��Ŀ����
13����ҵ�ϳ��ú����ˮ����Na2S2O3•5H2O��ʵ���ҿ�����ͼװ�ã���ȥ���ּг�������ģ���������̣�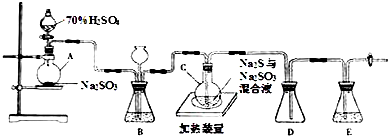
��ƿC�з�����Ӧ���£�
Na2S��aq��+H2O��l��+SO2��g���TNa2SO3��aq��+H2S��aq���� ����
2H2S��aq��+SO2��g���T3S��s��+2H2O��l���� ����
S��s��+Na2SO3��aq�� $\frac{\underline{\;\;��\;\;}}{\;}$Na2S2O3��aq���� ����
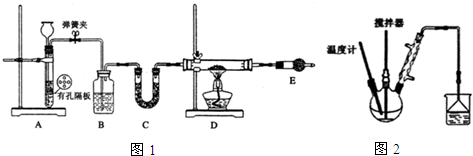
��1������A������Բ����ƿ��������װ��ɺر����˻�������װ��B�еij���©����ע��Һ�����γ�һ��Һ������Һ���߶ȱ��ֲ��䣬������װ�����������ã�װ��E��ΪNaOH��Һ��
��2��װ��B������֮һ�ǹ۲�SO2���������ʣ����е�Һ�����ѡ��c��
a������ˮ���� b������Na2SO3��Һ���� c������NaHSO3��Һ����d������NaHCO3��Һ
��֪��Ӧ������Խ���������ƿC�з�Ӧ�ﵽ�յ����������Һ����壨�������ʧ��������Ӧ���ڿ��þƾ����ʵ���������A��ʵ�����þƾ��Ƽ���ʱ����ʹ��ʯ��������������ad��
a���ձ�����������b��������������c���Թܡ���������d����ƿ
��3����Ӧ��ֹ������C�е���Һ������Ũ������ȴ�ᾧ��������Na2S2O3•5H2O�����п��ܺ���Na2SO3��Na2SO4�����ʣ����������Լ����ʵ�飬����Ʒ���Ƿ����Na2SO4����Ҫ˵��ʵ�����������ͽ���ȡ������Ʒ��������ϡ���ᡢ���á�ȡ�ϲ���Һ������ˣ�ȡ��Һ�����μ�BaCl2��Һ�������ֳ�����˵������Na2SO4���ʣ�
��֪Na2S2O3•5H2O�����ֽ⣺S2O 32-+2H+�TS��+SO2��+H2O
��ѡ����Լ���ϡ���ᡢϡ���ᡢϡ���ᡢBaCl2��Һ��AgNO3��Һ��
���� װ��A���Ʊ���������C�ж��������������������Ʒ�Ӧ����Na2S2O3��Eװ�ý���β����������ֹ��Ⱦ����������ʢ���������Ʒ�Ӧ��DΪ��ȫƿ����ֹ������װ��B�������۲�SO2���������ʣ�Ҳ���Ը��ݳ���©����Һ��仯�ж�װ���Ƿ������������ã�
��1������������������֪����A��Բ����ƿ��Һ���߶ȱ��ֲ��䣬˵�����������ã�װ��E������β����SO2��H2S�����ã�
��2��SO2������ˮ������Na2SO3��Һ��NaHCO3��Һ��Ӧ������NaHSO3��Һ���ܽ�Ƚ�С��
���з���S��g��+Na2SO3��aq��$\frac{\underline{\;\;��\;\;}}{\;}$Na2S2O3��aq������Ӧ�ﵽ�յ�ʱS��ȫ�ܽ⣻
�ձ�����ƿ��Ҫ����ʱ����ʹ��ʯ������
��4���������ᣬNa2S2O3��Ӧ����S���������Ծ��ú�ȡ�ϲ���Һ���μ�BaCl2��Һ��������Һ���Ƿ�����������ӣ�
��� �⣺װ��A���Ʊ���������C�ж��������������������Ʒ�Ӧ����Na2S2O3��Eװ�ý���β����������ֹ��Ⱦ����������ʢ���������Ʒ�Ӧ��DΪ��ȫƿ����ֹ������װ��B�������۲�SO2���������ʣ�Ҳ���Ը��ݳ���©����Һ��仯�ж�װ���Ƿ������������ã�
��1������������������֪����A��Բ����ƿ��
������װ��ɺر����˻�������װ��B�еij���©����ע��Һ�����γ�һ��Һ������Һ���߶ȱ��ֲ��䣬�����������ã�
װ��E������β����SO2��H2S�����ã���ѡ��NaOH��Һ��
�ʴ�Ϊ��Բ����ƿ��Һ���߶ȱ��ֲ��䣻NaOH��
��2���۲�SO2���������ʣ���ͨ���۲���Һ�в������ݵĿ������жϣ�a�ܽ����������ˮ�Ͷ�������Ӧ���������ᣬb�ж���������������Ʒ�Ӧ�������������ƣ�d��̼���������������Ӧ���ɶ�����̼��ֻ��c�б���NaHSO3��Һ���ܽ���������ʺ�ͨ���۲���Һ�в������ݵĿ������ж϶�����������ɿ��������з���S��g��+Na2SO3��aq��$\frac{\underline{\;\;��\;\;}}{\;}$Na2S2O3��aq������Ӧ�ﵽ�յ�ʱS��ȫ�ܽ⣬�ɹ۲쵽��Һ����壨�������ʧ����
�ձ�����ƿ��Ҫ����ʱ����ʹ��ʯ������
�ʴ�Ϊ��c����Һ����壨�������ʧ����ad��
��3������Ʒ���Ƿ����Na2SO4��ʵ�鷽��Ϊ��ȡ������Ʒ��������ϡ���ᡢ���á�ȡ�ϲ���Һ������ˣ�ȡ��Һ�����μ�BaCl2��Һ�������ֳ�����˵������Na2SO4���ʣ�
�ʴ�Ϊ��ȡ������Ʒ��������ϡ���ᡢ���á�ȡ�ϲ���Һ������ˣ�ȡ��Һ�����μ�BaCl2��Һ�������ֳ�����˵������Na2SO4���ʣ�
���� ���⿼��ʵ�鷽���ķ��������ۣ��漰����������Ʊ��������Լ��顢���Ӽ��顢�Բ����ķ������۵ȣ�����ʵ�����������֪ʶ�ۺ�Ӧ�������Ŀ��飬��Ŀ�Ѷ��еȣ�

| A�� | HCl | B�� | NH3•H2O | C�� | H2SO3 | D�� | HClO |
| A�� | 10 mol | B�� | 11 mol | C�� | 12 mol | D�� | 13 mol |
| Y | Z |
| R |
| A�� | R���������Ӧ��ˮ����һ����ǿ�� | |
| B�� | X��Y��Z����Ԫ�ؼ����γɹ��ۻ����������γ����ӻ����� | |
| C�� | ��X��Y��Z����Ԫ���е�����������ɵľ���10�����ӵ�����2�� | |
| D�� | Z��W�γɵĻ�������ֻ�ܺ������Ӽ� |
 ��֪�������ȼ��Ϊ�£�N2H4����������Ϊ�������⣬������ֹ��������ѺϽ���ϣ���ش��������⣺
��֪�������ȼ��Ϊ�£�N2H4����������Ϊ�������⣬������ֹ��������ѺϽ���ϣ���ش��������⣺��1��N2H4��H2O2�����Ԫ���е�һ����������Ԫ����N��
��2���ѵ�ԭ��������22�����̬ԭ�ӵĺ�������Ų�ʽΪ[Ar]3d24s2��
��3��1molN2H4�������������Լ�����ĿΪ4NA��
��4��H2O2�����ֽ�ΪH2O��H2O�Ŀռ乹��ΪV�Σ�������ԭ�ӵ��ӻ��������Ϊsp3��
��5��H2S��H2O2����Ҫ�������������ʾ��
| ���� | �۵�/K | �е�/K | ˮ���ܽ�ȣ���״���� |
| H2S | 187 | 202 | ÿ��ˮ���ܽ�2.6L |
| H2O2 | 272 | 423 | ������Ȼ��� |
��6������þ�͵���������������زյ������β��ϣ������ܣ�����þ���������ƣ�����ڡ���С�ڡ�����
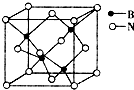
��7������������ľ�����ͼ��ʾ�����ھ��������ԭ�ӵ���λ��Ϊ4���������߳�Ϊa pm�����������������ܶ���$\frac{100}{{{N_A}{{��a��{{10}^{-10}}��}^3}}}$g•cm-3��ֻҪ������ʽ������٤������ΪNA����
| A�� | Q��Y��ɵij���������һ���������ʣ��ṹ�к��й��ۼ� | |
| B�� | ��֪WRZQ3��Һ�����ԣ�����WRZQ3��������ˮ���ܴٽ�ˮ�ĵ��� | |
| C�� | Ԫ��Q��W�γɵ����ֳ����������к�����ͬ���������������� | |
| D�� | Ԫ��T��Q��W��Y��ԭ�Ӱ뾶��СΪT��Q��Y��W |
| A�� | +45 kJ/mol | B�� | +32.7 kJ/mol | C�� | -45 kJ/mol | D�� | ������ |