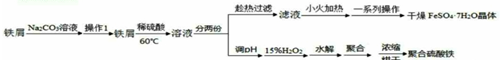
��Ŀ����
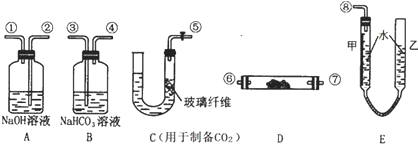
3��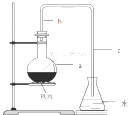 ��ѧʵ��������ͼ��ʾװ����ȡ�����屽������д���пհף�
��ѧʵ��������ͼ��ʾװ����ȡ�����屽������д���пհף���1������ƿa��װ���Լ��DZ��������м������b��������������һ�ǵ��������Ǽ����������������ã�
��2����Ӧ�������ڵ���c���¿ڸ������Թ۲쵽�������֣��������ڷ�Ӧ���ɵ�HBr��ˮ�������γɵģ�
��3����Ӧ��Ϻ�����ƿd�е���AgNO3��Һ�����廯������ɫ�������ɣ�
��4����Ӧ��Ϻ���ƿa�е�Һ�嵹��ʢ����ˮ���ձ�����Թ۲쵽�ձ��ײ��к�ɫ����
��ˮ��Һ�壮�����ܽ�����Ĵ��屽��
��5��д����ƿa�з�����Ӧ�Ļ�ѧ����ʽ��
 +Br2$\stackrel{FeBr_{3}}{��}$
+Br2$\stackrel{FeBr_{3}}{��}$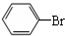 +HBr��
+HBr��
���� ��1���������ӷ�������b�������⣬�����������������ã����ٱ������ӷ���
��2��HBr��������ˮ���ʽ�HBrͨ��C�е�ˮ��ʱ��������ˮ�����ڿ������γɰ�����
��3����������Һ�еμ�AgNO3��Һʱ�ܲ����廯������ɫ������
��4�����屽Ϊ��ɫҺ�壬�ܶȱ�ˮ���屽����δ��Ӧ���壬���ʺ�ɫ��
��5���ڴ����������£������ϵ���ԭ�ӱ���ԭ����ȡ���������屽��ͬʱ���廯�����ɣ�
��� �⣺��1���������ӷ�������b�������⣬�����������������ã����ٱ������ӷ���
�ʴ�Ϊ������������
��2����Ӧ�������ڵ���c���¿ڸ������Թ۲쵽�������֣��������ڷ�Ӧ���ɵ�HBr��HBr��������ˮ��������ˮ�����ڿ������γɰ�������Ӧ�������ڵ���c���¿ڸ������Թ۲쵽�������֣��������ڷ�Ӧ���ɵ�HBr��
�ʴ�Ϊ��HBr��
��3��HBr��������ˮ�����������Һ���μ�AgNO3��Һʱ�ܲ����廯������ɫ������Ag++Br-=AgBr����
�ʴ�Ϊ���廯������ɫ������
��4�����屽Ϊ��ɫҺ�壬�ܶȱ�ˮ���屽����δ��Ӧ���壬���ʺ�ɫ����Ӧ��Ϻ���ƿa�е�Һ�嵹��ʢ����ˮ���ձ�����Թ۲쵽�ձ��ײ��к�ɫ������ˮ��Һ�壮�����ܽ����嵥�ʵ�ԭ��
�ʴ�Ϊ���֣��壻
��5���ڴ����������£������ϵ���ԭ�ӱ���ԭ����ȡ���������屽��ͬʱ���廯�����ɣ���Ӧ����ʽΪ�� +Br2$\stackrel{FeBr_{3}}{��}$
+Br2$\stackrel{FeBr_{3}}{��}$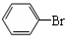 +HBr���ʴ�Ϊ��
+HBr���ʴ�Ϊ�� +Br2$\stackrel{FeBr_{3}}{��}$
+Br2$\stackrel{FeBr_{3}}{��}$ +HBr��
+HBr��
���� �����Ա������ʼ����ӵļ���Ϊ���壬����ѧ����˼ά����������ʵ����������ȣ��Ƕ�֪ʶ���ۺ����ã�����ʵ��ԭ���ǽ��Ĺؼ�����Ŀ�Ѷ��еȣ�

| A�� | 56gFe�����������ᷴӦ��ת�Ƶĵ�����Ϊ2NA | |
| B�� | ��״���£�22.4L��ˮ�к��е�ԭ����Ϊ3NA | |
| C�� | ���ʵ���Ũ��Ϊ2mol/L��BaCl2��Һ�У�����Cl-����Ϊ4NA | |
| D�� | 22.4 L CO��CO2�Ļ��������������̼ԭ����һ����NA |
| A�� |  ����ͼ֪��ʯīת��Ϊ���ʯ�Ƿ��ȷ�Ӧ | |
| B�� |  ����ͼ֪��S��g��+O2��g���TSO2��g����H1��S��s��+O2��g���TSO2��g����H2���H1����H2 | |
| C�� |  ����ͼ֪�����ױȺ����ȶ� | |
| D�� |  ����ͼ֪��CO��g��+H2O��g���TCO2��g��+H2��g����H��0 |
��1�����������������ڴ����еĺ������������ʱ�������������ȣ��漰���·�Ӧ��
��2NO2��g��+NaC1��s��?NaNO3��s��+ClNO��g�� K1
��4NO2��g��+2NaC1��s��?2NaNO3��s��+2NO��g��+Cl2��g�� K2
��2NO��g��+C12��g��?2C1NO��g�� K3
��K1��K2��K3֮��Ĺ�ϵΪK3=$\frac{{{K}^{2}}_{1}}{{K}_{2}}$��
��2����֪���ֻ�ѧ���ļ����������±����������ȵĽṹΪCl-N=O����
| ��ѧ�� | N��O | Cl-Cl | Cl-N | N=O |
| ����/kJ��mol-1 | 630 | 243 | a | 607 |
��3����1L�ĺ����ܱ������г���2molNO��g����1molC12��g�����ڲ�ͬ�¶��²��c��C1NO����ʱ��Ĺ�ϵ��ͼA��
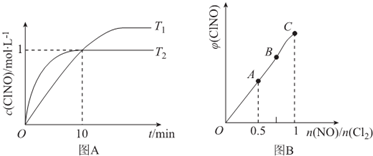
����ͼA���ж�T1��T2���÷�Ӧ�ġ�H��0 ���������������=������
�ڷ�Ӧ��ʼ��10minʱNO��ƽ����Ӧ����v��NO��=0.1mol/��L•min����
��T2ʱ�÷�Ӧ��ƽ�ⳣ��K=2��
��4��һ���������ں��º��ݵ��ܱ������а�һ����������NO��g����Cl2��g����ƽ��ʱClNO�����������$\frac{n��NO��}{n��C{1}_{2}��}$�ı仯ͼ����ͼB����A��B��C��״̬�У�NO��ת����������A�㣮
 ��úΪԭ�ϣ�������ѧ�ӹ�ʹúת��Ϊ���塢Һ�塢����ȼ���Լ����ֻ�����Ʒ�Ĺ�ҵ��ú������
��úΪԭ�ϣ�������ѧ�ӹ�ʹúת��Ϊ���塢Һ�塢����ȼ���Լ����ֻ�����Ʒ�Ĺ�ҵ��ú��������1����ˮ����ͨ�����ȵ�̼���ɲ���ˮú������ӦΪ��C��s��+H2O��g��?CO��g��+H2��g����H=+131.3kJ/mol
�ٸ÷�Ӧ�ڸ��������Է����е�ԭ���ǡ�S��0
��һ���¶��£���һ���ݻ��ɱ���ܱ������У�����������Ӧ���������жϸ÷�Ӧ�ﵽ��ѧƽ��״̬����BDF������ĸ����ͬ����
A�������е�ѹǿ���� B��1mol H-H �����ѵ�ͬʱ����2mol H-O ��
C��V ��CO��=V ��H2��D���ܱ��������ݻ����ٸı�
E������1mol ˮ��ͬʱ����1mol H2 F���������ܶȲ���
�۸÷�Ӧ��ƽ�ⳣ���ı���ʽK=$\frac{c��CO��c��{H}_{2}��}{c��{H}_{2}O��}$��
��2��ˮú���ٽ�һ����Ӧ����ȡ��������ӦΪH2O��g��+CO��g��?H2��g��+CO2��g����ij�¶��¸÷�Ӧ���淴Ӧƽ�ⳣ��K=$\frac{4}{9}$�����¶����ڼס��ҡ������������ܱ������У�ֻͶ��H2��g����CO2��g��������ʼŨ�����±���ʾ�������жϲ���ȷ����B��
| ��ʼ Ũ�� | �� | �� | �� |
| c��H2��/mol/L | 0.010 | 0.020 | 0.020 |
| c��CO2��/mol/L | 0.010 | 0.010 | 0.020 |
B��ƽ��ʱ�����кͱ���H2��ת���ʲ���
C��ƽ��ʱ������c��CO2���Ǽ��е�2 ��
D��ƽ��ʱ������CO2��ת���ʴ���60%
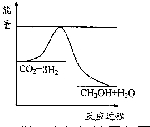
��3��Ŀǰ��ҵ����һ�ַ�������CO2�������״���һ�������·�����Ӧ��CO2��g��+3H2��g��?CH3OH��g��+H2O��g������ͼ��ʾ�÷�Ӧ���й�������������λΪkJ•mol-1���ı仯�������Ϊ1L �ĺ����ܱ������У�����1mol CO2��3mol H2�����д�ʩ����ʹc ��CH3OH����С����A��
A�������¶� ��
B������He��g����ʹ��ϵѹǿ����
C����H2O��g������ϵ�з������
D���ٳ���1mol CO2 ��3mol H2��
| A�� | CuO�ǻ�ԭ���������� | |
| B�� | H2�ǻ�ԭ���������� | |
| C�� | H2O�Ȳ�����������Ҳ���ǻ�ԭ���� | |
| D�� | Cu�ǻ�ԭ�������ԭ |
| A�� | ͭ��Ũ���ᷴӦ��3Cu2+ 8H++2 NO3-�T3Cu2++2NO��+4H2O | |
| B�� | ����ʯ����ᷴӦ��2H++CO32-�TCO2��+H2O | |
| C�� | ��������ϡ���ᷴӦ��2Fe+6H+�T2Fe3++3H2�� | |
| D�� | ����ˮ��Ӧ��2Na+2H2O�T2Na++2OH-+H2�� |