��Ŀ����
Na2S2O3���׳Ʊ��շۣ���ҽҩ��ӡȾ��Ӧ�ù㷺����ͨ�����з����Ʊ���ȡ15.1g Na2SO3����80.0mLˮ����ȡ5.0g��ۣ��������Ҵ���ʪ��ӵ�������Һ�У�С��������У���Ӧ1Сʱ����ˣ���Һ��100�澭������Ũ������ȴ��10�������Na2S2O3?5H2O��
��1�������������Ҵ���ʪ��Ŀ���� ��
��2����Һ�г�Na2S2O3�Ϳ���δ��Ӧ��ȫ��Na2SO3�⣬����ܴ��ڵ��������������� ������ķ����ǣ�ȡ��������Һ�������������Ժ��˳�ȥS���ټ�BaCl2��Һ�����������ᷢ��������Ӧ�Ļ�ѧ����ʽΪ��Na2SO3+2HCl�TSO2��+H2O+2NaCl�� ��
��3��ij�������С���ú�0.100mol?L-1 Na2S2O3��Һ[��������Na2SO3����n��Na2S2O3����n��Na2SO3��
=5��1]�ⶨij������ˮ��Ba2+��Ũ�ȣ�����ȡ��ˮ50.0mL�������ʵ�����ȼ���������K2Cr2O7��Һ����BaCrO4������������ϴ�ӡ����˺���������ϡ�����ܽ⣬��ʱCrO42-ȫ��ת��ΪCr2O72-���ټӹ���KI��Һ����ַ�Ӧ��������Na2S2O3��Һ���еζ�����Ӧ��ȫʱ���������Na2S2O3��Һ�����Ϊ36.0mL��
����֪�йط�Ӧ�����ӷ���ʽΪ����Cr2O72-+6I-+14H+�T2Cr3++3I2+7H2O����I2+2S2O32-�T2I-+S4O62-����I2+SO32-+H2O�T2I-+SO42-+2H+������ζ������п��� ��ָʾ��������ù�����ˮ��Ba2+�����ʵ���Ũ�ȣ�
��1�������������Ҵ���ʪ��Ŀ����
��2����Һ�г�Na2S2O3�Ϳ���δ��Ӧ��ȫ��Na2SO3�⣬����ܴ��ڵ���������������
��3��ij�������С���ú�0.100mol?L-1 Na2S2O3��Һ[��������Na2SO3����n��Na2S2O3����n��Na2SO3��
=5��1]�ⶨij������ˮ��Ba2+��Ũ�ȣ�����ȡ��ˮ50.0mL�������ʵ�����ȼ���������K2Cr2O7��Һ����BaCrO4������������ϴ�ӡ����˺���������ϡ�����ܽ⣬��ʱCrO42-ȫ��ת��ΪCr2O72-���ټӹ���KI��Һ����ַ�Ӧ��������Na2S2O3��Һ���еζ�����Ӧ��ȫʱ���������Na2S2O3��Һ�����Ϊ36.0mL��
����֪�йط�Ӧ�����ӷ���ʽΪ����Cr2O72-+6I-+14H+�T2Cr3++3I2+7H2O����I2+2S2O32-�T2I-+S4O62-����I2+SO32-+H2O�T2I-+SO42-+2H+������ζ������п���
���㣺�Ʊ�ʵ�鷽�������,̽�����ʵ���ɻ�������ʵĺ���
ר�⣺ʵ�������
��������1�����������ˮ�������Ҵ����Ҵ�ʪ�����ʹ������ڷ�ɢ����Һ�У�
��2������S2O32?���л�ԭ�ԣ��ױ�������������������ӿ�֪����Ϊ�����ƣ���������ƺ����ᷴӦ�����Ȼ��ơ��������������ˮ��
��3�����ɵĵⵥ���������۱���������������Ƶζ�ʱ������ɫ��ȥ����Ӳ��仯��˵����Ӧ�ﵽ�յ㣻��Һ�з����ķ�ӦΪ��2Ba2++Cr2O72-=2BaCrO4+2H+�� Cr2O72-+6I-+14H+=2Cr3++3I2+7H2O�� I2+2S2O32-=2I-+S4O62-��I2+SO32-+H2O�T2I-+SO42-+2H+�����ݷ���ʽ�г������ʵĹ�ϵΪ2Ba2+��2BaCrO4��Cr2O72-��3 I2��6S2O32-��2BaCrO4��Cr2O72-��3I2��3SO32-���㣮
��2������S2O32?���л�ԭ�ԣ��ױ�������������������ӿ�֪����Ϊ�����ƣ���������ƺ����ᷴӦ�����Ȼ��ơ��������������ˮ��
��3�����ɵĵⵥ���������۱���������������Ƶζ�ʱ������ɫ��ȥ����Ӳ��仯��˵����Ӧ�ﵽ�յ㣻��Һ�з����ķ�ӦΪ��2Ba2++Cr2O72-=2BaCrO4+2H+�� Cr2O72-+6I-+14H+=2Cr3++3I2+7H2O�� I2+2S2O32-=2I-+S4O62-��I2+SO32-+H2O�T2I-+SO42-+2H+�����ݷ���ʽ�г������ʵĹ�ϵΪ2Ba2+��2BaCrO4��Cr2O72-��3 I2��6S2O32-��2BaCrO4��Cr2O72-��3I2��3SO32-���㣮
���
�⣺��1�����������ˮ�����Ҵ�����������ڷ�Ӧǰ���Ҵ�ʪ����ʹ������ڷ�ɢ����Һ�У���������ۺ�Na2SO3��Һ��ֽӴ����ӿ췴Ӧ����
�ʴ�Ϊ��ʹ������ڷ�ɢ����Һ�У���������ۺ�Na2SO3��Һ��ֽӴ����ӿ췴Ӧ���ʣ�
��2��S2O32?���л�ԭ�ԣ��ܹ���������������������ӣ����Կ��ܴ��ڵ������������ƣ���������ƺ����ᷴӦ�����Ȼ��ơ��������������ˮ����Ӧ�ķ���ʽΪ��Na2S2O3+2HCl=S��+SO2��+H2O+2NaCl���ʴ�Ϊ��Na2SO4�� Na2S2O3+2HCl=S��+SO2��+H2O+2NaCl��
��3�����ݱ궨��ԭ����֪�����ɵĵⵥ���������۱���������������Ƶζ�����ɫ��ȥ����Ӳ��仯��˵����Ӧ�ﵽ�յ㣻��Һ�з����ķ�ӦΪ��2Ba2++Cr2O72-=2BaCrO4+2H+�� Cr2O72-+6I-+14H+=2Cr3++3I2+7H2O�� I2+2S2O32-=2I-+S4O62-��I2+SO32-+H2O�T2I-+SO42-+2H+��
n��Na2S2O3��=0.0360 L��0.100 mol?L-1=0.0036 mol
��n��Na2SO3��=0.00360 mol��5=0.00072 mol
�������⣺2Ba2+��2BaCrO4��Cr2O72-��3I2��6S2O32-
2 6
n1 0.0036 mol
��n1��Ba2+��=
=0.0012 mol
2Ba2+��2BaCrO4��Cr2O72-��3I2��3SO32-
2 3
n2 0.00072mol
�õ�n2��Ba2+��=
=0.00048 mol
��c��Ba2+��=
=3.36��10-2 mol?L-1��
�𣺵��ۣ��ù�����ˮ��Ba2+�����ʵ���Ũ��Ϊ3.36��10-2 mol?L-1��
�ʴ�Ϊ��ʹ������ڷ�ɢ����Һ�У���������ۺ�Na2SO3��Һ��ֽӴ����ӿ췴Ӧ���ʣ�
��2��S2O32?���л�ԭ�ԣ��ܹ���������������������ӣ����Կ��ܴ��ڵ������������ƣ���������ƺ����ᷴӦ�����Ȼ��ơ��������������ˮ����Ӧ�ķ���ʽΪ��Na2S2O3+2HCl=S��+SO2��+H2O+2NaCl���ʴ�Ϊ��Na2SO4�� Na2S2O3+2HCl=S��+SO2��+H2O+2NaCl��
��3�����ݱ궨��ԭ����֪�����ɵĵⵥ���������۱���������������Ƶζ�����ɫ��ȥ����Ӳ��仯��˵����Ӧ�ﵽ�յ㣻��Һ�з����ķ�ӦΪ��2Ba2++Cr2O72-=2BaCrO4+2H+�� Cr2O72-+6I-+14H+=2Cr3++3I2+7H2O�� I2+2S2O32-=2I-+S4O62-��I2+SO32-+H2O�T2I-+SO42-+2H+��
n��Na2S2O3��=0.0360 L��0.100 mol?L-1=0.0036 mol
��n��Na2SO3��=0.00360 mol��5=0.00072 mol
�������⣺2Ba2+��2BaCrO4��Cr2O72-��3I2��6S2O32-
2 6
n1 0.0036 mol
��n1��Ba2+��=
| 0.0036mol��2 |
| 6 |
2Ba2+��2BaCrO4��Cr2O72-��3I2��3SO32-
2 3
n2 0.00072mol
�õ�n2��Ba2+��=
| 0.00072mol��2 |
| 3 |
��c��Ba2+��=
| 0.0012mol+0.00048mol |
| 0.05L |
�𣺵��ۣ��ù�����ˮ��Ba2+�����ʵ���Ũ��Ϊ3.36��10-2 mol?L-1��
���������⿼�����Լ������á����ʵ��ƶϡ��ζ��ļ��㡢����ʽ����д��֪ʶ����3���������ϴ���Ŀ�ѶȽϴ�ע���ϵʽ��Ӧ�ã�

��ϰ��ϵ�д�
 �ƸԹھ��ο���ϵ�д�
�ƸԹھ��ο���ϵ�д�
�����Ŀ
����˵������ȷ���ǣ�������
| A������Cl-���ӵ��Լ�����������Һ��ϡ���� |
| B������SO42-���ӵ��Լ�һ�����Ȼ�����Һ��ϡ���� |
| C������CO32-���ӵ��Լ�������ͳ���ʯ��ˮ |
| D����ij��Һ�м���BaCl2��Һ������ɫ������֤����Һ����SO42- |
 ��H2C03��
��H2C03��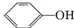 ����
����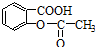 ת��Ϊ
ת��Ϊ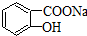 �ķ����У�������
�ķ����У�������