��Ŀ����
�Իش��������⣺
��1������ʵ������ѡ�õ�����������Լ����۲���������
A����������ƽ����11.7g�Ȼ��ƾ���
B���ü�ʽ�ζ�����ȡ20.00mL Na2CO3��Һ
C������NH4Cl����ʱ����ʪ���ɫʯ����ֽ�����Թܿڣ�����NH3������
D���ⶨ��Һ��pHʱ���ýྻ������IJ�����պȡ��Һ������������ˮʪ�����pH��ֽ�������ɫ���Ƚ�
E����������ˮ��pH��ֽ���Ϳ��Լ���pH��ȵ�H2SO4��CH3COOH��Һ
��2������ֲ�ﺣ���к��д����ĵ�Ԫ�أ���Ԫ���Ե����ӵ���ʽ���ڣ�ʵ������Ӻ�������ȡ���������ͼ��
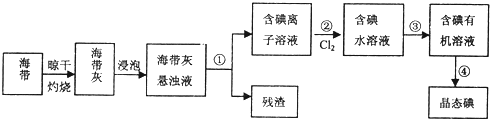
I��ָ����ȡ��Ĺ������йص�ʵ��������ƣ��� ����
����ȡ��Ĺ����У��ɹ�ѡ����л��Լ��� ������ţ�
A���ױ����ƾ�B�����Ȼ�̼����C�����͡�����D�����͡�����
��Ϊʹ��ˮ��Һת��Ϊ����л���Һ��ʵ�������ձ���������������ƿ���ƾ��ơ����ܡ�Բ����ƿ��ʯ�����Լ���Ҫ�ļг���������Ʒ����ȱ�ٵIJ��������� ��
��1������ʵ������ѡ�õ�����������Լ����۲���������
A����������ƽ����11.7g�Ȼ��ƾ���
B���ü�ʽ�ζ�����ȡ20.00mL Na2CO3��Һ
C������NH4Cl����ʱ����ʪ���ɫʯ����ֽ�����Թܿڣ�����NH3������
D���ⶨ��Һ��pHʱ���ýྻ������IJ�����պȡ��Һ������������ˮʪ�����pH��ֽ�������ɫ���Ƚ�
E����������ˮ��pH��ֽ���Ϳ��Լ���pH��ȵ�H2SO4��CH3COOH��Һ
��2������ֲ�ﺣ���к��д����ĵ�Ԫ�أ���Ԫ���Ե����ӵ���ʽ���ڣ�ʵ������Ӻ�������ȡ���������ͼ��

I��ָ����ȡ��Ĺ������йص�ʵ��������ƣ���
����ȡ��Ĺ����У��ɹ�ѡ����л��Լ���
A���ױ����ƾ�B�����Ȼ�̼����C�����͡�����D�����͡�����
��Ϊʹ��ˮ��Һת��Ϊ����л���Һ��ʵ�������ձ���������������ƿ���ƾ��ơ����ܡ�Բ����ƿ��ʯ�����Լ���Ҫ�ļг���������Ʒ����ȱ�ٵIJ���������
���㣺�����ɷ��е�ļ���,�ⶨ��ҺpH�ķ���,���ˡ�������ע����Һ������
ר�⣺
��������1��A��������ƽ�ĸ���Ϊ0.1g��
B����ʽ�ζ����ǿ���ȷ��ȡһ���������Һ��������
C���Ȼ���������ɵİ������Ȼ������Թܿڻ������Ȼ�粒��壻
D��pH��ֽʹ��ʱ����������ˮʪ��
E��pH��ȵ�H2SO4��CH3COOH��Һ�м���������ˮϡ�ͣ�����ϡ�ͺ�pHС���Ǵ��
��2������������Һͨ�����ù��˲��������뻥��Һ��ͨ����������ķ�����
����ȡ�Ļ���ԭ�������ܼ��������ܣ���������һ���ܼ��е��ܽ�ȱ�����һ�ִ�Ķࣻ
�Ӻ�����ȡ������̿�֪�����ݺ������Һ��Ҫ���˳��ӣ��Ժ������ӵ���Һ���������������������ɵⵥ�ʣ�Ȼ����ȡ�õ�����л���Һ���Դ���ѡ��ʵ��������
B����ʽ�ζ����ǿ���ȷ��ȡһ���������Һ��������
C���Ȼ���������ɵİ������Ȼ������Թܿڻ������Ȼ�粒��壻
D��pH��ֽʹ��ʱ����������ˮʪ��
E��pH��ȵ�H2SO4��CH3COOH��Һ�м���������ˮϡ�ͣ�����ϡ�ͺ�pHС���Ǵ��
��2������������Һͨ�����ù��˲��������뻥��Һ��ͨ����������ķ�����
����ȡ�Ļ���ԭ�������ܼ��������ܣ���������һ���ܼ��е��ܽ�ȱ�����һ�ִ�Ķࣻ
�Ӻ�����ȡ������̿�֪�����ݺ������Һ��Ҫ���˳��ӣ��Ժ������ӵ���Һ���������������������ɵⵥ�ʣ�Ȼ����ȡ�õ�����л���Һ���Դ���ѡ��ʵ��������
���
�⣺��1��A��������ƽ�ĸ���Ϊ0.1g������ȷ��
B����ʽ�ζ����ǿ���ȷ��ȡһ���������Һ�������������ü�ʽ�ζ�����ȡ20.00mL Na2CO3��Һ������ȷ��
C���Ȼ���������ɵİ������Ȼ������Թܿڻ������Ȼ�粒��壬������ʪ���ɫʯ����ֽ�����Թܿڣ�����NH3�����ɣ��ʴ���
D��pH��ֽʹ��ʱ����������ˮʪ�ʴ���
E��pH��ȵ�H2SO4��CH3COOH��Һ�м���������ˮϡ�ͣ�����ϡ�ͺ�pHС���Ǵ��ᣬ��������ˮ��pH��ֽ���Ϳ��Լ���pH��ȵ�H2SO4��CH3COOH��Һ������ȷ��
��ѡCD��
��2���������̿�֪�����벻��������Һ�ķ���Ϊ���ˣ���Ϊ�����л��ܼ��е��ܽ�ȱ�ˮ���ҵ���л��ܼ����ڷ��룬��������ȡ��
�ʴ�Ϊ�����ˣ���ȡ��
����ȡ��������ˮ���ܣ��ƾ������ᡢ���;�������������Һ����ȡ�����ʴ�Ϊ��B��
������ת��Ϊ����л���Һ����Ҫ�漰���ա��ܽ���˼���Һ������Ȳ���������ʹ��©�����ձ�������������Һ��Ҫ��Һ©���ȣ�ȱ�ٵ�����Ϊ��Һ©�����ʴ�Ϊ����Һ©����
B����ʽ�ζ����ǿ���ȷ��ȡһ���������Һ�������������ü�ʽ�ζ�����ȡ20.00mL Na2CO3��Һ������ȷ��
C���Ȼ���������ɵİ������Ȼ������Թܿڻ������Ȼ�粒��壬������ʪ���ɫʯ����ֽ�����Թܿڣ�����NH3�����ɣ��ʴ���
D��pH��ֽʹ��ʱ����������ˮʪ�ʴ���
E��pH��ȵ�H2SO4��CH3COOH��Һ�м���������ˮϡ�ͣ�����ϡ�ͺ�pHС���Ǵ��ᣬ��������ˮ��pH��ֽ���Ϳ��Լ���pH��ȵ�H2SO4��CH3COOH��Һ������ȷ��
��ѡCD��
��2���������̿�֪�����벻��������Һ�ķ���Ϊ���ˣ���Ϊ�����л��ܼ��е��ܽ�ȱ�ˮ���ҵ���л��ܼ����ڷ��룬��������ȡ��
�ʴ�Ϊ�����ˣ���ȡ��
����ȡ��������ˮ���ܣ��ƾ������ᡢ���;�������������Һ����ȡ�����ʴ�Ϊ��B��
������ת��Ϊ����л���Һ����Ҫ�漰���ա��ܽ���˼���Һ������Ȳ���������ʹ��©�����ձ�������������Һ��Ҫ��Һ©���ȣ�ȱ�ٵ�����Ϊ��Һ©�����ʴ�Ϊ����Һ©����
���������⿼�����ʵķ�����ᴿ֪ʶ���ǻ�ѧʵ�鷽�������ۣ��ۺ��Խ�ǿ��ѧ����ˡ���ȡ������Ȼ�������ע���ػ���֪ʶ�Ŀ��飬���������Ľṹ��ʹ�÷�������Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
���е��뷽��ʽ������ǣ�������
| A��CaCl2=Ca2++2Cl- |
| B��Ba��OH��2=Ba2++2OH- |
| C��HNO3=H++NO3- |
| D��NaHCO3=Na++H++CO32- |
�����������ֳƳ�������Һ�壨ionic liquid����1914�걨���ĵ�һ������Һ�������һ�李���C2H5NH3��NO3�������۵�Ϊ12�森��֪C2H5NH2������ӵ�������NH3��ǿ�������й������һ�淋�˵����ȷ���ǣ�������
| A��������Һ���к������� |
| B�������һ��ˮ��Һ�ʼ��� |
| C�������һ��ˮ������ӷ���ʽ�ɱ�ʾΪ��C2H5NH3++2H2O?C2H5NH2?H2O+H3O+ |
| D��ͬ�¶�����ͬ���ʵ���Ũ�ȵ������һ����Һ���������Һǰ�ߵ�pHС |
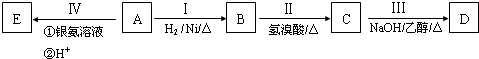
 ���ɵ���F���л������ķ�Ӧ������
���ɵ���F���л������ķ�Ӧ������ ��д����G��Ϊ������һ�����������ɾ����Ļ�ѧ����ʽ��
��д����G��Ϊ������һ�����������ɾ����Ļ�ѧ����ʽ��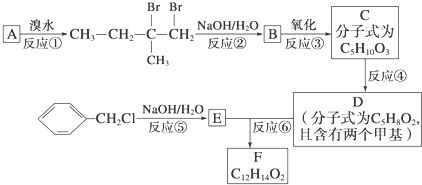
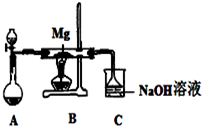 SiO2��SO2��CO2����������������ǵĻ�ѧ������һ���������ԣ�Mg��Na�Ļ�ѧ����Ҳ��һ�������ԣ�ij��ȤС������ͼ��ʾװ�ý���Mg��SO2��Ӧ��ʵ�飮
SiO2��SO2��CO2����������������ǵĻ�ѧ������һ���������ԣ�Mg��Na�Ļ�ѧ����Ҳ��һ�������ԣ�ij��ȤС������ͼ��ʾװ�ý���Mg��SO2��Ӧ��ʵ�飮