��Ŀ����
��������������ǣ�������
| A����a L 0.1 mol/L ��CH3COOH��Һ��b L 0.1 mol/L�� KOH��Һ��ϣ�������Һ��һ�����ڣ�c ��K+��+c ��H+��=c ��CH3COO-��+c ��OH-�� |
| B����ˮ�У�c��Cl-����c��H+����c��OH-����c��ClO-�� |
| C����0.1 mol/L ��NaHCO3��Һ��0.3 mol/L ��Ba��OH��2��Һ�������ϣ�������Һ��һ�����ڣ�c ��OH-����c ��Ba2+����c ��Na+����c ��H+�� |
| D�������£���pH=3��CH3COOH��Һ��pH=11��NaOH��Һ�У�ˮ�ĵ���̶���ͬ |
���㣺����Ũ�ȴ�С�ıȽ�
ר�⣺����ƽ������Һ��pHר��,�����ˮ��ר��
������A�����ݵ���غ��жϣ�
B��������ˮ��Ӧ��������ʹ����ᣬ����ǿ����ʣ�����Һ����ȫ���룬����������������ʣ�����Һ�в��ֵ��룻
C��0.1 mol/L ��NaHCO3��Һ��0.3 mol/L ��Ba��OH��2��Һ�������ϣ�������Һ�е�����Ϊ0.1mol/L ��Ba��OH��2��0.05mol/L ��NaOH���ݴ��жϣ�
D��������ͼ��ˮ�ĵ����Ӱ���֪����pH=3��CH3COOH��Һ�У�ˮ�ĵ��밴��Һ�����������Ӽ��㣬pH=11��NaOH��Һ�У�ˮ�ĵ��밴�����Ӽ��㣮
B��������ˮ��Ӧ��������ʹ����ᣬ����ǿ����ʣ�����Һ����ȫ���룬����������������ʣ�����Һ�в��ֵ��룻
C��0.1 mol/L ��NaHCO3��Һ��0.3 mol/L ��Ba��OH��2��Һ�������ϣ�������Һ�е�����Ϊ0.1mol/L ��Ba��OH��2��0.05mol/L ��NaOH���ݴ��жϣ�
D��������ͼ��ˮ�ĵ����Ӱ���֪����pH=3��CH3COOH��Һ�У�ˮ�ĵ��밴��Һ�����������Ӽ��㣬pH=11��NaOH��Һ�У�ˮ�ĵ��밴�����Ӽ��㣮
���
�⣺A��CH3COOH��Һ�� KOH��Һ��ϣ����ݵ���غ��֪c ��K+��+c ��H+��=c ��CH3COO-��+c ��OH-������A��ȷ��
B��������ˮ��Ӧ��������ʹ����ᣬ����ǿ����ʣ�����Һ����ȫ���룬����������������ʣ�����Һ�в��ֵ��룬��������Һ����c��H+����c��Cl-����c��ClO-����c��OH-������B����
C��0.1 mol/L ��NaHCO3��Һ��0.3 mol/L ��Ba��OH��2��Һ�������ϣ�������Һ�е�����Ϊ0.1mol/L ��Ba��OH��2��0.05mol/L ��NaOH��������Һ��һ�����ڣ�c ��OH-����c ��Ba2+����c ��Na+����c ��H+������C��ȷ��
D��������ͼ��ˮ�ĵ����Ӱ���֪����pH=3��CH3COOH��Һ�У�ˮ�ĵ��밴��Һ�����������Ӽ��㣬��Ũ��Ϊ10-11mol/L��pH=11��NaOH��Һ�У�ˮ�ĵ��밴�����Ӽ��㣬��Ũ��Ϊ10-11mol/L����D��ȷ��
��ѡB��
B��������ˮ��Ӧ��������ʹ����ᣬ����ǿ����ʣ�����Һ����ȫ���룬����������������ʣ�����Һ�в��ֵ��룬��������Һ����c��H+����c��Cl-����c��ClO-����c��OH-������B����
C��0.1 mol/L ��NaHCO3��Һ��0.3 mol/L ��Ba��OH��2��Һ�������ϣ�������Һ�е�����Ϊ0.1mol/L ��Ba��OH��2��0.05mol/L ��NaOH��������Һ��һ�����ڣ�c ��OH-����c ��Ba2+����c ��Na+����c ��H+������C��ȷ��
D��������ͼ��ˮ�ĵ����Ӱ���֪����pH=3��CH3COOH��Һ�У�ˮ�ĵ��밴��Һ�����������Ӽ��㣬��Ũ��Ϊ10-11mol/L��pH=11��NaOH��Һ�У�ˮ�ĵ��밴�����Ӽ��㣬��Ũ��Ϊ10-11mol/L����D��ȷ��
��ѡB��
���������⿼������Ũ�ȵĴ�С�Ƚϣ��Ѷ��еȣ�����Ũ�ȴ�С�Ƚ�Ҫ���ǵ���ʵĵ��롢����ˮ�⡢ˮ�ĵ��롢����غ㡢�����غ㡢���Ӻ��ʽ���ڻ����Һ�л�Ҫע��������֮��ķ�Ӧ��

��ϰ��ϵ�д�
 ����ѧ��Ӧ�����ϵ�д�
����ѧ��Ӧ�����ϵ�д�
�����Ŀ
�����������Ҵ������顢��ϩ������ɫ��Һ��һ���Լ��ǣ�������
| A�������� | B����ˮ |
| C�������� | D������������Һ |
����ʵ�飬�ڵμ��й��Լ�֮ǰ�����ȼӼ�Һʹ��Һ����ǣ�������
| A������������ˮ��ˮԡ�м���ˮ�� |
| B���屽ˮ��μ���������Һ����ˮ������е������� |
| C������ˮ��μ�����������ͭ����Һ����ˮ������е������� |
| D������������ˮ���μ�������Һ������ȩ���Ĵ��� |
Ϊ�˳�ȥ�����е�CaCl2��MgCl2��Na2SO4����ɳ���ɽ���������ˮ��ͨ�����¼���ʵ�鲽�裬���Ƶô�����ʳ��ˮ���ټ����Թ�����Na2CO3��Һ���ڼ����Թ�����NaOH��Һ���ۼ����Թ�����BaCl2 ��Һ���ܵ���ϡ�����������ݲ������ݹ��ˣ�����ȷ�IJ���˳���ǣ�������
| A���ݢۢڢ٢ݢ� |
| B���ݢۢ٢ڢݢ� |
| C���ݢڢۢ٢ݢ� |
| D���ݢڢ٢ۢݢ� |
���Ե缫����������ʵ���Һ�����һ��ʱ�䣬�����������ӣ����Һ��pH�½����ǣ�������
| A��CuSO4 |
| B��NaCl |
| C��NaOH |
| D��H2SO4 |
ʵ�����ﲻͬ��ѧ�Լ��ı��淽��������ͬ����NaOH��Һ���ڴ���ʯ����NaCl��Һ��������4���Լ�ͨ�����Դ������ͼ��ʾ�IJ����Լ�ƿ�У������Լ�ƿ��˳�����Լ������ȷ���ǣ�������
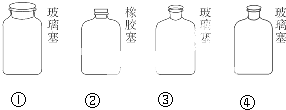

| A���٢ڢۢ� | B���ڢ٢ܢ� |
| C���ڢۢܢ� | D���ڢ٢ۢ� |
�����йط�Ӧ�ȵ�������ȷ���ǣ�������
| A����֪2H2��g��+O2��g���T2H2O��g����H=-483.6kJ/mol����������ȼ����Ϊ241.8kJ/mol |
| B��C��ʯī��s���TC�����ʯ��s����H��0�����Խ��ʯ��ʯī�ȶ� |
| C����֪̼��ȼ����ΪykJ/mol����7.2g��̼��12.8g��������ȼ�գ�����Ӧ��ľ����ų�xkJ��������������1mol̼��������Ӧ����1molһ����̼����ʱ��H=-��2.5x-0.5y��kJ/mol |
| D����֪2C��s��+2O2��g���T2CO2��g����H1��2C��s��+O2��g���T2CO��g����H2�����H1����H2 |

