��Ŀ����
����С���������ʵ�飺ȡһ������Ϊ200mL�Ĵ�ע����A����ע��������Ͳ��Ԥ��ʢ��0.300g��Ư�۾���Ȼ���������������ᣬ��Ӧ��ֺ�ע�����ڵ�Һ��С�ĵ��Ƴ�����ʱע�����ڲ�������������ۺ�Ϊ��״���������44.8mL���ش�
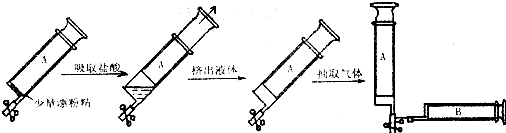
��1����Bע������Aע��������Ƥ���ϳ�ȡ�ۺ�Ϊ��״���µ�����4.48mL��������ˮ���ɣ���Bע����������ǡ��ȫ������ˮ��ʱ���γ���ˮ�����ԼΪ mL������ʱ���������ܽ��0.710g/100gˮ���������ˮ���ܶ�Ϊ1.00g/cm3����
��2��Ư�۾���NaClO��NaCl�Ļ��������ʵ���֮����1��1������Ư�۾�ʱ�������м��������ռ���࣬��������Ư�۾��г�NaClO��NaCl֮������ʺ���Ϊ g��
��3����������ˮ��ʱ����������Bע�����ڳ�����������Լ mL��

��1����Bע������Aע��������Ƥ���ϳ�ȡ�ۺ�Ϊ��״���µ�����4.48mL��������ˮ���ɣ���Bע����������ǡ��ȫ������ˮ��ʱ���γ���ˮ�����ԼΪ
��2��Ư�۾���NaClO��NaCl�Ļ��������ʵ���֮����1��1������Ư�۾�ʱ�������м��������ռ���࣬��������Ư�۾��г�NaClO��NaCl֮������ʺ���Ϊ
��3����������ˮ��ʱ����������Bע�����ڳ�����������Լ
���㣺��ѧ����ʽ���йؼ���,�ȡ��塢�⼰�仯������ۺ�Ӧ��,�йػ���ﷴӦ�ļ���
ר�⣺������
��������1������n=
�����������ʵ������ٸ���m=nM������������������ܽ�ȼ���ˮ������������������Һ�������ٸ���V=
������Һ�����
��2����������������Ӧ��ClO-+Cl-+2H+=Cl2��+H2O�������������ʵ��������ݷ���ʽ����NaClO�����ʵ������ٸ���m=nM����Ư����NaClO��NaCl��������������������NaOH��������
��3����ʱ�������ˮ�������ܷ�ӦΪ��2Cl2+2H2O=4HCl+O2������������������������������
| V |
| Vm |
| m |
| �� |
��2����������������Ӧ��ClO-+Cl-+2H+=Cl2��+H2O�������������ʵ��������ݷ���ʽ����NaClO�����ʵ������ٸ���m=nM����Ư����NaClO��NaCl��������������������NaOH��������
��3����ʱ�������ˮ�������ܷ�ӦΪ��2Cl2+2H2O=4HCl+O2������������������������������
���
�⣺��1���������ʵ���Ϊ
=2.0��10-4mol����������Ϊ2.0��10-4mol��71g/mol=0.0142g����Ҫˮ������Ϊ
=2g����Һ����Ϊ2g+0.0142g=2.0142g����Һ���Ϊ
=2.01mL��
�ʴ�Ϊ��2.01��
��2�������������ʵ���Ϊ
=0.002mol����ClO-+Cl-+2H+=Cl2��+H2O����֪n��NaClO��=n��Cl2��=0.002mol��Ư�۾���NaClO��NaCl�Ļ��������ʵ���֮����1��1����n��NaCl��=0.002mol�����ʵ�����Ϊ0.300g-0.002mol����74.5g/mol+58.5g/mol��=0.0340g��
�ʴ�Ϊ��0.0340��
��3����ʱ�������ˮ�������ܷ�ӦΪ��2Cl2+2H2O=4HCl+O2����V��O2��=
V��Cl2��=
��4.48mL=2.24mL��
�ʴ�Ϊ��2.24��
| 0.00448L |
| 22.4L/mol |
| 0.0142g |
| 0.710g/100g |
| 2.0142g |
| 1.00g/mL |
�ʴ�Ϊ��2.01��
��2�������������ʵ���Ϊ
| 0.0448L |
| 22.4L/mol |
�ʴ�Ϊ��0.0340��
��3����ʱ�������ˮ�������ܷ�ӦΪ��2Cl2+2H2O=4HCl+O2����V��O2��=
| 1 |
| 2 |
| 1 |
| 2 |
�ʴ�Ϊ��2.24��
���������⿼����ҺŨ�ȼ��㡢�������㡢��ѧ����ʽ����ȣ��Ƕ�ѧ���ۺ������Ŀ��飬��3����ע�������ܷ�Ӧ����ʽ����Ѷ��еȣ�

��ϰ��ϵ�д�
 ���Ž�����ٰθ��νӹ㶫���������ϵ�д�
���Ž�����ٰθ��νӹ㶫���������ϵ�д� �����������ҵ�������������ϵ�д�
�����������ҵ�������������ϵ�д� ѧ���������ν��Ͼ���ѧ������ϵ�д�
ѧ���������ν��Ͼ���ѧ������ϵ�д�
�����Ŀ
���¸��ֱ�ʾʽ�У����ܷ�ӳ��ԭ�Ӻ�������˶�״̬���ǣ�������
A��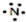 |
B��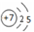 |
| C��1s22s22p3 |
D��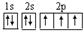 |
���б仯���������仯���ǣ�������
| A��ʯ�ͷ��� | B��ʯ���ѻ� |
| C��ú���� | D��ʯ��绯 |
��һ��������þ��ͭ�Ͻ���뵽ϡ������Һ�У�����ǡ����ȫ��Ӧ��������NO����4.48L����״��������������Һ�м������ʵ���Ũ��Ϊ3mol?L-1��NaOH��Һ��������ȫ���������й������в���ȷ���ǣ�������
| A����ʼ����Ͻ�����ʵ���֮��Ϊ0.3mol |
| B����Ӧ�б���ԭ��δ����ԭ���������ʵ���֮��Ϊ3��1 |
| C��������ȫʱ����NaOH��Һ�����Ϊ200mL |
| D�����ɳ�����������ԭ�Ͻ����������10.2g |
 ���Ⱥͷ�̪��ˮ�к��л��ܼ��е��ܽ�Ȳ�ͬ�����ܼ�����ֽ������������ʱ�����Ⱥͷ�̪���Բ�ͬ����������ֽ���ƶ����Ӷ��ﵽ�����Ŀ�ģ�ijУ��ѧ��ȤС����ͨ����ԭ�������Ⱥͷ�̪�ӻ����ҺA�з��뿪����
���Ⱥͷ�̪��ˮ�к��л��ܼ��е��ܽ�Ȳ�ͬ�����ܼ�����ֽ������������ʱ�����Ⱥͷ�̪���Բ�ͬ����������ֽ���ƶ����Ӷ��ﵽ�����Ŀ�ģ�ijУ��ѧ��ȤС����ͨ����ԭ�������Ⱥͷ�̪�ӻ����ҺA�з��뿪����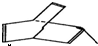 �л���X�Ľṹ�ü���ʽ��ʾ��ͼ
�л���X�Ľṹ�ü���ʽ��ʾ��ͼ