��Ŀ����
������������ȷ����(�� ��)
�ٱ�״���£�1 L HCl��1 L H2O�����ʵ�����ͬ
�ڱ�״���£�1 g H2��14 g N2�������ͬ
��28 g CO�����Ϊ22.4 L
���������ʵ����ʵ�����ͬ���������ڱ�״���µ����Ҳ��ͬ
����ͬ��ͬ���ʱ���������ʵ����ʵ���Խ����ѹǿԽ��
��ͬ��ͬѹ�£�������ܶ����������Է�������������
A���٢ڢۡ�����B���ڢݢ� C���ڢۢ� D���ܢݢ�
B
��������
����������ٱ�״����H2O��Һ̬������ʹ������Ħ����������ڱ�״���£�1 g H2��14 g N2�����ʵ�����ͬ�����ڶ��ߵ�״̬�������壬������ǵ������ͬ����ȷ����28 g CO������ڱ�״�������ԼΪ22.4 L ����������ȷ������Ĵ�С�������������ʵ����ʵ�����ͬ�������ǵ�״̬���������壬�������ڱ�״���µ����Ҳ��һ����ͬ��������ͬ��ͬ���ʱ���������ʵ����ʵ���Խ��λ���������ķ�������Խ�࣬��ѹǿԽ����ȷ����ͬ��ͬѹ�£�������ܶ����������Է������������ȣ���ȷ��
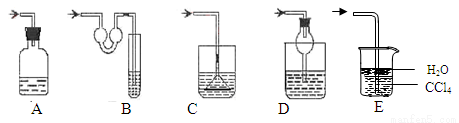
���㣺�������ʵ���������٤�����ɺ������۵�Ӧ�õ�֪ʶ��

��ϰ��ϵ�д�
�����Ŀ
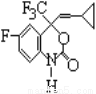
 NH4+ +OH��
NH4+ +OH�� Cu2+ +2Cl��
Cu2+ +2Cl��
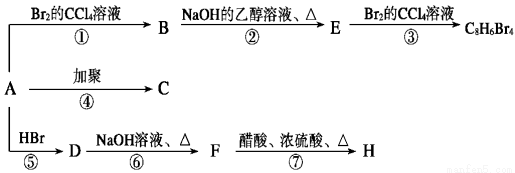
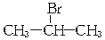 (��Ҫ����),1 molij������A���ȼ�պ���Եõ�8 mol CO2��4 mol H2O������A�ڲ�ͬ�������ܷ�����������ʾ��һϵ�б仯��
(��Ҫ����),1 molij������A���ȼ�պ���Եõ�8 mol CO2��4 mol H2O������A�ڲ�ͬ�������ܷ�����������ʾ��һϵ�б仯��