��Ŀ����
������������ʽ����ƽ�������(N2H4)��Ϊȼ�ϣ�NO2��Ϊ����������Ӧ����N2��ˮ��������֪��N2(g) + 2O2(g) ��2NO2(g)�� ��H����67.7 kJ/mol
N2H4(g) + O2(g) �� N2(g) + 2H2O(g)�� ��H����534 kJ/mol
���й����º�NO2��Ӧ���Ȼ�ѧ����ʽ�У���ȷ����
| A��2N2H4(g) + 2NO2(g) �� 3N2(g) + 4H2O(l)����H����1135.7 kJ/mol |
| B��N2H4(g) + NO2(g) �� 3/2N2(g) + 2H2O(g)����H����567.85 kJ/mol |
| C��N2H4(g) + NO2(g) �� 3/2N2(g) + 2H2O(l)����H����1135.7 kJ/mol |
| D��2N2H4(g) + 2NO2(g) = 3N2(g) + 4H2O(g)����H��+1135.7 kJ/mol |
B
����

��ϰ��ϵ�д�
�����Ŀ
���꣬�ҹ��ں�����ҵ��ȡ������������Ŀ�ijɾͣ����۷ɴ���α�����ϵ
�л������̫�ա�
(1)������������ʽ����ƽ���������(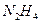 )��Ϊȼ�ϣ�
)��Ϊȼ�ϣ� ��Ϊ�ƽ�����
��Ϊ�ƽ�����
�� ����Ҫ��������ȼ�������ڹ���ʱ���������ɫ����
����Ҫ��������ȼ�������ڹ���ʱ���������ɫ����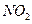 ���Ի�������
���Ի�������
����Ⱦ��Ϊ������Ⱦ��ʹ������ (����ĸ)����֮��
| A��Һ̬�� | B��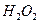 | C��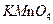 | D��Һ̬�� |
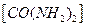 �ʹ������Ʒ�Ӧ������ȡ����(������
�ʹ������Ʒ�Ӧ������ȡ����(������ͬʱ����������)����÷�Ӧ�����ӷ���ʽ�� ��
(2)�ɴ������ڿ����ĸ��¹�������ͼ��ʾ��
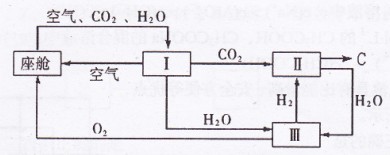
�������ڿ������¹��̿���ѭ�����õ�����Ϊ
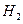 ��
��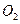 �� ��װ�â���
�� ��װ�â���������Ӧ�Ļ�ѧ����ʽΪ ��
�ڴ�װ��I����ɿ���
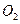 ����Դ�����Աÿ������35mol
����Դ�����Աÿ������35mol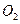 ��ÿ�����
��ÿ������������к�18 mol
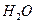 ��������������к�
��������������к�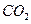 mol��
mol�� 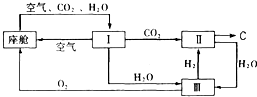 ���꣬�ҹ��ں�����ҵ��ȡ������������Ŀ�ijɾͣ����۷ɴ���α�����ϵ�л������̫�գ�
���꣬�ҹ��ں�����ҵ��ȡ������������Ŀ�ijɾͣ����۷ɴ���α�����ϵ�л������̫�գ�