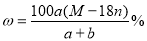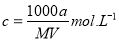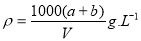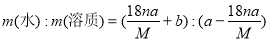��Ŀ����
���������п�ͼ��ϵ��գ���֪��Ӧ�١������ҹ���ҵ�����е���Ҫ��Ӧ��X������Ϊ��ɫ��ζ��Һ�壻C��ɫ��Ӧ����ʻ�ɫ��JΪ���ɫ������D��E������Ϊ���壬��E��ʹƷ����Һ��ɫ��A�����н�������Ԫ�أ����н���Ԫ�ص���������ԼΪ46.7%��
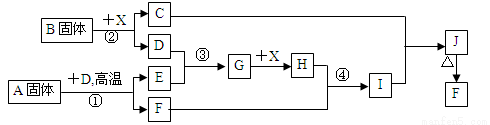
��1��A�Ļ�ѧʽΪ �� F�Ļ�ѧʽΪ ��
��2����Ӧ �۵Ļ�ѧ����ʽ�� ��
��3����Ӧ�ڵ����ӷ���ʽ�� ��
��4����Ӧ�ܵ����ӷ���ʽ�� ��
��5����֪ÿ����16 g E���ų�106.5 kJ��������Ӧ�ٵ��Ȼ�ѧ����ʽΪ��
��
 Сѧ��10���ӿ������100��ϵ�д�
Сѧ��10���ӿ������100��ϵ�д�����ѧ��ѡ��3�����ʽṹ�����ʡ�
����ʯ�������鱦�繫��Ϊ�Ĵ�����ʯ֮һ����Ҫ�ɷ�ΪBe3Al2[Si6O18]����������Cr2O3��0.15��0.6%�������γ���ĸ�̡��Իش��������⣺
��1����̬Alԭ���У�������������ܼ���_______����̬Crԭ�ӵļ۵����Ų�ʽ��_____��
��2���á�����������գ�
��һ������ | ���� | �е� | ���Ӱ뾶 |
Be_____B | C��C_____Si��Si | H2S_____H2O | Al3+_____O2- |
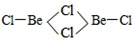
��3��BeCl2���ӵĿռ乹����______�����Ķ�����Be2Cl4�ṹ����ͼ��ʾ������Beԭ�ӵ��ӻ���ʽ��_____��
��4��ǿ��ԭ��LiAlH4�ܽ�SiCl4��ԭ��SiH4����д��SiH4�ڿ�������ȼ�Ļ�ѧ����ʽ______��
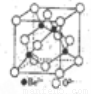
��5��Be������������������ṹ����ͼ��ʾ����֪�����뾧����ܶ�Ϊ��g��cm-3��������Ϊ___cm(��NAΪ�����ӵ�������ֵ���ú�NA���ѵĴ���ʽ��ʾ)��
���������ˮ��Һ��Ϊ˫��ˮ����������������ɱ����Ư����ҵ����ԭ�ϵȡ�
ijʵ��С��ȡһ�����Ĺ���������Һ���ⶨH2O2�ĺ�������̽��H2O2��ijЩ���ʡ�Ӧ�á�
�ⶨ���۹���������Һ��H2O2����������
��1����ȡ10.00mL�ܶ�Ϊ�� g/mL�����۹���������Һ��Ӧѡ��������_______������ţ���
A��10mL��Ͳ B����ʽ�ζ��� C����ʽ�ζ��� D��50mL�ձ�
��2����������Һϡ����250mL���������õ��IJ��������ǣ��ձ�����������_________�������ƣ���
��3��ȡ25.00mL��2����ϡ��Һ����ƿ�У�������ϡ�����ữ����c mol/L KMnO4��Һ�ζ���
����ɷ�Ӧ�����ӷ���ʽ��____MnO4-+___H2O2+___H+��___Mn2++___H2O+___��
�ڸ�С��һ���������Ĵβⶨʵ�飬ʵ������¼���£�
ʵ����� | ��һ�� | �ڶ��� | ������ | ���Ĵ� |
����KMnO4��Һ���/mL | 25.00 | 25.02 | 20.00 | 24.98 |
�����������ݣ�����ԭ����������Һ��H2O2��������____________________________��
��4�����в����ᵼ�²ⶨ���ƫ�ߵ���_____________��
A���ζ�ǰ�ζ��ܼ����������ݣ��ζ���������ʧ
B����H2O2��Һϡ�ͳ�250��00mL��Һʱ�����ݸ��ӿ̶���
C���յ�ʱ������һ��KMnO4��Һ����Һ�ʺ�ɫ�����ɫ��Һ����һ��H2O2��Һ�Գʺ�ɫ
��.̽��H2O2������
��5�������ⶨԭ����H2O2����__________�ԣ�
��6����Ҫ��֤H2O2���ȶ��ԣ�������____________________________��
��.̽��H2O2��Ӧ��
��7��Ϊ�о���ҵ����������˫��ˮ�����������ڵ�pH����Ӧʱ�������ؼ����������Բ�Ʒ������Ӱ�죬��Ҫ��������ʵ�飬��ʵ�����1�Ǹ����̵�����������������������±�������ʵ�����Ϊ5��6��7������ʵ��������
ʵ����� | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
˫��ˮ��mL�� | 0.5 | 0.4 | 0.6 | 0.5 | |||
pH | 11 | 11 | 11 | 10 | |||
��Ӧʱ�� | 3 | 3 | 3 | 3 |


 3NA
3NA