��Ŀ����
4��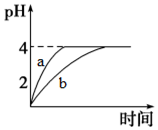 ����ƽ�⡢ˮ��ƽ�⡢�����ܽ�ƽ������Һ�е�����ƽ�⣮��ش��������⣺
����ƽ�⡢ˮ��ƽ�⡢�����ܽ�ƽ������Һ�е�����ƽ�⣮��ش��������⣺��1�������£�ȡpH=2������ʹ�����Һ��100mL�������зֱ����������Zn������Ӧ����������Һ��pH�仯��ͼ��ʾ����ͼ�б�ʾ������Һ��pH�仯���ߵ���b���a����b�������������вμӷ�Ӧ��Zn������Ϊm1��������Һ�вμӷ�Ӧ��Zn������Ϊm2����m1��m2��ѡ���������=����������
��2��25��ʱ����ˮ�ĵ���ƽ����ϵ�м�������̼���ƹ��壬��ˮ�ⷽ��ʽΪCO32-+H2O?HCO3-+OH-��HCO3-+H2O?H2CO3+OH-�������Һ��pH=12������ˮ�������c��OH-��=0.01mol•L-1��
��3�������г����ô����ȥˮ������Ҫ�ɷ�CaCO3�������ƽ��ǶȽ���ԭ��̼��ƴ��ڳ����ܽ�ƽ�⣬CaCO3��s��?Ca2+��aq��+CO32-��aq��������Ϊ������ڵ���ƽ�⣬CH3COOH?CH3COO-+H+�������ӽ��̼������Ӵٽ��̶��ܽ�ƽ��������У�̼����ܽ⣮
��4������ƽ�ⳣ���Ǻ���������ʵ���̶�ǿ��������������֪��
| ��ѧʽ | ���볣����25�棩 |
| HCN | K=4.9��10-10 |
| CH3COOH | K=1.8��10-5 |
| H2CO3 | K1=4.3��10-7��K2=5.6��10-11 |
����NaCN��Һ��ͨ��������CO2��������Ӧ�����ӷ���ʽΪCO2+H2O+CN-=HCO3-+HCN��
��25��ʱ����CH3COOH��CH3COONa�Ļ����Һ�У������pH=6������Һ��c��CH3COO-��-c��Na+��=9.9��10-7 mol•L-1���ȷֵ����
���� ��1���Ӵ�����������ʵĽǶȷ�����������Zn��Ӧͬʱ�������H+��pH�仯�ϻ�����pHֵ�仯��ͬ������μӷ�Ӧ�������ӱ�����ࣻ
��2��̼������Һ����̼�������ˮ�����Һ�Լ��ԣ�����Һ���ε�ˮ��ٽ�ˮ�ĵ��룻
��3��̼��ƴ��ڳ����ܽ�ƽ�⣬����Ϊ������ڵ���ƽ�������
��4����ĵ��볣��Խ������Խǿ���ɵ��볣����֪����CH3COOH��H2CO3��HCN��HCO3-��
����ͬ���ʵ���Ũ�ȵ�������Һ����Ӧ���������Խ�������������ˮ��̶�Խ������Һ��pHԽ��
�۸�������ǿ���ж����������д���ӷ���ʽ��
�ۻ��Һ�еĵ���غ���㣻
��� �⣺��1�����ڴ�����������ʣ���Zn��Ӧͬʱ�������H+������pH�仯�ϻ���������B�����Ǵ�����Һ��pH�仯���ߣ���ͼ֪����ʹ����pH�仯������2��4��������������Ũ����С���������д��ڵ���ƽ�⣬�����Ӻ�п��Ӧʱ�ٽ�������룬���䷴Ӧ�������ӣ����Դ����DZ߷�Ӧ�ߵ���H+�������ĵ�Zn�࣬����m1��m2��
�ʴ�Ϊ��b������
��2��̼������Һ����̼�������ˮ�����Һ�Լ��ԣ�̼���ˮ������ӷ���ʽΪ��CO32-+H2O?HCO3-+OH-��HCO3-+H2O?H2CO3+OH-����pH=12����c��OH-��=0.01mol/L��ȫ����ˮ���������
�ʴ�Ϊ��CO32-+H2O?HCO3-+OH-��HCO3-+H2O?H2CO3+OH-��0.01mol/L��
��3�������г����ô����ȥˮ������Ҫ�ɷ�CaCO3����̼��ƴ��ڳ����ܽ�ƽ�⣬CaCO3��s��?Ca2+��aq��+CO32-��aq��������Ϊ������ڵ���ƽ�⣬CH3COOH?CH3COO-+H+�������ӽ��̼������Ӵٽ��̶��ܽ�ƽ��������У�̼����ܽ⣬
�ʴ�Ϊ��̼��ƴ��ڳ����ܽ�ƽ�⣬CaCO3��s��?Ca2+��aq��+CO32-��aq��������Ϊ������ڵ���ƽ�⣬CH3COOH?CH3COO-+H+�������ӽ��̼������Ӵٽ��̶��ܽ�ƽ��������У�̼����ܽ⣻
��4������ͬ���ʵ���Ũ�ȵ�������Һ����Ӧ���������Խ�������������ˮ��̶�Խ������Һ��pHԽ����˵���̶�CO32-��CN-��HCO3-��CH3COO-����pH��С�����˳��Ϊc��a��b��
�ʴ�Ϊ��c��a��b��
������H2CO3��HCN��HCO3-������NaCN��Һ��ͨ��������CO2��������Ӧ�����ӷ���ʽΪCO2+H2O+CN-=HCO3-+HCN���ʴ�Ϊ��CO2+H2O+CN-=HCO3-+HCN��
��pH=6����c��H+��=10-6mol•L-1��c��OH-��=10-8mol•L-1��
��Һ�ʴ��ڵ���غ㣺c��CH3COO-��+c��OH-��=c��H+��+c��Na+����
��c��CH3COO-��-c��Na+��=c��H+��-c��OH-��=10-6mol•L-1-10-8mol•L-1=9.9��10-7mol•L-1��
�ʴ�Ϊ��9.9��10-7��
���� ���⿼������ˮ�⼰���ԵıȽϡ�pH�����ϡ�͵ȣ�ע��ˮ�������Խ��Խˮ���ϡ����ǿ�ı仯������������ۺ��Խϴ���Ŀ�Ѷ��еȣ�

 ��У����ϵ�д�
��У����ϵ�д�| A�� | 2KMnO4$\frac{\underline{\;\;��\;\;}}{\;}$K2MnO4+MnO2+O2�� | B�� | IBr+H2O�THIO+HBr | ||
| C�� | 2Na2O2+4HCl�T4NaCl+O2+2H2O | D�� | NaH+H2O�TNaOH+H2 |
| A�� | ��������Ȼ��ƿ��ƽ����� | |
| B�� | ����������Ӧ�IJ����뷴Ӧ������ | |
| C�� | ClO2����������ˮ������ | |
| D�� | ���ȼҵ����ָ���ʳ��ˮ��ȡ�����Ȳ�Ʒ�Ĺ�ҵ |
| A�� | W���⻯���X���⻯���ȶ� | |
| B�� | Y�����ڱ��е�λ��Ϊ��3���ڡ�IB�� | |
| C�� | Z������������Ӧ��ˮ����ļ��Ա�Y���� | |
| D�� | X�ĵ����뺬W�����ﲻ���ܷ����û���Ӧ |
| A�� | CuSO4 | B�� | HCl | C�� | BaCl2 | D�� | Na2SO4 |
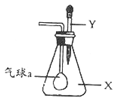 ��ͼ����ƿ��ʢ������X���ι���ʢ��Һ��Y������ѹ��ͷ�ιܵĽ�ͷ��ʹҺ�����ƿ�У������ڲ�����һ�˵�С������������������X��Һ��Y�������ǣ�������
��ͼ����ƿ��ʢ������X���ι���ʢ��Һ��Y������ѹ��ͷ�ιܵĽ�ͷ��ʹҺ�����ƿ�У������ڲ�����һ�˵�С������������������X��Һ��Y�������ǣ�������| A�� | X�ǰ�����Y����������Һ | B�� | X��SO2��Y��Ba��OH��2��Һ | ||
| C�� | X��CO2��Y��������Һ | D�� | X��HC1��Y���Ȼ�����Һ |
| A�� | KAl��SO4��2•12H2O��ˮ������Al��OH��3���壬��������ˮ�� | |
| B�� | SO2��Ư�ס�ɱ�����ܣ�����ʳƷ�ӹ��д���ʹ�� | |
| C�� | MnO2�н�ǿ�������ԣ�����H2O2�ֽ�������� | |
| D�� | Si�ǰ뵼����ϣ��������ƹ��ά |
| ʵ��Ŀ�� | ʵ����� | |
| A | �Ƚ�ˮ���Ҵ��������ǻ�����ԭ�ӵĻ����� | ��������ȡ���״����Ľ����Ʒֱ���ˮ���Ҵ���Ӧ |
| B | �����������ϩ | �ֱ�����ͨ��Ʒ����Һ |
| C | ��ȥ���л��е��������� | ��������������������Һ��������Һ |
| D | ���������ϡ������Ⱥ��Ƿ�ˮ�� | �����������Ƶ�������ͭ����Һ������ |
| A�� | A | B�� | B | C�� | C | D�� | D |