��Ŀ����
10����ͼ��ʾ��������Ϊ��Һ��pH��������ΪZn2+��[Zn��OH��4]2-�����ʵ���Ũ�ȵĶ�����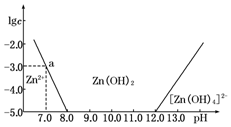
�ش��������⣺
��1����ZnCl2��Һ�м�������������������Һ����Ӧ�����ӷ���ʽ�ɱ�ʾΪZn2++4OH-�T[Zn��OH��4]2-��
��2����ͼ�����ݼ���ɵ�Zn��OH��2���ܶȻ�Ksp=1.00��10-17��
��3��ij��Һ�к�Zn2+��Ϊ��ȡZn2+���Կ�����Һ��pH�ķ�Χ��8.0��pH��12.0��
ij��������ˮ���Ȼ�þ�ʹ�ʯ����ȡ��������þ�������������������ʣ�ͨ���������̽����ᴿ���ƣ������ȼ��������þ��
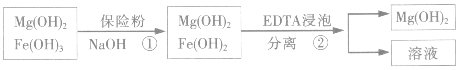
��4��������м��뱣�շۣ�Na2S2O4�������ã�������������ԭΪ����������
��5����֪EDTAֻ������Һ�е�Fe2+��Ӧ����������ˮ�����ʣ�����Mg��OH��2��Ӧ����ȻFe��OH��2������ˮ���������������EDTA�ļ��룬�����ܹ���Fe��OH��2��ȥ����ô��ȸߵ�Mg��OH��2����ӳ����ܽ�ƽ��ĽǶȼ��Խ���Fe��OH��2����Һ�д�������ƽ�⣺Fe��OH��2��s��=Fe2+��aq��+2OH-��aq�������ϵ���EDTAʱ��EDTA�����Fe 2+��ʹƽ�������ƶ���ʹFe��OH��2�����ܽ⣻��
����Ϊ�о���ͬ�����ᴿ���������Ƶ���ȼ���Ĵ��ȴӶ�ȷ������ᴿ������ij�о�С���ȡ������������4���������Ƶõ���ȼ�����к������IJⶨ����������
| ������ȼ�������� | ��ȼ�������� | |||
| ��� | �ᴿ��ϵ�¶�/�� | ����EDTA����/g | ���뱣�շ�����/g | W��Fe��/��10-4g�� |
| 1 | 40 | 0.05 | 0.05 | 7.63 |
| 2 | 40 | 0.05 | 0.10 | 6.83 |
| 3 | 60 | 0.05 | 0.10 | 6.83 |
| 4 | 60 | 0.10 | 0.10 | 6.51 |
��40���60���EDTA����ΪO.05g ��EDTA����Ϊ0.10g �ݱ��շ�����Ϊ0.05g�ޱ��շ�����Ϊ0.10g
A���٢ۢ�B���ڢܢ�C���٢ܢ�D���ڢ�
���� ��1����ͼ���֪��Һ�ļ��Խ�ǿʱ��п�Ĵ�����ʽΪ[Zn��OH��4]2-�����Ԫ���غ���д���̣�
��2������ͼ�����ݣ�����Ksp=c��Zn2+����c2��OH-�����㣻
��3��Ϊ��ȡZn2+�ɽ�Zn2+ת��ΪZn��OH��2����������ͼ��ȷ����ʱ������Һ��pH��Χ��
��4�����շۣ�Na2S2O4�����л����ԣ����Խ�����������ԭΪ������������
��5��Fe��OH��2����Һ�д�������ƽ�⣺Fe��OH��2��s��=Fe2+��aq��+2OH-��aq�������ϵ���EDTAʱ��EDTA�����Fe 2+��ʹƽ�������ƶ���
��6���۲�ʵ��2��3֪������������ͬʱ���¶Ȳ�ͬ����������ͬ����ȡ����������������þ���ܽ⣬���Բ��õ��½Ϻã�ѡ40�棬�۲�ʵ��3��4֪������������ͬʱ��EDTA����Խ������Խ�ͣ�����ѡȡEDTA����Ϊ0.10g���۲�ʵ��1��2֪������������ͬʱ�����շ�����Խ������Խ�ͣ�����ѡȡ���շ�����Ϊ0.10g���ݴ˴��⣮
��� �⣺��1����ͼ���֪��Һ�ļ��Խ�ǿʱ��п�Ĵ�����ʽΪ[Zn��OH��4]2-�������䷴Ӧ�����ӷ���ʽΪ��Zn2++4OH-�T[Zn��OH��4]2-��
�ʴ�Ϊ��Zn2++4OH-�T[Zn��OH��4]2-��
��2������Һ��pH=7.0ʱ��c��Zn2+��=10-3mol•L-1��Ksp=c��Zn2+����c2��OH-��=10-3•��10-7��2=10-17��
�ʴ�Ϊ��1.00��10-17��
��3������ͼ��֪����Һ��pH��ΧΪ8��pH��12ʱ��Zn2+�ɽ�Zn2+ת��ΪZn��OH��2��������Һ��п����Ũ��С��10-5 mol•L-1�����Խ�����Ϊп���Ӳ����ڣ�����ij��Һ�к�Zn2+���ӣ�Ϊ��ȡZn2+���ӿ��Կ�����Һ��pHֵ�ķ�Χ��8.0��pH��12.0��
�ʴ�Ϊ��8.0��pH��12.0��
��4�����շۣ�Na2S2O4�����Խ�����������ԭΪ���������������������ӷ���ʽΪ��6Fe��OH��3+S2O42-+2OH-=6Fe��OH��2+2SO42-+4H2O��
�ʴ�Ϊ��������������ԭΪ������������
��5��Fe��OH��2����Һ�д�������ƽ�⣺Fe��OH��2��s��=Fe2+��aq��+2OH-��aq�������ϵ���EDTAʱ��EDTA�����Fe 2+��ʹƽ�������ƶ���ʹFe��OH��2�����ܽ⣬
�ʴ�Ϊ��Fe��OH��2����Һ�д�������ƽ�⣺Fe��OH��2��s��=Fe2+��aq��+2OH-��aq�������ϵ���EDTAʱ��EDTA�����Fe 2+��ʹƽ�������ƶ���ʹFe��OH��2�����ܽ⣻
��6���۲�ʵ��2��3֪������������ͬʱ���¶Ȳ�ͬ����������ͬ����ȡ����������������þ���ܽ⣬���Բ��õ��½Ϻã�ѡ40�棬�۲�ʵ��3��4֪������������ͬʱ��EDTA����Խ������Խ�ͣ�����ѡȡEDTA����Ϊ0.10g���۲�ʵ��1��2֪������������ͬʱ�����շ�����Խ������Խ�ͣ�����ѡȡ���շ�����Ϊ0.10g
��ѡC��
���� ���⿼�������ܵ���ʵ��ܽ�ƽ�⣬ע���ͼ��ͱ����е����ݵķ����������Ʊ�ʵ�鷽����������ɵļ���Ӧ�ã�������ԭ��Ӧ�ĵ���ת�Ƽ��㣬þ�������仯�������ʵķ����жϣ���Ŀ�Ѷ��еȣ�

| A�� | ��ͬ���������һ����ͬ | B�� | ��ԭ�Ӹ���һ����ͬ | ||
| C�� | ���Ӹ���һ����ͬ | D�� | ��������22��9 |
| ��� | �� | �� | �� | �� |
| ʵ�� |  |  |  |  |
��2��ʵ����Ŀ������֤Cl-��Cu2+��������Ӧ��
��3��ʵ��������֤������Һ�е�H+��OH-��������֮�䷢���˻�ѧ��Ӧ��
��4��ʵ����з�����Ӧ�����ӷ���ʽ��CaCO3+2H+�TCa2++CO2��+H2O��
CH3OH ��l��+$\frac{3}{2}$ O2��g��=CO2��g��+2H2O��l����H=-726.5KJ/mol��
��2���ӻ�ѧ���ĽǶȷ�������ѧ��Ӧ�Ĺ��̾��Ƿ�Ӧ��Ļ�ѧ�����ƻ���������Ļ�ѧ�����γɹ��̣���֪��Ӧ��N2��g��+3H2��g��?2NH3��g����H=a kJ•mol-1���йؼ������������
| ��ѧ�� | H-H | N-H | N��N |
| ���ܣ�kJ•mol-1�� | 436 | 391 | 945 |
��3�����ݸ�˹���ɿ��Զ�ijЩ����ͨ��ʵ��ֱ�Ӳⶨ�Ļ�ѧ��Ӧ�ķ�Ӧ�Ƚ������㣮��֪��
C��s��ʯī��+O2��g���TCO2��g����H1=-393.5kJ•mol-1
2H2��g��+O2��g���T2H2O��l����H2=-571.6kJ•mol-1
2C2H2��g��+5O2��g���T4CO2��g��+2H2O��l����H3=-2599kJ•mol-1
���ݸ�˹���ɣ�����298Kʱ��C��s��ʯī����H2��g������1mol C2H2��g����Ӧ�ķ�Ӧ��Ϊ��
��H=+226.7 KJ/mol��
��4�����������õ������£�NH4+����������Ӧ��������NO3-��������Ӧ�������仯ʾ
��ͼ��ͼ��

��һ����Ӧ�Ƿ��ȷ�Ӧ������ȡ������ȡ�����ԭ���Ƿ�Ӧ��������������������������
| A�� | ���� | B�� | �춡�� | C�� | ������ | D�� | 2��3-�������� |
| A�� | �ų�248 kJ������ | B�� | ����248 kJ������ | ||
| C�� | �ų�183 kJ������ | D�� | ����183 kJ������ |
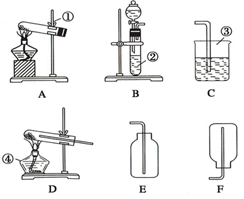 ����A��B��C��D��E��F����װ�ã���ͼ��ʾ�������������ܿ�����ѡ�ã�
����A��B��C��D��E��F����װ�ã���ͼ��ʾ�������������ܿ�����ѡ�ã�