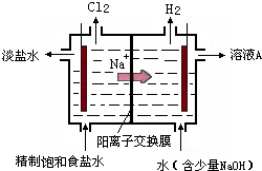
��Ŀ����
��ͼ1ΪԪ�����ڱ���һ���֣�����Ԫ�آ١����ڱ��е�λ�ã����û�ѧ��������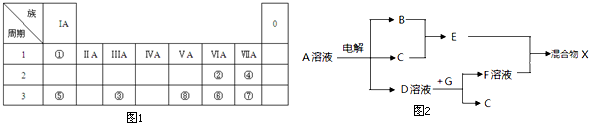
��1���ܡ��ݡ��ߵ����Ӱ뾶�ɴ�С��˳��Ϊ ���������ӷ��ű��
��2���͢ߵ���ۺ����������ǿ��Ϊ �����û�ѧʽ��
��3���ɱ���Ԫ���γɵ����ʿɷ���ͼ2�еķ�Ӧ������B��C��G�ǵ��ʣ�BΪ����ɫ���壬D��Һ�Լ��ԣ�
��д�����A��Һ�Ļ�ѧ����ʽ ����д��D��Һ��G��Ӧ�����ӷ���ʽ ��
��4���飨As���Ļ����������ɱ�漰ҽ�ƣ�
��As��ԭ�ӽṹʾ��ͼΪ �������⻯��Ļ�ѧʽΪ ��
��Y�ɢڢݢ�����Ԫ����ɣ�����ˮ��Һ�������г�������������As����Y��ˮ��Һ��Ӧ��������As����ۺ����ᣬ�÷�Ӧ�Ļ�ѧ����ʽΪ ��

��1���ܡ��ݡ��ߵ����Ӱ뾶�ɴ�С��˳��Ϊ
��2���͢ߵ���ۺ����������ǿ��Ϊ
��3���ɱ���Ԫ���γɵ����ʿɷ���ͼ2�еķ�Ӧ������B��C��G�ǵ��ʣ�BΪ����ɫ���壬D��Һ�Լ��ԣ�
��д�����A��Һ�Ļ�ѧ����ʽ
��4���飨As���Ļ����������ɱ�漰ҽ�ƣ�
��As��ԭ�ӽṹʾ��ͼΪ
��Y�ɢڢݢ�����Ԫ����ɣ�����ˮ��Һ�������г�������������As����Y��ˮ��Һ��Ӧ��������As����ۺ����ᣬ�÷�Ӧ�Ļ�ѧ����ʽΪ
���㣺Ԫ�������ɺ�Ԫ�����ڱ����ۺ�Ӧ��,������ƶ�
ר�⣺Ԫ����������Ԫ�����ڱ�ר��
��������Ԫ�������ڱ���λ�ã���֪��ΪH����ΪO����ΪAl����ΪF����ΪNa����ΪS����ΪCl����ΪP��
��1�����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ��
��2���ǽ�����Խǿ����ۺ����������Խǿ��
��3���ɱ���Ԫ���γɵ����ʣ�����B��C��G�ǵ��ʣ�BΪ����ɫ���壬��BΪCl2��D��Һ�Լ��ԣ����A����Һ�õ�B��C��D������֪AΪNaCl��DΪNaOH��CΪH2����ת����ϵ��֪��EΪHCl��GΪAl��FΪNaAlO2��
��4����Asԭ�Ӻ��������Ϊ33����4�����Ӳ㣬���������Ϊ2��8��18��5��
��As���ڢ�A�壬�����Ϊ-3��������д���⻯��Ļ�ѧʽ��
��Y�ɢڢݢ�����Ԫ����ɣ�����ˮ��Һ�������г���������������YΪNaClO��As����NaClO��ˮ��Һ��Ӧ��������As����ۺ����ᣬ��Ԫ�ر���ԭΪ�����ӣ�
��1�����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ��
��2���ǽ�����Խǿ����ۺ����������Խǿ��
��3���ɱ���Ԫ���γɵ����ʣ�����B��C��G�ǵ��ʣ�BΪ����ɫ���壬��BΪCl2��D��Һ�Լ��ԣ����A����Һ�õ�B��C��D������֪AΪNaCl��DΪNaOH��CΪH2����ת����ϵ��֪��EΪHCl��GΪAl��FΪNaAlO2��
��4����Asԭ�Ӻ��������Ϊ33����4�����Ӳ㣬���������Ϊ2��8��18��5��
��As���ڢ�A�壬�����Ϊ-3��������д���⻯��Ļ�ѧʽ��
��Y�ɢڢݢ�����Ԫ����ɣ�����ˮ��Һ�������г���������������YΪNaClO��As����NaClO��ˮ��Һ��Ӧ��������As����ۺ����ᣬ��Ԫ�ر���ԭΪ�����ӣ�
���
�⣺��Ԫ�������ڱ���λ�ã���֪��ΪH����ΪO����ΪAl����ΪF����ΪNa����ΪS����ΪCl����ΪP��
��1�����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ�����Ӱ뾶��Cl-��F-��Na+���ʴ�Ϊ��Cl-��F-��Na+��
��2���ǽ�����S��Cl���ǽ�����Խǿ����ۺ����������Խǿ��������HClO4��H2SO4���ʴ�Ϊ��HClO4��H2SO4��
��3���ɱ���Ԫ���γɵ����ʣ�����B��C��G�ǵ��ʣ�BΪ����ɫ���壬��BΪCl2��D��Һ�Լ��ԣ����A����Һ�õ�B��C��D������֪AΪNaCl��DΪNaOH��CΪH2����ת����ϵ��֪��EΪHCl��GΪAl��FΪNaAlO2���ٵ��A��Һ�Ļ�ѧ����ʽΪ��2NaCl+2H2O
H2��+Cl2��+2NaOH��
��D��Һ��G��Ӧ�����ӷ���ʽΪ��2Al+2OH-+2H2O=2AlO2-+3H2����
�ʴ�Ϊ��2NaCl+2H2O
H2��+Cl2��+2NaOH��2Al+2OH-+2H2O=2AlO2-+3H2����
��4����Asԭ�Ӻ��������Ϊ33����4�����Ӳ㣬���������Ϊ2��8��18��5��ԭ�ӽṹʾ��ͼΪ��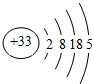 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
��
��As���ڢ�A�壬�����Ϊ-3�����⻯��Ļ�ѧʽΪAsH3���ʴ�Ϊ��AsH3��
��Y�ɢڢݢ�����Ԫ����ɣ�����ˮ��Һ�������г���������������YΪNaClO��As����NaClO��ˮ��Һ��Ӧ��������As����ۺ����ᣬ��Ԫ�ر���ԭΪ�����ӣ���Ӧ����ʽΪ��2As+5NaClO+3H2O=5NaCl+2H3AsO4���ʴ�Ϊ��2As+5NaClO+3H2O=5NaCl+2H3AsO4��
��1�����Ӳ�ṹ��ͬ���˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ�����Ӱ뾶��Cl-��F-��Na+���ʴ�Ϊ��Cl-��F-��Na+��
��2���ǽ�����S��Cl���ǽ�����Խǿ����ۺ����������Խǿ��������HClO4��H2SO4���ʴ�Ϊ��HClO4��H2SO4��
��3���ɱ���Ԫ���γɵ����ʣ�����B��C��G�ǵ��ʣ�BΪ����ɫ���壬��BΪCl2��D��Һ�Լ��ԣ����A����Һ�õ�B��C��D������֪AΪNaCl��DΪNaOH��CΪH2����ת����ϵ��֪��EΪHCl��GΪAl��FΪNaAlO2���ٵ��A��Һ�Ļ�ѧ����ʽΪ��2NaCl+2H2O
| ||
��D��Һ��G��Ӧ�����ӷ���ʽΪ��2Al+2OH-+2H2O=2AlO2-+3H2����
�ʴ�Ϊ��2NaCl+2H2O
| ||
��4����Asԭ�Ӻ��������Ϊ33����4�����Ӳ㣬���������Ϊ2��8��18��5��ԭ�ӽṹʾ��ͼΪ��
 ���ʴ�Ϊ��
���ʴ�Ϊ�� ��
����As���ڢ�A�壬�����Ϊ-3�����⻯��Ļ�ѧʽΪAsH3���ʴ�Ϊ��AsH3��
��Y�ɢڢݢ�����Ԫ����ɣ�����ˮ��Һ�������г���������������YΪNaClO��As����NaClO��ˮ��Һ��Ӧ��������As����ۺ����ᣬ��Ԫ�ر���ԭΪ�����ӣ���Ӧ����ʽΪ��2As+5NaClO+3H2O=5NaCl+2H3AsO4���ʴ�Ϊ��2As+5NaClO+3H2O=5NaCl+2H3AsO4��
���������⿼��Ԫ�����ڱ���Ԫ�������ɡ������ƶϵȣ��Ѷ��еȣ����ضԻ�ѧ����Ŀ��飬��Ҫѧ���������ջ���֪ʶ��

��ϰ��ϵ�д�
 ������ĩ��ϰ��ѵ��ϵ�д�
������ĩ��ϰ��ѵ��ϵ�д� С��ʿ��ĩ����100��ϵ�д�
С��ʿ��ĩ����100��ϵ�д�
�����Ŀ
����������ɵĺϽ�19�ˣ�������11.2������ǡ����ȫ��Ӧ����ô�˺Ͻ�����ǣ�������
| A���ƺ��� | B�������� |
| C��ͭ���� | D�������� |
����˵����ȷ���ǣ�������
| A����⾫��ͭʱ��ͬһʱ���������ܽ�Ĵ�ͭ������������������ͭ�������� |
| B���ڶƼ��ϵ��п��������п��������Ҳ�����ö��Բ��������� |
| C�����ε�س������ǻ�ѧ��ת��Ϊ���ܵĹ��� |
| D�����ε�س��ʱ������ϱ��С�+���ĵ缫Ӧ����ӵ�Դ�ĸ������� |
���������У���ѡװ�ò��������ǣ�������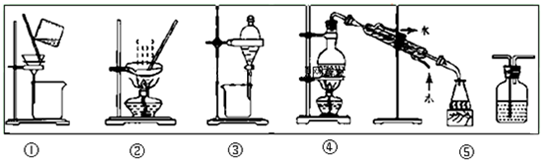

| A�������ᴿ��ѡ�ٺ͢� |
| B����CC14��ȡ��ˮ�еĵ⣬ѡ�� |
| C������Na2CO3��Һ��CH3COOC2H5��ѡ�� |
| D����FeC12��Һ����C12ѡ�� |
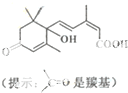 �������˻��ڼ�Դ��������ʻ�ʩ����S-�տ����Ƽ����Ա����ʻ�ʢ����S-�տ��صķ��ӽṹ��ͼ�����й��ڸ÷���˵����ȷ���ǣ�������
�������˻��ڼ�Դ��������ʻ�ʩ����S-�տ����Ƽ����Ա����ʻ�ʢ����S-�տ��صķ��ӽṹ��ͼ�����й��ڸ÷���˵����ȷ���ǣ�������| A��1 mol S-�տ��صķ�������2molNaOH��Ӧ |
| B���ܷ����ӳɡ�ȡ������ȥ��Ӧ |
| C��S-�տ��صķ���ʽΪC14H19O4 |
| D������̼̼˫�����������ǻ����ʻ����Ȼ� |
����ʹ��ˮ��ɫ������ʹ������Һ��ɫ���ǣ�������
| A���� | B����ϩ | C���Ҵ� | D���Ȼ��� |