��Ŀ����
17��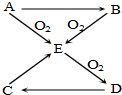 ��֪A��B��C��D��E������������ͼ��ʾ��ת����ϵ�����ַ�Ӧ�P��Ӧ����δ�г���������ʱ��Ҫ�������������裩�������������о�����AԪ�أ�
��֪A��B��C��D��E������������ͼ��ʾ��ת����ϵ�����ַ�Ӧ�P��Ӧ����δ�г���������ʱ��Ҫ�������������裩�������������о�����AԪ�أ���1����AΪ���嵥�ʣ����B��E�Ļ�ѧ����ʽ4NH3+5O2$\frac{\underline{����}}{��}$4NO+6H2O��
�ڱ�״���£�������������D�������ƿ������ˮ���У���ƿ��Һ��������������������ƿ����Һ��ɢ���أ�����������ƿ��������Һ�����ʵ���Ũ��Ϊ0.045 mol/L
��2����AΪ���嵥�ʣ��������ڶ���̼���� ��D�ķ���ʽ ΪSO3��
��C��E�Ļ�ѧ����ʽΪCu+2H2SO4��Ũ�� CuSO4+SO2��+2H2O��
�۽�Eͨ��ijЩ���ʵ���Һ�У��ɷ�����Ӧ��������ǿ�ᣬ�Ծ�һ��д����ѧ����ʽSO2+Cl2+2H2O=2HCl+H2SO4��
���� A��B��C��D��E������AԪ�أ�A����������������Ӧ�õ�D��Aת���õ�B����B��������Ӧ���Եõ�E��˵��A�е�Ԫ���DZ��Ԫ�أ�
��1����AΪ���嵥�ʣ��ƶ�AΪN2����BΪNH3��EΪNO��DΪNO2��CΪHNO3��
��2����AΪ���嵥�ʣ��������ڶ���̼����AΪS����֪BΪH2S��EΪSO2��DΪSO3��CΪH2SO4��
��� �⣺A��B��C��D��E������AԪ�أ�A����������������Ӧ�õ�D��Aת���õ�B����B��������Ӧ���Եõ�E��˵��A�е�Ԫ���DZ��Ԫ�أ�
��1����AΪ���嵥�ʣ��ƶ�AΪN2����BΪNH3��EΪNO��DΪNO2��CΪHNO3��
��B��E�Ļ�ѧ����ʽ��4NH3+5O2$\frac{\underline{����}}{��}$4NO+6H2O��
�ʴ�Ϊ��4NH3+5O2$\frac{\underline{����}}{��}$4NO+6H2O��
�ڷ�����Ӧ��3NO2+H2O=2HNO3+NO��3�������������Ӧ����1���NO����Һ���Ϊ2��������������Ϊ3mol������������Ϊ2mol����Һ���Ϊ2L��22.4L/mol����������ƿ��������Һ�����ʵ���Ũ��Ϊ$\frac{2mol}{2mol��22.4L/mol}$=0.045mol/L��
�ʴ�Ϊ��0.045 mol/L��
��2����AΪ���嵥�ʣ��������ڶ���̼����AΪS����֪BΪH2S��EΪSO2��DΪSO3��CΪH2SO4��
��D�ķ���ʽΪ��SO3���ʴ�Ϊ��SO3��
��C��E�Ļ�ѧ����ʽΪ��Cu+2H2SO4��Ũ�� CuSO4+SO2��+2H2O�ȣ�
�ʴ�Ϊ��Cu+2H2SO4��Ũ�� CuSO4+SO2��+2H2O��
�۽�Eͨ��ijЩ���ʵ���Һ�У��ɷ�����Ӧ��������ǿ�ᣬ�Ծ�һ��д����ѧ����ʽΪ��SO2+Cl2+2H2O=2HCl+H2SO4��
�ʴ�Ϊ��SO2+Cl2+2H2O=2HCl+H2SO4��
���� ���⿼���������ʵ��ƶϣ��������ڿ�������Ŀ�����ؿ�����Ԫ�ص��ʼ��仯�������ʣ���Ҫѧ����������Ԫ�ػ��������ʣ��Ѷ��еȣ�

| A�� | 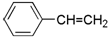 | B�� | 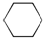 | C�� | 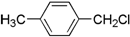 | D�� |  |
| A�� | Na2O2+2H2O�T2Na++2OH-+H2�� | B�� | 2O22-+4H+�T4OH-+O2�� | ||
| C�� | Na2O2+2H2O�T2Na++2OH-+O2�� | D�� | 2Na2O2+2H2O�T4Na++4OH-+O2�� |
 ����β����ȼúβ��������ҳ�����Ⱦ������ɿ�����Ⱦ����Ҫԭ��
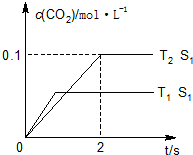
����β����ȼúβ��������ҳ�����Ⱦ������ɿ�����Ⱦ����Ҫԭ����1������β����������Ҫԭ��Ϊ��2NO��g��+2CO��g��$\stackrel{����}{?}$2CO2��g��+N2��g�������ܱ������з����÷�Ӧʱ��c��CO2�����¶ȣ�T���������ı������S����ʱ�䣨t���ı仯���ߣ���ͼ��ʾ���ݴ��жϣ�
�ٸ÷�Ӧ�ġ�H��0�����������������
����T2�¶��£�0��2s�ڵ�ƽ����Ӧ����v��N2��=0.025mol/��L•s����
�۵��������������һ��ʱ�����������������ѧ��Ӧ���ʣ��������ı����S1��S2������ͼ�л���c��CO2����T1��S2�����´ﵽƽ������еı仯���ߣ�
�����÷�Ӧ�ھ��ȡ����ݵ��ܱ���ϵ�н��У�����ʾ��ͼ��ȷ����˵����Ӧ�ڽ��е�t1ʱ�̴ﵽƽ��״̬����BD������ţ���
��2��ֱ���ŷ�úȼ�ղ������������������صĻ������⣮úȼ�ղ����������������������CH4����ԭNOx�������������������Ⱦ�����磺
CH4��g��+2NO2��g���TN2��g��+CO2��g��+2H2O��g����H1=-867kJ/mol
2NO2��g���TN2O4��g����H=-56.9kJ/mol
д��CH4��g������ԭN2O4��g������N2��g����H2O��g�����Ȼ�ѧ����ʽCH4��g��+N2O4��g���TN2��g��+2H2O��l��+CO2��g����H=-898.1kJ/mol��
��3����֪��Ӧ��CO2��g��+H2��g��?CO��g��+H2O��g�����ֽ���ͬ����CO2��g����H2��g���ֱ�ͨ�뵽�ݻ�Ϊ2L�ĺ����ܱ������н��з�Ӧ���õ������������ݣ�
| ʵ���� | �¶�/�� | ��ʼ��/mol | ƽ����/mol | �ﵽƽ�� ����ʱ��/min | ||
| CO2��g�� | H2��g�� | H2O��g�� | CO2��g�� | |||
| 1 | 650 | 2 | 1 | 0.4 | 1.6 | 6 |
| 2 | 900 | 4 | 2 | 1.6 | 2.4 | 2 |
�ڸ÷�Ӧ�ġ�H��0���������������
| A�� | ��ϩ����Ȳ | B�� | 1-��ϩ���ױ� | C�� | ���������� | D�� | ���� ��Ȳ |
| A�� | С�մ�NaHCO3 | B�� | �ռ�NaOH | C�� | �̷�FeSO4•7H2O | D�� | ����BaCO3 |
| A�� | ÿת��0.1mol���ӣ�����������20.7g Pb | |
| B�� | ����ʱ��������������ϵ�����������ƣ�ָ�벻ƫת | |
| C�� | ������Ӧʽ��Pb2++2e-�TPb | |
| D�� | �ŵ�����У�Li+���ƶ� |
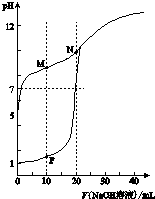 ��0.1mol•L-1 NaOH��Һ�ֱ�ζ������Ϊ20mL��Ũ�Ⱦ�Ϊ0.1mol•L-1 HCl��Һ��HX��Һ����Һ��pH�����NaOH��Һ����仯��ͼ��
��0.1mol•L-1 NaOH��Һ�ֱ�ζ������Ϊ20mL��Ũ�Ⱦ�Ϊ0.1mol•L-1 HCl��Һ��HX��Һ����Һ��pH�����NaOH��Һ����仯��ͼ��