��Ŀ����
2009�꣬��λ������ѧ�Һ�һλ��ɫ�п�ѧ���Ժ�����Ľṹ���ܵ��о��������ŵ������ѧ���������屻��Ϊ������ѧ�����еĹ���ʦ��������к��к��ǣ��������йغ��ǵ�ת����ϵͼ��A��E��Ϊ�л��

��֪�����ǵĽṹ��ʽΪ��

�Իش��������⣺
��1�������к��������ŵ�����Ϊ ��
��2��1mol��������� mol���ᷴӦ��������
��3��д����Ӧ�ٵĻ�ѧ����ʽ��
��4��A�Ľṹ��ʽΪ �������� �����л���Ӧ���ͣ���
��5��д��B������������Һ��Ӧ�Ļ�ѧ����ʽ��
��6��ij�л�����C��ͬϵ�����Է�������Ϊ88����ͬ���칹��������ܷ���ˮ�ⷴӦ���ڷ���ˮ�ⷴӦ��IJ���֮һ����NaOH��Һ��Ӧ�����ͬ���칹��Ľṹ��ʽΪ��д�ṹ��ʽ�� �� �� �� ��

��֪�����ǵĽṹ��ʽΪ��

�Իش��������⣺
��1�������к��������ŵ�����Ϊ
��2��1mol���������
��3��д����Ӧ�ٵĻ�ѧ����ʽ��
��4��A�Ľṹ��ʽΪ
��5��д��B������������Һ��Ӧ�Ļ�ѧ����ʽ��
��6��ij�л�����C��ͬϵ�����Է�������Ϊ88����ͬ���칹��������ܷ���ˮ�ⷴӦ���ڷ���ˮ�ⷴӦ��IJ���֮һ����NaOH��Һ��Ӧ�����ͬ���칹��Ľṹ��ʽΪ��д�ṹ��ʽ��
���㣺�л�����ƶ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
�����������ڸ����Ȼ���������·�Ӧ������Ԫ��������������Ϣ֪��������ȩ������ȩ����Զ��̼ԭ���ϵ��ǻ���Ӧ������Ԫ�������ṹ��ʽΪ��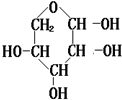 ��
�� �ߵ��ᷴӦ����A��
�ߵ��ᷴӦ����A��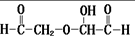 �����������Ϣ֪��A�Ľṹ��ʽΪ��HCOOH��
�����������Ϣ֪��A�Ľṹ��ʽΪ��HCOOH��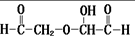 �����������º�ˮ��Ӧ����B��C�����������Ϣ֪����������OHCCH2OH��OHCCHO��B�ܷ���������Ӧ����D��C�ܱ���������B����B��OHCCHO��C��OHCCH2OH��DΪHOOCCOOH��E��HOCH2CH2OH��������ʵĽṹ�����ʷ������
�����������º�ˮ��Ӧ����B��C�����������Ϣ֪����������OHCCH2OH��OHCCHO��B�ܷ���������Ӧ����D��C�ܱ���������B����B��OHCCHO��C��OHCCH2OH��DΪHOOCCOOH��E��HOCH2CH2OH��������ʵĽṹ�����ʷ������
 ��
�� �ߵ��ᷴӦ����A��
�ߵ��ᷴӦ����A��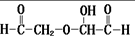 �����������Ϣ֪��A�Ľṹ��ʽΪ��HCOOH��
�����������Ϣ֪��A�Ľṹ��ʽΪ��HCOOH��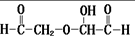 �����������º�ˮ��Ӧ����B��C�����������Ϣ֪����������OHCCH2OH��OHCCHO��B�ܷ���������Ӧ����D��C�ܱ���������B����B��OHCCHO��C��OHCCH2OH��DΪHOOCCOOH��E��HOCH2CH2OH��������ʵĽṹ�����ʷ������
�����������º�ˮ��Ӧ����B��C�����������Ϣ֪����������OHCCH2OH��OHCCHO��B�ܷ���������Ӧ����D��C�ܱ���������B����B��OHCCHO��C��OHCCH2OH��DΪHOOCCOOH��E��HOCH2CH2OH��������ʵĽṹ�����ʷ���������
�⣺�����ڸ����Ȼ���������·�Ӧ������Ԫ��������������Ϣ֪��������ȩ������ȩ����Զ��̼ԭ���ϵ��ǻ���Ӧ������Ԫ�������ṹ��ʽΪ�� ��
�� �ߵ��ᷴӦ����A��
�ߵ��ᷴӦ����A��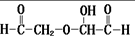 �����������Ϣ֪��A�Ľṹ��ʽΪ��HCOOH��
�����������Ϣ֪��A�Ľṹ��ʽΪ��HCOOH��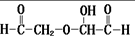 �����������º�ˮ��Ӧ����B��C�����������Ϣ֪����������OHCCH2OH��OHCCHO��B�ܷ���������Ӧ����D��C�ܱ���������B����B��OHCCHO��C��OHCCH2OH��DΪHOOCCOOH��E��HOCH2CH2OH��
�����������º�ˮ��Ӧ����B��C�����������Ϣ֪����������OHCCH2OH��OHCCHO��B�ܷ���������Ӧ����D��C�ܱ���������B����B��OHCCHO��C��OHCCH2OH��DΪHOOCCOOH��E��HOCH2CH2OH��
��1�����ݺ��ǵĽṹ֪�������еĹ��������ǻ���ȩ�����ʴ�Ϊ���ǻ���ȩ����
��2��1mol���Ǻ���4mol�ǻ�������1mol��������ܺ�4mol���ᷢ��������Ӧ���ʴ�Ϊ��4��
��3�������ڸ����Ȼ��������������·����ķ�ӦΪ��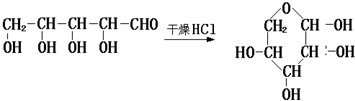 ��
��
�ʴ�Ϊ�� ��
��
��4��ͨ�����Ϸ���֪��A�Ǽ��ᣬ��ṹ��ʽΪ��HCOOH��C�����ӳɷ�Ӧ����E���ʴ�Ϊ��HCOOH���ӳɷ�Ӧ��
��5��B��OHCCHO��B��������Һ����������Ӧ����Ӧ����ʽΪ��OHC-CHO+4Ag��NH3��2OH
NH4OOC-COONH4+4Ag��+6NH3+2H2O���ʴ�Ϊ��OHC-CHO+4Ag��NH3��2OH
NH4OOC-COONH4+4Ag��+6NH3+2H2O��
��6��C��OHCCH2OH��ij�л�����C��ͬϵ�����Է�������Ϊ88����ͬ���칹��������ܷ���ˮ�ⷴӦ˵�������������ڷ���ˮ�ⷴӦ��IJ���֮һ����NaOH��Һ��Ӧ��˵��ˮ�����������ʹ����÷�����̼ԭ�Ӹ���=
+2=4������������C��ͬϵ��Ľṹ��ʽΪ��HCOOCH2CH2CH3��HCOOCH��CH3��2��CH3COOCH2CH3��CH3CH2COOCH3��
�ʴ�Ϊ��HCOOCH2CH2CH3��HCOOCH��CH3��2��CH3COOCH2CH3��CH3CH2COOCH3��
 ��
�� �ߵ��ᷴӦ����A��
�ߵ��ᷴӦ����A��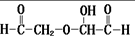 �����������Ϣ֪��A�Ľṹ��ʽΪ��HCOOH��
�����������Ϣ֪��A�Ľṹ��ʽΪ��HCOOH��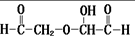 �����������º�ˮ��Ӧ����B��C�����������Ϣ֪����������OHCCH2OH��OHCCHO��B�ܷ���������Ӧ����D��C�ܱ���������B����B��OHCCHO��C��OHCCH2OH��DΪHOOCCOOH��E��HOCH2CH2OH��
�����������º�ˮ��Ӧ����B��C�����������Ϣ֪����������OHCCH2OH��OHCCHO��B�ܷ���������Ӧ����D��C�ܱ���������B����B��OHCCHO��C��OHCCH2OH��DΪHOOCCOOH��E��HOCH2CH2OH����1�����ݺ��ǵĽṹ֪�������еĹ��������ǻ���ȩ�����ʴ�Ϊ���ǻ���ȩ����
��2��1mol���Ǻ���4mol�ǻ�������1mol��������ܺ�4mol���ᷢ��������Ӧ���ʴ�Ϊ��4��
��3�������ڸ����Ȼ��������������·����ķ�ӦΪ��
 ��
���ʴ�Ϊ��
 ��
����4��ͨ�����Ϸ���֪��A�Ǽ��ᣬ��ṹ��ʽΪ��HCOOH��C�����ӳɷ�Ӧ����E���ʴ�Ϊ��HCOOH���ӳɷ�Ӧ��
��5��B��OHCCHO��B��������Һ����������Ӧ����Ӧ����ʽΪ��OHC-CHO+4Ag��NH3��2OH
| �� |
| �� |
��6��C��OHCCH2OH��ij�л�����C��ͬϵ�����Է�������Ϊ88����ͬ���칹��������ܷ���ˮ�ⷴӦ˵�������������ڷ���ˮ�ⷴӦ��IJ���֮һ����NaOH��Һ��Ӧ��˵��ˮ�����������ʹ����÷�����̼ԭ�Ӹ���=
| 88-16��2-1��4-12��2 |
| 14 |
�ʴ�Ϊ��HCOOCH2CH2CH3��HCOOCH��CH3��2��CH3COOCH2CH3��CH3CH2COOCH3��
���������⿼�����л�����ƶϣ���ȷ���������Ϣ�жϼ��ͳɼ���ʽ�ǽⱾ��ؼ����ٽ�����ʵĹ����ż���������������ɣ��ѵ���ȷ������������C��ͬϵ��ṹ��ʽ��ע�����ͬϵ�����ʽ�ص���ȷ��C��ͬϵ����̼ԭ�Ӹ�������Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
2.8g Feȫ������һ��Ũ�ȡ�200mL��HNO3��Һ�У��õ���״���µ�����1.12L����÷�Ӧ����Һ��pHΪ1������Ӧǰ����Һ����仯���Բ��ƣ��������й��ж���ȷ���ǣ�������
| A����Ӧ����Һ��c��NO3-��=0.85mol/L |
| B����Ӧ�����Һ�����ܽ�1.4gFe |
| C����Ӧ����Һ����Ԫ�ؿ�����Fe2+��ʽ���� |
| D��1.12L���岻������NO��NO2�Ļ������ |
��NAΪ�����ӵ�������������������ȷ���� ��������
| A����״���£�22.4L�ױ��к�C-H����ĿΪ8NA |
| B�����³�ѹ�£�7.8g���к���˫������ĿΪ0.3NA |
| C�����ͱ�����Ļ����1mol����ȫȼ������O2�ķ�����Ϊ7.5NA |
| D�����³�ѹ�£�5.2g���ͱ���ϩ�Ļ�����к���ԭ����ĿΪ0.8NA |
����ͼװ�ò��ܴﵽʵ��Ŀ���ǣ�������


| A���ü�ͼװ�ñȽ�NaHCO3��Na2CO3�����ȶ��� |
| B������ͼװ���Ʊ�Fe��OH��2 |
| C���ñ�ͼװ���Ʊ����ռ�����NO2���� |
| D���ö�ͼװ����ȡ���������� |
������CO2�ֱ�ͨ�����и���Һ�У������������ܴ���������ǣ�������
| A��K+��AlO2-��Cl-��NO3- |
| B��Na+��CH3COO-��C6H5O-��HCO3- |
| C��Na+��ClO-��SO42-��Cl- |
| D��NH4+��Al3+��SO42-��H+ |
���������ĸ�˵����ȷ���ǣ�������

| A����֪ͼ������ϵFe3++3SCN-?Fe��SCB��3��c[Fe��SCN��3]���¶�T��ƽ��ͼ����A����B����ȣ�A���Fe3+Ũ�ȴ� |
| B��ͼ�ڱ�ʾþ�����ֱ����Ũ�ȡ�������Ĺ���ϡ���ᷴӦ��������������V��H2�� ��ʱ��t�Ĺ�ϵ����Ӧ��þ�����ķ�Ӧ����֮��Ϊ2��3 |
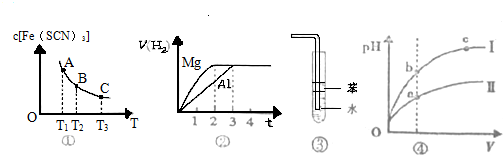
| C��ͼ�ۿ���������HCl���壬�������� |
| D��ͼ����ij�¶��£���ͬ�������ͬpH������ʹ�����Һ�ֱ��ˮϡ�ͣ�pH����Һ���V�仯�����ߣ�IIΪ����ϡ��ʱpH�ı仯���� |
 ��֪A��B��D��E�������ʾ�������Ԫ�أ�DΪ���嵥�ʣ�EΪ��ɫ��ĩ��A��B�ֱ�E������ֽ����D��Bͨ��ΪҺ̬��������Ԫ����ɣ�ԭ�Ӹ�����Ϊ1��1����1molB��18mol���ӣ��Իش��������⣮
��֪A��B��D��E�������ʾ�������Ԫ�أ�DΪ���嵥�ʣ�EΪ��ɫ��ĩ��A��B�ֱ�E������ֽ����D��Bͨ��ΪҺ̬��������Ԫ����ɣ�ԭ�Ӹ�����Ϊ1��1����1molB��18mol���ӣ��Իش��������⣮