��Ŀ����
7����֪E1=134kJ/mol��E2=368kJ/mol����ο�����ͼ������Ҫ����գ�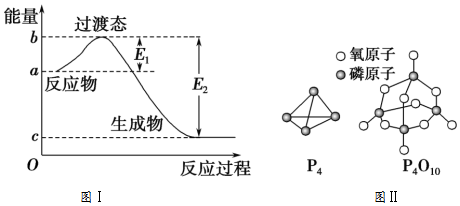
��1��ͼ����1mol NO2��g����1mol CO��g����Ӧ����CO2��g����NO��g�������е������仯ʾ��ͼ��NO2��CO��Ӧ���Ȼ�ѧ����ʽΪ��NO2��g��+CO��g���TNO��g��+CO2��g����H=-234kJ•mol-1��
��2����̼��������Ҫָ����CO2���ڽ������������ŷ��о�����Ҫ�����ã�ĿǰNH3�ͣ�NH4��2CO3�Ѿ���������ҵ��̼����������CO2�ɷ������¿��淴Ӧ��
��Ӧ��2NH3��l��+H2O��l��+CO2��g��?��NH4��2CO3��aq����H1
��Ӧ��NH3��l��+H2O��l��+CO2��g��?NH4HCO3��aq����H2
��Ӧ��NH4��2CO3��aq��+H2O��l��+CO2��g��?2NH4HCO3��aq����H3
���H3���H1����H2֮��Ĺ�ϵ�ǣ���H3=2��H2-��H1��
��3���±���ʾ�Dz��ֻ�ѧ���ļ��ܲ�����
| ��ѧ�� | P-P | P-O | O�TO | P�TO |
| ����/��kJ/mol�� | a | b | c | x |
���� ��1�����������仯ͼ����Ӧ�ȵ�������Ӧ�Ļ�ܼ�ȥ�淴Ӧ�Ļ�ܣ�
��2����2NH3��l��+H2O��l��+CO2��g��?��NH4��2CO3��aq����H1
��NH3��l��+H2O��l��+CO2��g��?NH4HCO3��aq����H2��2����-�ٵã���NH4��2CO3��aq��+H2O��l��+CO2��g��?2NH4HCO3��aq����H3=2��H2-��H1���ݴ˽��з�����
��3������ȼ�յķ���ʽΪP4+5O2=P4O10�����ݻ�ѧ���Ķ��Ѻ��γɵ���Ŀ���м��㣮
��� �⣺��1���÷�Ӧ���ʱ��H=E1-E2=134KJ/mol-368KJ/mol=-234KJ/mol�������Ȼ�ѧ����ʽΪ��NO2��g��+CO��g���TNO��g��+CO2��g����H=-234kJ•mol-1��
�ʴ�Ϊ��NO2��g��+CO��g���TNO��g��+CO2��g����H=-234kJ•mol-1��
��2����2NH3��l��+H2O��l��+CO2��g��?��NH4��2CO3��aq����H1
��NH3��l��+H2O��l��+CO2��g��?NH4HCO3��aq����H2��2����-�ٵã���NH4��2CO3��aq��+H2O��l��+CO2��g��?2NH4HCO3��aq����H3=2��H2-��H1��
�ʴ�Ϊ��2��H2-��H1��
��3������ȼ�յķ���ʽΪP4+5O2=P4O10��1mol������ȫȼ�����6mol P-P��5mol O=O���γ�12molP-O��4mol P=O��
����12mol��bkJ/mol+4mol��xkJ/mol-��6mol��a kJ/mol+5 mol��c kJ/mol��=dkJ/mol��x=$\frac{1}{4}$��6a+5c+d-12b����
�ʴ�Ϊ��$\frac{1}{4}$��6a+5c+d-12b����
���� ���⿼���Ϊ�ۺϣ���Ŀ�Ѷ��еȣ�����ע���Ȼ�ѧ����ʽ����д�����ͼ���Ӧ�ã�ע���ʱ��������ʾۼ�״̬�ı�ע��ѧϰ��Ҫȷ���գ�����ע�ⷴӦ�ȵļ��㣬�ر���ע��������������ķ��ӽṹ����ȷ�жϹ��ۼ������ͺ���Ŀ��

 53���ò�ϵ�д�
53���ò�ϵ�д�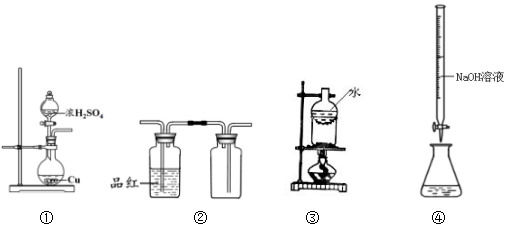
| A�� | ��װ�â��Ʊ�SO2 | B�� | ��װ�âڼ�����ռ�SO2 | ||
| C�� | ��װ�â۷���NaCl��I2 | D�� | ��װ�âܲⶨ��Һ��c��H+�� |
| A�� | CO2�ĵ���ʽΪ | |
| B�� | ŨH2SO4�Լ�ƿ��Ӧ����Σ�վ����ǩ | |
| C�� | ���ݷ�ɢ����ֱ����С���ɽ���ɢϵ��Ϊ��Һ���������Һ | |
| D�� | ���ڴ�8�����ӵ�̼ԭ�ӿɱ�ʾΪ86C |
| A�� | 100g��������Ϊ46%�ľƾ�ˮ��Һ�е���ԭ����Ϊ4NA | |
| B�� | 500mL 2mol/L̼������Һ�е���������С��NA | |
| C�� | �ϳɰ���Ӧ�д��������˷�Ӧ�Ļ�ܣ������˷�Ӧ���ʱ� | |
| D�� | �ݡ���H-T•��S��0���ж�ij��Ӧ���Է�����֪�÷�Ӧһ���ܷ��� |
 ���顢һ����̼���������״��ȼ�����Ҫ��ȼ��Ҳ����Ҫ�Ļ���ԭ�ϣ���֪��
���顢һ����̼���������״��ȼ�����Ҫ��ȼ��Ҳ����Ҫ�Ļ���ԭ�ϣ���֪����2H2��g��+O2��g���T�T2H2O��l����H1=-571.6kJ•mol-1
��CH4��g��+$\frac{1}{2}$O2��g���T�TCO��g��+2H2��g����H2=-36kJ•mol-1
��CH4��g��+H2O��g���T�TCO��g��+3H2��g����H3=+216kJ•mol-1
��1��д�����鲻��ȫȼ������һ����̼��Һ̬ˮ���Ȼ�ѧ����ʽ��CH4��g��+$\frac{3}{2}$O2��g���TCO��g��+2H2O��l����H=-607.6 kJ•mol-1��
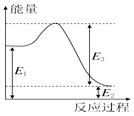
��2�����ڷ�Ӧ����������ת��������ͼ��ʾ�������й�˵������ȷ����C��
A��E1=36kJ
B��E2=36kJ
C��E1-E2=36kJ
D��E3-E1=36kJ
��3����ҵ�Ͽ�ͨ��CO��H2�����Ƶ�CH3OH��CO��g��+2H2��g��?CH3OH��g����H1=a kJ•mol-1��CO�ṹʽΪC��O������֪ijЩ��ѧ���ļ�����ֵ�����
| ��ѧ�� | C-C | C-H | H-H | C-O | C��O | H-O |
| ���� ��kJ•mol-1�� | 348 | 413 | 436 | 358 | 1072 | 463 |
��Q��|a|���ã�������=��գ���
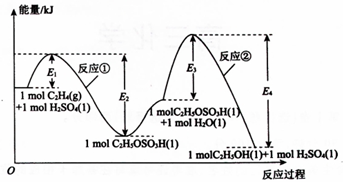 ��ϩ���ˮ�Ϸ��ϳ��Ҵ���������ɣ���Ӧ�����е������仯��ͼ��ʾ���������ܱ������н��еĺϳɷ�Ӧ������˵������ȷ���ǣ�������
��ϩ���ˮ�Ϸ��ϳ��Ҵ���������ɣ���Ӧ�����е������仯��ͼ��ʾ���������ܱ������н��еĺϳɷ�Ӧ������˵������ȷ���ǣ�������| A�� | H2SO4����ϩ���ˮ�Ϸ��ϳ��Ҵ��Ĵ��� | |
| B�� | ��С�����ݻ�����ϩ��ת�������� | |
| C�� | ��Ӧ�ٺͷ�Ӧ�ھ�Ϊ���ȷ�Ӧ | |
| D�� | ��Ӧ�ڵķ�Ӧ�ȡ�H=-��E3-E4��kJ•mol-1 |
| X | |
| Y | Z |
| A�� | �� X ͨ���������Ե��ӿ��γ� X2 ���ӣ�Z �����ڱ��е�λ���ǵ������ڢ��� | |
| B�� | �� X��Y��Z ���ǽ����������ǵ�����������ˮ���ﶼ��ǿ�� | |
| C�� | ������ԭ������������֮��Ϊ 13�������Ǿ�Ϊ�ǽ���Ԫ�� | |
| D�� | �� X��Y��Z ���Ƿǽ������������γɵļ��⻯���У�Z ���⻯��е���� |
| A�� | ��ƽ�����ש�������� | |
| B�� | ҩƷ���治�ƣ�¶��ʱ����� | |
| C�� | ����ʱҺ��������ƿ�ڽ��紦����̶ȾͶ������ | |
| D�� | �ܽⲽ����ɺ�ϴ�Ӵ������� |
| A�� | 67.2 L | B�� | 44.8 L | C�� | 33.6 L | D�� | 22.4 L |