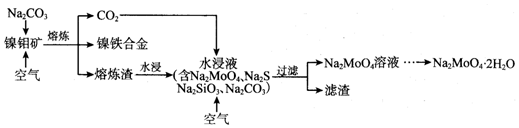
��Ŀ����
6�����з�Ӧ�����ӷ���ʽ��ȷ���ǣ�������| A�� | ����ӡˢ��·�壺Fe3++Cu�TCu2++Fe2+ | |
| B�� | �����ʵ�����Ba��OH��2��NH4HSO4��ϡ��Һ�з�Ӧ��Ba2++2OH-+2H++SO42-�TBaSO4��+2H2O | |
| C�� | Ca��OH��2��Һ������Ca��HCO3��2��Һ��Ӧ��Ca2++2HCO3-+2OH-�T2CaCO3��+2H2O+CO32- | |
| D�� | ���Ʊ�����ˮ�м���ʯ��ʯ�������Һ��HClOŨ�ȣ�CaCO3+2Cl2+H2O�TCa2++2Cl-+CO2��+2H2O |
���� A�����ӷ���ʽ��������ɲ���ȣ�Υ���˵���غ㣻
B���������ʵ������ʱ��笠�����Ҳ���뷴Ӧ��
C��̼�������������Ӧ�����в������̼������ӣ�
D��HCl��̼��Ʒ�Ӧ���ٽ���������ˮ�ķ�Ӧ��������HClO�����ɣ�
��� �⣺A����������ͭ��Ӧ�����������Ӻ�ͭ���ӣ���ȷ�����ӷ���ʽΪ��Fe3++Cu�TCu2++Fe2+����A����
B�������ʵ�����Ba��OH��2��NH4HSO4��ϡ��Һ�з�Ӧ��NH4++Ba2++2OH-+H++SO42-�TBaSO4��+H2O+NH3•H2O����B����
C��Ca��OH��2��Һ������Ca��HCO3��2��Һ��Ӧ����̼��Ƴ�����ˮ����ȷ�����ӷ���ʽΪ��Ca2++HCO3-+OH-�TCaCO3��+H2O+����C����
D�����Ʊ�����ˮ�м���ʯ��ʯ�������Һ��HClOŨ�ȣ���Ӧ�����ӷ���ʽΪ��CaCO3+2Cl2+H2O�TCa2++2Cl-+CO2��+2H2O����D��ȷ��
��ѡD��
���� ���⿼�������ӷ���ʽ����д�жϣ�Ϊ�߿��ĸ�Ƶ�⣬��Ŀ�ѶȲ���ע����ȷ���ӷ���ʽ�����жϳ��÷�������鷴Ӧ��������Ƿ���ȷ���������ʲ���Ƿ���ȷ���������������ʵ���Ҫ������ѧʽ������Ƿ�����غ��ϵ���磺�����غ�͵���غ�ȣ��ȣ�

 53������ϵ�д�
53������ϵ�д�| A�� | �ô����ȥˮ���е�̼��ƣ�CaCO3+2H+�TCa2++H2O+CO2�� | |
| B�� | ����ˮ��Һ�ʼ��Ե�ԭ��S2-+2H2O?H2S��+2OH- | |
| C�� | ��NaHCO3��Һ�м���������Ba��OH��2��Һ��Ba2++OH-+HCO3-�TBaCO3��+2H2O | |
| D�� | ��Na2FeO4��Һ�м���ϡ�������������4FeO42-+20H+�T4Fe3++3O2��+10H2O |
| A�� | Na+��I-��K+��Br2 | B�� | Mg2+��Cu2+��SO42-��NO3- | ||
| C�� | Fe2+��Cl2��K+��SO42- | D�� | ClO-��Cl-��CO2��Na+ |
| A�� | ���0.10mol/LCH3COOH��Һ�д��ڽ϶��CH3COOH���� | |
| B�� | �Ƚϵ�Ũ�ȵ���������ᡢCH3COOH��Һ������Zn��Ӧ����H2����� | |
| C�� | �Ƚϵ�Ũ�ȵ���������ᡢCH3COOH��Һ�����NaOH��Һ��Ӧ��ų������� | |
| D�� | 0.10mol/LNH4Cl�����ԣ�0.1mol/LCH3COONH4��Һ������ |
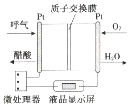
| A�� | �����ɺ������ڵIJ��缫���� | |
| B�� | H+�����ӽ���Ĥ�����������ڵIJ��缫 | |
| C�� | ��·������2mol����ʱ������11.2LO2 | |
| D�� | �õ�صĸ�����ӦΪ��CH3CH2OH+3H2O-12e-�T2CO2��+12H+ |