ǿ�Ӧ����1molˮʱ�ų����ȣ������������أ��к�����ֵ��ȣ�
�ʴ�Ϊ������ȣ���Ӧ�ų����������������Լ�������Ķ����йأ�
��4����ˮΪ����������Ϊ���ȹ��̣������ð�ˮ����ϡ����������Һ��Ӧ����Ӧ�ų�������ƫС���к��ȵ���ֵ��ƫС��
�к�����ǿ���ǿ�Ӧ����1molˮʱ�ų��������������������أ�������5mL 0.50mol/L NaOH��Һ��������ʵ�飬��õ��к��ȵ���ֵ��Ӱ�죻
�ʴ�Ϊ��ƫС����Ӱ�죮
���� ���⿼��ѧ���й��к��ȵIJⶨ֪ʶ��ע���к������ᡢ������ʵ����أ��ѶȲ���

| A�� | pH=5��H2S��Һ�У�c��H+��=c��HS-��=1��10-5mol•L-1 | |
| B�� | pH=a�İ�ˮ��Һ��ϡ��10������pH=b����a=b+1 | |
| C�� | pH=2��CH3COOH��Һ��pH=12��NaOH��Һ�������ϣ����Һ��pH��7 | |
| D�� | a1mLb1mol•L-1�����a2mLb2mol•L-1��ˮ��������ԣ�b1=b2����a1��a2 |
| A�� | ETFE�����п��ܴ��ڡ�-CH2-CH2-CF2-CF2-�������ӷ�ʽ | |
| B�� | �ϳ�ETFE���ϳɾ�ȫ���ұ�ϩ�ķ�Ӧ��Ϊ�Ӿ۷�Ӧ | |
| C�� | ��ȫ���ұ�ϩ���ӵĽṹ��ʽ����Ϊ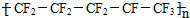 | |
| D�� | �ķ���ϩ������ԭ�Ӿ���ͬһƽ���� |
| A�� | ���� | B�� | ���� | C�� | �ϳ��� | D�� | �ϳ���ά |
| A�� | һ���¶��£���ӦMgCl2��l���TMg��l��+Cl2��g���ġ�H��0��S��0 | |
| B�� | ˮ�ⷴӦNH4++H2O?NH3•H2O+H+�ġ�H��0 | |
| C�� | ˮ�ܡ����ܡ���Ȼ�����ǿ�������Դ | |
| D�� | ���ǻ��Ϸ�Ӧ���Ƿ��ȷ�Ӧ���ֽⷴӦ�������ȷ�Ӧ |
| A�� | IVA��Ԫ���⻯��е�˳���ǣ�GeH4��SiH4��CH4����VIIA��Ԫ���⻯��е�˳��Ҳ�ǣ�HBr��HCl��HF | |
| B�� | CH4����������ṹ����SiH4Ҳ����������ṹ | |
| C�� | ���������Ӧ��4Li+O2$\frac{\underline{\;����\;}}{\;}$2 Li2O��������������Ӧ��4Na+O2$\frac{\underline{\;����\;}}{\;}$2Na2O | |
| D�� | CaCO3��ϡ���ᷴӦ����CO2����CaSO3��ϡ���ᷴӦ����SO2 |
| A�� | F-�Ľṹʾ��ͼ��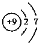 | |
| B�� | ԭ�Ӻ�����10�����ӵ���ԭ�ӣ�${\;}_{8}^{18}$O | |
| C�� | �����ĵ���ʽ�� | |
| D�� | ��ϩ�Ľṹ��ʽ��CH2CH2 |
| A�� | �÷�ӦΪ�û���Ӧ | B�� | �þ���Ϊ���Ӿ��� | ||
| C�� | ��������һ���µĻ����� | D�� | �����ʵ���Է���������2400 |

 ��
��

 ���������ᣨŨ������Ũ��������ڼ��������£�����������Ӧ���ɵ�ԭ�Ӽ�λ��һȡ����Ļ�ѧ����ʽΪ
���������ᣨŨ������Ũ��������ڼ��������£�����������Ӧ���ɵ�ԭ�Ӽ�λ��һȡ����Ļ�ѧ����ʽΪ ��
��
 ��
��