��Ŀ����
����ʵ�������Ԥ��ʵ��Ŀ�Ļ���۾���ȷ���ǣ���������
| ѡ�� | ʵ����� | Ԥ��ʵ��Ŀ�Ļ���� |
| A | �����£���pH��ֽ�ⶨŨ��Ϊ0.1mol?L-1 Na2SiO3��Һ��Na2CO3��Һ��pH | �Ƚ�H2SiO3��H2CO3������ǿ�� |
| B | ij��Һ�м��������ữ���Ȼ�����Һ���а�ɫ�������� | ����Һ�к���SO42- |
| C | ��ij��Һ�м���2��KSCN��Һ����Һ���Ժ�ɫ��������Һ�м��뼸�����Ƶ���ˮ����Һ��Ϊ��ɫ | ����Һ��һ������Fe2+ |
| D | ��ij����ͨ����ۺ�KI�Ļ����Һ����Һ����ɫ | ������һ����Cl2 |
| A��A | B��B | C��C | D��D |
���㣺���ʵļ���ͼ����ʵ�鷽�����,��ѧʵ�鷽��������
ר�⣺���ʼ��������
������A����pH��ֽ�ⶨŨ��Ϊ0.1mol?L-1Na2SiO3��Һ��Na2CO3��Һ��pH�����ݷǽ���Ԫ����ۺ���������Խǿ��Ԫ�صķǽ�����Խǿ�����жϹ�����̼������ԣ�
B�����������ữ���Ȼ�����Һ���а�ɫ�������ɣ���������ΪAgCl�������ᱵ�����������ǿ�����ԣ�
C������2��KSCN��Һ����Һ���Ժ�ɫ��������Һ�м��뼸�����Ƶ���ˮ����Һ��Ϊ��ɫ�����������ӱ���ˮ�������������ӣ�
D��ij����ͨ����ۺ�KI�Ļ����Һ����Һ����ɫ����������������ԣ��������������ɵⵥ�ʣ�
B�����������ữ���Ȼ�����Һ���а�ɫ�������ɣ���������ΪAgCl�������ᱵ�����������ǿ�����ԣ�
C������2��KSCN��Һ����Һ���Ժ�ɫ��������Һ�м��뼸�����Ƶ���ˮ����Һ��Ϊ��ɫ�����������ӱ���ˮ�������������ӣ�
D��ij����ͨ����ۺ�KI�Ļ����Һ����Һ����ɫ����������������ԣ��������������ɵⵥ�ʣ�
���
A����pH��ֽ�ⶨŨ��Ϊ0.1mol?L-1Na2SiO3��Һ��Na2CO3��Һ��pH�����жϹ�����̼��������ӵ����ԣ�pHԽ��Ӧ������Խ������A��ȷ��
B�����������ữ���Ȼ�����Һ���а�ɫ�������ɣ���������ΪAgCl�������ᱵ�����������ǿ�����ԣ���ԭ��Һ�п��ܴ��������ӣ�����������ӡ�����������ӣ���B����
C������2��KSCN��Һ����Һ���Ժ�ɫ��������Һ�м��뼸�����Ƶ���ˮ����Һ��Ϊ��ɫ�����������ӱ���ˮ�������������ӣ����Ը���Һ��һ������Fe2+����C��ȷ��
D��ij����ͨ����ۺ�KI�Ļ����Һ����Һ����ɫ����������������ԣ��������������ɵⵥ�ʣ�������������Cl2����NO2�ȣ���D����
��ѡAC��
B�����������ữ���Ȼ�����Һ���а�ɫ�������ɣ���������ΪAgCl�������ᱵ�����������ǿ�����ԣ���ԭ��Һ�п��ܴ��������ӣ�����������ӡ�����������ӣ���B����
C������2��KSCN��Һ����Һ���Ժ�ɫ��������Һ�м��뼸�����Ƶ���ˮ����Һ��Ϊ��ɫ�����������ӱ���ˮ�������������ӣ����Ը���Һ��һ������Fe2+����C��ȷ��
D��ij����ͨ����ۺ�KI�Ļ����Һ����Һ����ɫ����������������ԣ��������������ɵⵥ�ʣ�������������Cl2����NO2�ȣ���D����
��ѡAC��
���������⿼�黯ѧʵ�鷽�������ۣ�Ϊ��Ƶ���㣬�漰���Լ��ǽ����ԱȽϡ����Ӽ��鼰������ԭ��Ӧ�ȣ��������ʵ����ʼ���ѧ��Ӧԭ��Ϊ���Ĺؼ����ۺ��Խ�ǿ����Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
��NAΪ�����ӵ�������ֵ��������˵����ȷ���ǣ�������
| A����⾫��ͭʱ���������õ�������Ϊ2NA����������64g |
| B��200mLij����ǿ�����к���1.5NA��SO42-���ӣ�ͬʱ����NA�����������ӣ��������ʵ���Ũ����2.5mol/L |
| C�����³�ѹ��78g Na2O2������������������������Ϊ4NA |
| D��һ��������������Fe����Ũ���ᷴӦ��ת�Ƶ�����һ��Ϊ2NA |
�����и��ֵ���Ͷ���ͨ��CuSO4��Һ�У��ܲ���ͭ���ʵ��ǣ�������
| A��Fe | B��Na |
| C��H2 | D��Ag |
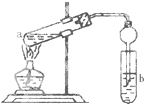 ��ͼ�����Թ�a���ȼ���3mL 95%���Ҵ�����ҡ����������2mLŨH2SO4�����ҡ�ȣ���ȴ���ټ���2mL�����ᣬ�ò�������ֽ�����Թ̶ܹ�������̨�ϣ����Թ�b�м���7mL����̼������Һ�����Ӻ�װ�ã��þƾ��ƶ��Թ�a���ȣ����۲쵽�Թ�b������������ʱֹͣʵ�飮
��ͼ�����Թ�a���ȼ���3mL 95%���Ҵ�����ҡ����������2mLŨH2SO4�����ҡ�ȣ���ȴ���ټ���2mL�����ᣬ�ò�������ֽ�����Թ̶ܹ�������̨�ϣ����Թ�b�м���7mL����̼������Һ�����Ӻ�װ�ã��þƾ��ƶ��Թ�a���ȣ����۲쵽�Թ�b������������ʱֹͣʵ�飮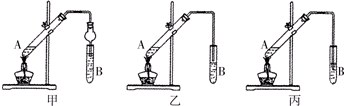
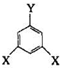 ��ʾ������X��Y����ΪH�������ܷ���������Ӧ��������
��ʾ������X��Y����ΪH�������ܷ���������Ӧ��������