��Ŀ����
6��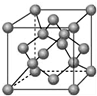 ��֪A��B��C��D����Ԫ�����ڱ���ǰ36�ŵ�Ԫ�أ�AԪ�ص�һ�ֺ���û�����ӣ�B�Ļ�̬ԭ�Ӻ��������7�ֲ�ͬ���˶�״̬��CԪ�������ǽ������ϵ����ǣ����ĵ��ʿ�����������оƬ��DԪ���ǵؿ��к����ڶ��Ľ���Ԫ�أ���ش�
��֪A��B��C��D����Ԫ�����ڱ���ǰ36�ŵ�Ԫ�أ�AԪ�ص�һ�ֺ���û�����ӣ�B�Ļ�̬ԭ�Ӻ��������7�ֲ�ͬ���˶�״̬��CԪ�������ǽ������ϵ����ǣ����ĵ��ʿ�����������оƬ��DԪ���ǵؿ��к����ڶ��Ľ���Ԫ�أ���ش���1��BA3���ӵ�����ṹ�������Σ��û�����������ˮ��ԭ������ǣ���ѡ����ĸ��ABC
A���û���������Ǽ��Է���
B���û�������ӿ���ˮ����֮���γ����
C���û����������ˮ��Һ�в�����ˮ������Ӧ
��2��BA3���۵��CA4���۵�ߣ�����ߡ��͡���ԭ���ǰ�������֮���γ������
��3��Dλ��Ԫ�����ڱ��еڢ��壬����+3�����ӵļ��鷽���ǣ�ȡ���������Һ�������Թ��У��μ�1-2��KSCN��Һ������Һ���Ѫ��ɫ�������Һ�к���Fe3+��
��4����ͼ����C���ʹ��ɵľ����һ������������þ����ı߳�Ϊa cm��NA��ʾ�����ӵ�������ֵ����þ�����ܶ���$\frac{8��28}{{a}^{3}��{N}_{A}}$g/cm3����ֻҪ���г���ʽ����
���� A��B��C��D����Ԫ�����ڱ���ǰ36�ŵ�Ԫ�أ�AԪ�ص�һ�ֺ���û�����ӣ���AΪHԪ�أ�B�Ļ�̬ԭ�Ӻ��������7�ֲ�ͬ���˶�״̬����BΪNԪ�أ�CԪ�������ǽ������ϵ����ǣ����ĵ��ʿ�����������оƬ����CΪSi��DԪ���ǵؿ��к����ڶ��Ľ���Ԫ�أ���DΪFe��
��1��NH3����Ϊ�����νṹ������ˮ��Ӧ����һˮ�ϰ�����ˮ����֮���γ����������������ˮ���Ӷ��Ǽ��Է��ӣ��������ܣ�
��2����������֮���γ������SiH4����֮��Ϊ���»���������ȷ��»�����ǿ��
��3��DΪFe���������ڱ��е������ڵڢ��壬��KSCN��Һ���������ӣ�
��4�����ݾ�̯�����㾧����Siԭ����Ŀ���ټ��㾧�����������ݦ�=$\frac{m}{V}$���㾧���ܶȣ�
��� �⣺A��B��C��D����Ԫ�����ڱ���ǰ36�ŵ�Ԫ�أ�AԪ�ص�һ�ֺ���û�����ӣ���AΪHԪ�أ�B�Ļ�̬ԭ�Ӻ��������7�ֲ�ͬ���˶�״̬����BΪNԪ�أ�CԪ�������ǽ������ϵ����ǣ����ĵ��ʿ�����������оƬ����CΪSi��DԪ���ǵؿ��к����ڶ��Ľ���Ԫ�أ���DΪFe��
��1��NH3����Ϊ�����νṹ����������ˮ��Ӧ����һˮ�ϰ�����ˮ����֮���γ����������������ˮ���Ӷ��Ǽ��Է��ӣ��������ܣ�������������ˮ��
�ʴ�Ϊ�������Σ�ABC��
��2����������֮���γ������SiH4����֮��Ϊ���»���������ȷ��»�����ǿ���ʰ����ķе���ڵ�SiH4��
�ʴ�Ϊ���ߣ���������֮���γ������
��3��DΪFe���������ڱ��е������ڵڢ��壬��KSCN��Һ���������Ӿ��巽����ȡ���������Һ�������Թ��У��μ�1-2��KSCN��Һ������Һ���Ѫ��ɫ�������Һ�к���Fe3+��
�ʴ�Ϊ������ȡ���������Һ�������Թ��У��μ�1-2��KSCN��Һ������Һ���Ѫ��ɫ�������Һ�к���Fe3+��
��4��������Siԭ����ĿΪ4+8��$\frac{1}{8}$+6��$\frac{1}{2}$=8����������Ϊ8��$\frac{28}{{N}_{A}}$g�����ܶ�=8��$\frac{28}{{N}_{A}}$g�£�a cm��3=$\frac{8��28}{{a}^{3}��{N}_{A}}$g/cm3��
�ʴ�Ϊ��$\frac{8��28}{{a}^{3}��{N}_{A}}$��
���� �����Ƕ����ʽṹ�����ʵĿ��飬ע������������γɵ�Ӱ�죬���ճ������Ӽ��鷽�����Ƚϻ�����

| A�� | �������Һ�м���������ϡ���ᣬ���ȼ����ӣ���ȴ���������Һ�м�������Cu��OH��2��û�к�ɫ����������˵������û��ˮ�� | |
| B�� | .����ˮ�Ҵ��м���ŨH2SO4��������170�棬����������ͨ������KMnO4��Һ���Ϻ�ɫ��ȥ�� ��ʹ��Һ��ɫ����������ϩ | |
| C�� | CO2ͨ�뵽���͵�Na2CO3��Һ���о�������������ΪNaHCO3 | |
| D�� | ����Fe2+ʱ������KSCN��Һ������Һ��죬��Fe2+�ѱ����� |
| A�� | �ڳ��³�ѹ�£�1mol�������еķ�����ΪNA | |
| B�� | �ڳ��³�ѹ�£�11.2L�������еķ�����Ϊ0.5NA | |
| C�� | 15.6gNa2O2�����CO2��Ӧʱ��ת�Ƶĵ�����ΪNA | |
| D�� | ��4gNaOH����1Lˮ�У��ɵõ�0.1mol/LNaOH��Һ |
| A�� | 115��Ԫ��λ�����ڱ��ڰ����� | |
| B�� | ��115�ŵ�Ԫ�ص����ֺ��ص�ԭ���У��������������֮��Ϊ174 | |
| C�� | 1��115��Ԫ�ص�ԭ����1��12Cԭ������֮��Ϊ115��12 | |
| D�� | 115��Ԫ�ؿ���+5�� |
| A�� | ��ϩ������7���Ҽ���1���м� | |
| B�� | ��ϩ������3��̼ԭ�Ӷ���sp2�ӻ� | |
| C�� | ��ϩ������3��̼ԭ����ͬһֱ���� | |
| D�� | ��ϩ�����мȴ��ڷǼ��Լ��ִ��ڼ��Լ� |
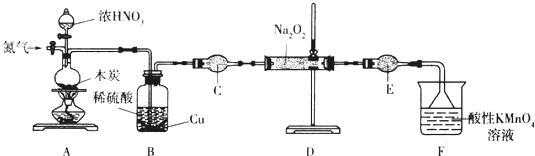
��֪����2NO+Na2O2�T2NaNO2��
��3NaNO2+3HCl�T3NaCl+HNO3+2NO��+H2O��
�����������£�NO��NO2������MnO4-��Ӧ����NO3-��Mn2+��Na2O2��ʹ���Ը��������Һ��ɫ��
��1������װ��Aǰ����ͨһ��ʱ��N2��Ŀ�����ų�װ���еĿ�����
��2��װ��A�з�����Ӧ�Ļ�ѧ����ʽΪC+4HNO3��Ũ��$\frac{\underline{\;\;��\;\;}}{\;}$CO2��+4NO2��+2H2O��ʵ�������Bƿ�е���Һ������Ũ������ȴ�ᾧ����������ƣ������˿ɻ��CuSO4•5H2O��
��3������C������Ϊ����ܣ�����ʢ�ŵ�ҩƷΪ��ʯ�ң������ƣ���
��4����ַ�Ӧ����װ��D�в���ķ����ǣ�ȡ�������������Թ��У�����ϡ������Һ�������ݲ��������Թܿ��Ϸ����ֺ���ɫ���壬�������NaNO2��ע���Լ�������
��5��Ϊ�ⶨ�������Ƶĺ�������ȡ4.000g��Ʒ����ˮ���250mL��Һ��ȡ25.00mL��Һ����ƿ�У���0.1000mol•L-1����KMnO4��Һ���еζ���ʵ�������������±���ʾ��
| ����� | 1 | 2 | 3 | 4 |
| KmnO4��Һ���/mL | 20.60 | 20.02 | 20.00 | 19.98 |
a����ʽ�ζ���������ˮϴ����δ�ñ�Һ��ϴ
b����ƿϴ����δ����
c���ζ��յ�ʱ���Ӷ���
�ڸ��ݱ������ݣ��������ù������������Ƶ���������86.25%��
��6����ƺ���ʵ��Ƚ�0.1mol•L-1NaNO2��Һ��NO2-��ˮ��̶Ⱥ�0.1mol•L-1HNO2��Һ��HNO2�ĵ���̶���Դ�С������Ҫ˵��ʵ�鲽�衢����ͽ��ۣ�������ҩƷ��ѡ��25��C��0.1mol/LHNO2��0.1mol/LNaNO2��Һ�������ϣ����ⶨ��ҺPH��7��˵��HNO2�ĵ���̶ȴ���NO2-���ӵ�ˮ��̶ȣ����ⶨ��ҺpH��7��˵��NO2-����ˮ��̶ȴ���HNO2�ĵ���̶ȣ�
 ij��ѧ��ȤС��Ϊ̽��Ԫ�����ʵĵݱ���ɣ���ƿ�����ϵ��ʵ�飮
ij��ѧ��ȤС��Ϊ̽��Ԫ�����ʵĵݱ���ɣ���ƿ�����ϵ��ʵ�飮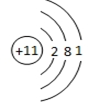 ��
��