��Ŀ����
5��Ǧ�ֲ��㷺��������ȡ�����ڼӹ���Ϊ����ʹ�õĵ�һ�ֽ�������1��Ǧ��Ԫ�����ڱ���λ��Ϊ�������ڵ�IVA�壮
��2����Ǧ����Ǧ����ʹˮ�����ؽ���Ǧ�ĺ���������������Ⱦ��ˮ��Һ��Ǧ�Ĵ�����̬��Ҫ��Pb2+��Pb��OH��+��Pb��OH��2��Pb��OH��3-��Pb��OH��42-������̬Ǧ��Ũ�ȷ���x����ҺpH�仯�Ĺ�ϵ��ͼ1��ʾ��
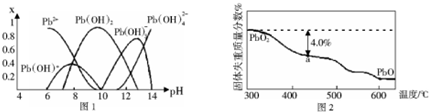
����Pb2+����Һ����εμ�NaOH����Һ����ǣ������μ�NaOH��Һ�ֱ���壻pH��13ʱ����Һ�з�������Ҫ��Ӧ�����ӷ���ʽΪPb��OH��3-+HO-=Pb��OH��42-
�ڿ���С�鷢��һ�������Լ�DH��s��ȥ��Pb2+��Ч����ã��ɳ�ȥˮ�еĺ���Ǧ�������������ӣ���DH��s������Ǧ�����з�������Ҫ��ӦΪ2DH��s��+Pb2+?D2Pb��s��+2H+������Ǧʱ����ʵ�pHԼΪ6��
��3��PbO2����PbO�����������Һ��Ӧ�Ƶã���Ӧ�����ӷ���ʽΪPbO+ClO-=PbO2+Cl-��
��4��PbO2Ҳ����ͨ��ʯī�缫���Pb��NO3��2��Cu��NO3��2����Һ��ȡ�������ĵ缫��ӦʽΪPb2++2H2O-2e-=PbO2��+4H+�������Ϲ۲쵽��������ʯī�Ϻ�ɫ���������������Һ�в�����Cu��NO3��2�����������ĵ缫��ӦʽΪPb2++2e-=Pb��������������Ҫȱ����Pb2+�������ʽ��ͣ�
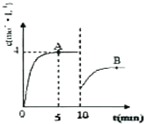
��5��Pb02�ڼ��ȹ��̷����ֽ��ʧ��������ͼ2��ʾ����֪ʧ�������ϵ�a��Ϊ��Ʒʧ��4.0%���� $\stackrel{��Ʒ��ʼ����-a���������}{\overline{��Ʒ��ʼ����}}$��100%���IJ������壬��a�������ɿɱ�ʾΪmPbO2•nPbO����m��n=2��3��������������ȣ���
���� ��1��Ǧ��̼ͬһ���壬�����ڱ���λ�ڵ������ڣ�
��2����pH��13ʱ��Pb��OH��3-Ũ�ȼ�С��Pb��OH��42-Ũ������Pb��OH��3-�����������ӽ��ΪPb��OH��42-��
�ڲμӷ�Ӧ����Pb2+����ͼ���֪��ѡ��PHҪʹǦȫ����Pb2+��ʽ���ڣ�
��3��PbO�ʹ������Ʒ�Ӧ���ɶ�����Ǧ���Ȼ��ƣ�
��4��������Pb2+ʧȥ���ӣ��õ�PbO2����Ҫˮ���뷴Ӧ��ͬʱ���������ӣ������ϣ�Cu2+��õ�������Cu��������ʯī�缫�ϣ�
�����Һ�в�����Cu��NO3��2��������Pb2+��õ�������Pb��Pb2+����PbO2�������ʽ��ͣ�
��5����a�������ɱ�ʾΪPbOx������PbO2$\frac{\underline{\;\;��\;\;}}{\;}$PbOx+$\frac{2-x}{2}$O2���з��̼���x��ֵ������ɱ�ʾΪmPbO2•nPbO����Oԭ����Pbԭ�ӵı�ֵΪx���ݴ˼�����
��� �⣺��1��Ǧ��̼ͬһ���壬λ�ڵ�IVA�壬�����ڱ���λ�ڵ������ڣ�
�ʴ�Ϊ���������ڵ�IVA�壻
��2����pH��13ʱ��Pb��OH��3-Ũ�ȼ�С��Pb��OH��42-Ũ������Pb��OH��3-�����������ӽ��ΪPb��OH��42-����Ӧ���ӷ���ʽΪ��Pb��OH��3-+HO-=Pb��OH��42-��
�ʴ�Ϊ��Pb��OH��3-+HO-=Pb��OH��42-��
�ڷ�ӦΪ2EH��s��+Pb2+?E2Pb��s��+2H+���μӷ�Ӧ����Pb2+����ͼ���֪��ѡ��PHҪʹǦȫ����Pb2+��ʽ���ڣ�����Ǧʱ����ʵ�pHԼ6��
�ʴ�Ϊ��6��
��3��PbO�ʹ������Ʒ�Ӧ���ɶ�����Ǧ���Ȼ��ƣ����ӷ���ʽΪPbO+ClO-=PbO2+Cl-��
�ʴ�Ϊ��PbO+ClO-=PbO2+Cl-��
��4��������Pb2+ʧȥ���ӣ��õ�PbO2����Ҫˮ���뷴Ӧ��ͬʱ���������ӣ��缫��ӦʽΪ��Pb2++2H2O-2e-=PbO2��+4H+�������ϣ�Cu2+��õ�������Cu��������ʯī�缫�ϣ���ʯī�Ϻ�ɫ����������
�����Һ�в�����Cu��NO3��2��������Pb2+��õ�������Pb�������缫��ӦʽΪ��Pb2++2e-=Pb����Pb2+�������ʽ��ͣ�
�ʴ�Ϊ��Pb2++2H2O-2e-=PbO2��+4H+��ʯī�Ϻ�ɫ����������Pb2++2e-=Pb����Pb2+�������ʽ��ͣ�
��5����a�������ɱ�ʾΪPbOx������PbO2$\frac{\underline{\;\;��\;\;}}{\;}$PbOx+$\frac{2-x}{2}$O2���У�$\frac{2-x}{2}$��32=239��4.0%�����x=1.4��
����ɱ�ʾΪmPbO2•nPbO����Oԭ����Pbԭ�ӵı�ֵΪx����$\frac{2m+n}{m+n}$=1.4��������m��n=2��3��
�ʴ�Ϊ��2��3��
���� ���⿼��̼��Ԫ�����ʣ���Ŀ�Ѷ��еȣ��漰���ʵ������йؼ��㡢���ӷ�Ӧ�����ԭ����֪ʶ�㣬ע�⣨4���������������缫��Ӧʽ����д��ע�⣨5���еļ��㣬Ϊ�״��㣬�����ڿ���ѧ���ķ��������ͼ���������

 ��ҵ����ϵ�д�
��ҵ����ϵ�д�| A�� | �Դ��ʹ�������ǽ��еġ���������������������������Ӧ����ԭ���ԭ�� | |
| B�� | �Ȼ������۵���������ͣ���˹�ҵ����ò��õ�������Ȼ������Ʊ������� | |
| C�� | ����ұ�����ơ��ơ�þ�������������õĽ�������ⷨ������Ψһ���еĹ�ҵ���� | |
| D�� | ���ʱ��ͨ���Ѵ��ƵĽ�����Ʒ���������ѶƲ���������� |
| A�� | ����ʱ��Ӧ���������е���Һȫ���������ɺ�ֹͣ���� | |
| B�� | ����ʵ�鿪ʼʱ��ͨ������ˮ���ٵ�ȼ�ƾ��Ƽ��� | |
| C�� | ��ȡʱ��Ӧѡ���л���ȡ��������ȡ�����ܶȱ����ˮ���ܶ�С | |
| D�� | ��Һʱ����Һ©�����²�Һ����¿ڷų����ϲ�Һ��Ҳ���¿����� |
| A�� | ����4.48L | B�� | ��2.24L | C�� | ����2.24L | D�� | ����2.24L |
 ��֪����Ϊ��Ԫ���H2C2O4�THC2O4-+H+Ka 1�� HC2O4-�TC2O42-+H +Ka 2�����£���ijŨ�ȵIJ�����Һ����μ���һ����Ũ�ȵ�KOH��Һ��������Һ�� H2C2O4��HC2O4-��C2O42- �����������ʵ����������ģ�����Һ pH �Ĺ�ϵ��ͼ��ʾ��������˵���в���ȷ���ǣ�������
��֪����Ϊ��Ԫ���H2C2O4�THC2O4-+H+Ka 1�� HC2O4-�TC2O42-+H +Ka 2�����£���ijŨ�ȵIJ�����Һ����μ���һ����Ũ�ȵ�KOH��Һ��������Һ�� H2C2O4��HC2O4-��C2O42- �����������ʵ����������ģ�����Һ pH �Ĺ�ϵ��ͼ��ʾ��������˵���в���ȷ���ǣ�������| A�� | �����£�Ka 1��Ka 2=1000 | |
| B�� | ����ͬ���ʵ��� KHC2O4 �� K2C2O4������ȫ����ˮ����� pH Ϊ 4.2 �Ļ��Һ | |
| C�� | pH=1.2 ��Һ�У�c��K +��+c��H + ��=c��OH-��+c��H2C2O4�� | |
| D�� | �� pH=1.2 ����Һ�м� KOH ��Һ��pH������4.2�Ĺ�����ˮ�ĵ����һֱ���� |
 ��1913�깤ҵ�ϳɰ�Ͷ���������ϳɰ���ҵ���Ϸ�չ�����ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ�Ͽɽ���������������ش��������⣺
��1913�깤ҵ�ϳɰ�Ͷ���������ϳɰ���ҵ���Ϸ�չ�����ֿ��Խ�һ���Ʊ����ᣬ�ڹ�ҵ�Ͽɽ���������������ش��������⣺��1����֪��N2��g��+O2��g���T2NO��g����H=+180.5kJ/mol
N2��g��+3H2��g��?2NH3��g����H=-92.4kJ/mol
2H2��g��+O2��g���T2H2O��g����H=-483.6kJ/mol
д������������������һ�����������ˮ�������Ȼ�ѧ����ʽ��4NH3��g��+5O2��g���T4NO��g��+6H2O��g����H=-905.0kJ/mol��
��2����һ�������£��ϳ����е�������������ʼŨ�ȷֱ�Ϊa mol•L-1��b mol•L-1����ӦΪ��N2+3H2?2NH3��������Ũ����ʱ��仯��ͼ��ʾ����10minʱ��ȡ�Ĵ�ʩ����ȥ���ְ�������A��ƽ�ⳣ��K=�����������������=����B���ƽ�ⳣ����
��3����һ������ĺ����ܱ������У��������»�ѧ��Ӧ��N2��g��+3H2��g��?2NH3��g�����仯ѧƽ�ⳣ��K���¶�t�Ĺ�ϵ�����
| t/K | 298 | 398 | 498 | �� |
| K | 4.1��106 | K1 | K2 | �� |
���жϸ÷�Ӧ�ﵽ��ѧƽ��״̬��������AC������ţ���
A��2v����H2��������=3v����NH3�� B��v����N2��=3v����H2��
C��������ѹǿ���ֲ��� D�����������ܶȱ��ֲ���
��4����һ���¶Ⱥʹ����£���6.4mol H2��2.4molN2�����һ���ݻ�Ϊ4L���ܱ������з�����Ӧ����3minĩʱ��Ӧǡ�ô�ƽ�⣬��ʱ������1.6mol NH3������������µ�ƽ�ⳣ��Ϊ0.4��L/mol��2��
| A�� | pH��ͬ�Ģ�CH3COONa����NaHCO3������Һ�е�c��Na+�����ڣ��� | |
| B�� | 0.1mol•L-1ij��Ԫ����ǿ����NaHA��Һ�У�c��Na+��=2c��A2-��+c��HA-��+c��H2A�� | |
| C�� | 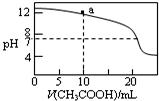 ͼ��ʾ��0.1 mol/L CH3COOH��Һ�ζ�20 mL 0.1mol/L NaOH��Һ�ĵζ����ߣ���pH=7ʱ��c��Na+����c��CH3COO-����c��OH-��=c��H+�� | |
| D�� | ��ͼa����Һ�и�����Ũ�ȵĹ�ϵ�ǣ�c��OH-��=c��H+��+c��CH3COO-��+2c��CH3COOH�� |

| A�� | SO2��NxOy���������������� | |
| B�� | ������ķ����� | |
| C�� | �ؽ������ӿɵ��µ����ʱ��� | |
| D�� | ����β���Ĵ����ŷ������������������Ϊ����֮һ |