��Ŀ����
��80��ʱ����0.20mol�������������������1L�ѳ�յĹ̶��ݻ����ܱ������У���һ��ʱ��Ը������ڵ����ʽ��з������õ��������ݣ�
���ݱ������ݺͱ������ṩ��������գ�C1��C2��C3��A��B��ʾ��Ӧ��Ũ�ȣ���ش����и�С�⣺
��1���÷�Ӧ�Ļ�ѧ����ʽΪ ���ﵽƽ��ʱ������������ת����Ϊ %������C2 C3 A���������������=��
��2��20sʱ��������������Ũ��C1= mol?L-1����0s��20s��������������ƽ����Ӧ����Ϊ ��
| ʱ�䣨s�� Ũ�ȣ�mol?L-1�� | 0 | 20 | 40 | 60 | 80 | 100 |
| c��N2O4�� | 0.20 | C1 | 0.10 | C3 | A | B |
| c��NO2�� | 0.00 | 0.12 | C2 | 0.22 | 0.22 | 0.22 |
��1���÷�Ӧ�Ļ�ѧ����ʽΪ
��2��20sʱ��������������Ũ��C1=
���㣺��ѧƽ�⽨���Ĺ���,��ѧƽ��ļ���
ר�⣺��ѧƽ��ר��
��������1������ͼ�����ݷ�������Ӧ�������������жϣ��ɱ���֪��60sʱ��Ӧ��ƽ�⣬���ݷ���ʽ�����c��N2O4��������ת���ʼ���ƽ��ʱN2O4��ת���ʣ�60s��Ӧ��ƽ�⣬��Ӧ��������ֵ�Ũ�Ȳ��䣻
��2����ѧƽ�������ʽ��ʽ���㣬��ϻ�ѧ��Ӧ���ʸ������0��20s��N2O4��ƽ����Ӧ���ʣ�
��2����ѧƽ�������ʽ��ʽ���㣬��ϻ�ѧ��Ӧ���ʸ������0��20s��N2O4��ƽ����Ӧ���ʣ�
���
�⣺��1���ɱ���֪��60sʱ��Ӧ��ƽ�⣬c��NO2��=0.22mol/L����Ӧ��N2O4Ũ��Ϊ0.11mol/L����Ӧ�Ļ�ѧ����ʽΪ��N2O4?2 NO2��
N2O4?2 NO2��
Ũ�ȱ仯��0.11mol/L 0.22mol/L
����ƽ��ʱN2O4��ת����Ϊ
��100%=55%��
60s��Ӧ��ƽ�⣬��Ӧ��������ֵ�Ũ�Ȳ��䣬����C3=A=B=0.20mol/L-0.11mol/L=0.09mol��40sʱ��
N2O4?2 NO2��
0.2mol/L 0
0.1mol/L 0.2mol/L
0.1mol/L C2=0.2mol/L
C2=0.2mol/L��C3=A=B=0.09mol/L��
�ʴ�Ϊ��N2O4?2NO2��55%������=��
��2�����е�20s��
N2O4 ?2NO2
��ʼ����mol/L�� 0.20 ���������� 0
�仯����mol/L�� 0.06 ����������0.12
20Sĩ��mol/L�� 0.14 ������������ 0.12
20sʱ��N2O4��Ũ��=C1=0.14mol/L��
0��20s��N2O4��ƽ����Ӧ����=
=0.003mol/��L?s����
�ʴ�Ϊ��0.14��0.003mol/��L?s����
N2O4?2 NO2��
Ũ�ȱ仯��0.11mol/L 0.22mol/L
����ƽ��ʱN2O4��ת����Ϊ
| 0.11mol/L |
| 0.20mol/L |
60s��Ӧ��ƽ�⣬��Ӧ��������ֵ�Ũ�Ȳ��䣬����C3=A=B=0.20mol/L-0.11mol/L=0.09mol��40sʱ��
N2O4?2 NO2��
0.2mol/L 0
0.1mol/L 0.2mol/L
0.1mol/L C2=0.2mol/L
C2=0.2mol/L��C3=A=B=0.09mol/L��
�ʴ�Ϊ��N2O4?2NO2��55%������=��
��2�����е�20s��
N2O4 ?2NO2
��ʼ����mol/L�� 0.20 ���������� 0
�仯����mol/L�� 0.06 ����������0.12
20Sĩ��mol/L�� 0.14 ������������ 0.12
20sʱ��N2O4��Ũ��=C1=0.14mol/L��
0��20s��N2O4��ƽ����Ӧ����=
| 0.06mol/L |
| 20s |
�ʴ�Ϊ��0.14��0.003mol/��L?s����
���������⿼���˻�ѧ��Ӧ���ʵļ��㡢��ѧƽ�ⳣ���ĺ����֪ʶ�㣬ע�⻯ѧƽ�ⳣ��ֻ���¶��йأ������ʵ�Ũ���أ�

��ϰ��ϵ�д�
 ��У����ϵ�д�
��У����ϵ�д�
�����Ŀ
���л�ѧ�������д��ȷ���ǣ�������
| A���������ӵĵ���ʽ Cl��Cl |
| B���������ӵĵ���ʽ H��H |
C���Ȼ�þ�ĵ���ʽ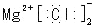 |
D���Ȼ��Ƶĵ���ʽ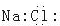 |
���£�1100�棩�����ܱ������з�����Ӧ��Na2SO4��s��+4H2��g��?Na2S��s��+4H2O��g��������˵����ȷ���ǣ�������
A���÷�Ӧ��ƽ�ⳣ������ʽK=
| ||
| B����������������ܶȻ�ѹǿ���ֲ��䣬��˵���÷�Ӧ�Ѵﵽƽ��״̬ | ||
| C����Na2SO4�������ı���ʼ����H2��Ũ�ȣ���ƽ��ʱH2��ת���ʲ��� | ||
| D������ʼʱͶ��2.84gNa2SO4��һ����H2����Ӧ��ƽ��ʱ�����ڹ��干��2.264g����Na2SO4��ת����Ϊ55% |
���º�ѹ�£����ݻ��ɱ���ܱ������з�����Ӧ��A��g��+B��g��?C��g��������ʼʱͨ��1molA��1mol B���ﵽƽ�������a mol C��������˵��������ǣ�������
| A������ʼͨ��3mol A��3mol B���ﵽƽ�������3a mol C | ||
| B������ʼͨ��3mol A��2mol B��1mol C���ﵽƽ����ܱ�������C�����ʵ�������Ϊ3mol | ||
| C������ʼʱͨ��2mol A��2mol B��1mol C���ﵽƽ��ʱ��B�����ʵ���һ��С��2mol | ||
D������ʼʱͨ��2molA��2mol B��1mol C���ﵽƽ�����ͨ��2 mol C�����ٴε���ƽ���C�����ʵ�������Ϊ
|
�����ֱ���һԪ����ɵĻ����1.82g �������Ľ��������ã�����448mL H2����״���£����û��������ǣ�������
| A��CH3OH��CH3CH2CH2OH |
| B��CH3CH2OH��CH3CH��OH��CH3 |
| C��CH3CH2OH��CH3��CH2��3CH2OH |
| D��CH3CH2CH2OH��CH3��CH2��3CH2OH |
 ����ͼװ�ã�X��Y��ֱ����Դ���������ֱ�������и���ʵ�飬���±������и����Ӧ��ϵ����ȷ��һ���ǣ�������
����ͼװ�ã�X��Y��ֱ����Դ���������ֱ�������и���ʵ�飬���±������и����Ӧ��ϵ����ȷ��һ���ǣ�������