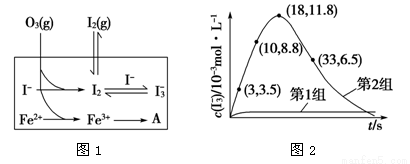
��Ŀ����
����ͼ���ס��ҡ����ֱ��ʾ�ڲ�ͬ�����¿��淴Ӧ��A(g)+B(g)  xC(g)�������� C�ڷ�Ӧ������еİٷֺ�����C%���ͷ�Ӧʱ�䣨t���Ĺ�ϵ��
xC(g)�������� C�ڷ�Ӧ������еİٷֺ�����C%���ͷ�Ӧʱ�䣨t���Ĺ�ϵ��
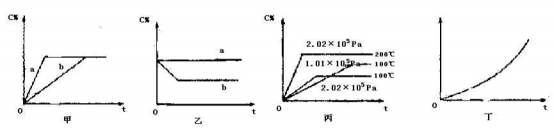
��1������ͼ���������߷ֱ�����д������������������ ���߱�ʾ����ʱ����������ʣ�a b������ڡ�С�ڻ���ڣ�
��2������ͼ��ʾ��Ӧ�ﵽƽ��ֱ��ں��º�ѹ�����ºͺ��º�����������ƽ���������г���He������������ ���߱�ʾ���º��ݵ��������ʱ�ú��º�����C% ������С�䣩
��3�����ݱ�ͼ�����жϸÿ��淴Ӧ������Ӧ�� ��Ӧ������ȡ����ȣ���������x��ֵ�� ��
��4����ͼ��ʾ��ij�̶��������ܱ������У��������淴Ӧ�ﵽƽ���ij�����������¶ȣ�T���ı仯���������������⣬��ͼ������������� ��[���C% ��A��ת���� ��B��ת���� ��ѹǿ ��c��A�� ��c(B)]������ƽ���ƶ��ķ���Ϊ ��������ƻ����ƣ�
21. ��1�� b ���� ��2�� a ���� ��3������ X>2 ��4����, �� , ��. �� ����
��������
�����������1�������ı䷴Ӧ���̣���߷�Ӧ���ʣ����̵���ƽ���ʱ�䣬a���ߵ���ƽ��ʱ��̣�b���ߵ���ƽ��ʱ�䳤������a���߱�ʾʹ�ô�����b����δʹ�ô��������ʣ�a����b��Ϊ��b�����ڣ�2�����º����£����벻��Ӧ�����壬��Ӧ��������ֵ�Ũ�Ȳ��䣬ƽ�ⲻ�ƶ�����Ӧ��������ֵĺ������䣮��ͼ��֪��a����C�ĺ������䣬b���ߵ�����ƽ���C�ĺ������ͣ�����a���߱�ʾ���º�����������ƽ���������г���He������Ϊ��a�����䣻��3����ͼ��֪��ѹǿ��ͬʱ���¶�Խ�ߣ�C�ĺ���Խ�ߣ��������¶ȣ�ƽ��������Ӧ�����ƶ��������¶ȣ�ƽ�������ȷ����ƶ�����������ӦΪ���ȷ�Ӧ���¶���ͬ��ѹǿԽ��ƽ��ʱC�ĺ���Խ�ͣ�������ѹǿ��ƽ�����淴Ӧ�����ƶ�������ѹǿƽ���������С�ķ����ƶ�����x��2��xΪ����������Ϊ�����ȣ�x��2��xΪ��������
��4���÷�Ӧ����ӦΪ���ȷ�Ӧ����Ӧǰ�������������������¶ȣ�ƽ�������ȷ����ƶ����������ƶ�����ͼ��֪����������ʾ�������¶����߶����������¶ȣ�ƽ�������ȷ����ƶ����������ƶ���C�ĺ�������A��B��ת��������ƽ��ʱA��BŨ�Ƚ��ͣ���Ӧǰ���������������ں��������£������¶ȣ�ƽ�������ȷ����ƶ����������ƶ���������ܵ����ʵ������ӣ�����ƽ��ʱ��������ѹǿ���٢ڢܷۢ��ϣ���Ϊ���٢ڢۢܣ����ơ�
���㣺�������������ƽ��״̬��Ӱ���Լ�ͼ��ʶ��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�


 (aq)����ƽ�ⳣ������ʽΪ________��
(aq)����ƽ�ⳣ������ʽΪ________��